What is in this leaflet
This leaflet answers some common questions about MENVEO.
It does not contain all the available information. It does not take the place of talking to your doctor or pharmacist.
The information in this leaflet was last updated on the date listed on the final page. More recent information on the medicine may be available.
You should ensure that you speak to your pharmacist or doctor to obtain the most up-to-date information on the medicine.
Those updates may contain important information about the medicine and its use of which you should be aware.
All medicines, including vaccines, have risks and benefits. Your doctor has weighed the risks of you or your child having MENVEO against the benefits they expect it will have for you.
If you have any concerns about this vaccine, ask your doctor, nurse or pharmacist.
Keep this leaflet. You may need to read it again.
What MENVEO is used for
MENVEO is a vaccine used for active immunisation of children (from 2 months of age), adolescents and adults to prevent invasive disease caused by Neisseria meningitidis serogroups A, C, W-135 and Y. The use of this vaccine should be in accordance with the official recommendations.
Meningitis is an infection of the spinal fluid and the tissues that surround the brain. Of the microbes that cause meningitis, one of the most severe is Neisseria meningitidis (meningococcus). Meningococcal meningitis is an infectious disease that is spread from person to person. It is a very serious disease that is sometimes fatal, and may cause permanent physical and neurological damage among survivors.
Please note that MENVEO can only protect you/ your child from meningitis caused by four groups of Neisseria meningitidis (A, C, W-135 and Y).
It will not protect you/ your child from Meningitis caused by other groups of Neisseria meningitidis, or meningitis caused by different kinds of microbes.
How it works
MENVEO works by causing your body to produce its own protection (or antibodies), against meningococcal bacteria. Your body usually takes a couple of weeks after vaccination to develop protection against Neisseria meningitidis.
If a vaccinated person comes into contact with Neisseria meningitidis, the body is usually able to destroy it. However, as with all vaccines, 100% protection cannot be guaranteed.
As with all vaccines, occasionally, individuals may react unfavourably to the vaccine.
The chance of a severe reaction from MENVEO is very small, but the risks from not being vaccinated against meningococcal disease may be very serious.
MENVEO should not be given to a person who has:
- Ever had an allergic reaction to MENVEO or any of the ingredients listed at the end of this leaflet.
- Ever had an allergic reaction to diphtheria toxoid (a substance used in a number of other vaccines)
- A very high fever.
In this case vaccination with MENVEO may have to be delayed. Please discuss this with your doctor. - Ever had an allergic reaction or became unwell after receiving any other meningococcal vaccine.
- A severe infection with a high temperature.
A minor infection such as a cold should not be a problem, but talk to your doctor, nurse or pharmacist about this before being vaccinated.
In addition, MENVEO should not be administered if:
- the expiry date printed on the pack has passed
- the packaging is torn or shows signs of tampering.
If you are not sure whether you or your child should have MENVEO talk to your doctor, nurse or pharmacist.
This medicine should be administered only to the person for whom it was prescribed.
Before You Receive MENVEO:
Tell your doctor or pharmacist if you have/your child has allergies to:
- any other medicines
- latex
Tell your doctor if you have/your child has had any of the following reactions after other vaccinations:
- life-threatening allergic reaction
- difficulty breathing
- swelling of the throat
- fits (convulsions), including fits associated with fever
- high fevers
- severe reactions at the injection site.
Tell your doctor if you have/your child has an infection or high temperature. Your doctor may decide to delay vaccination until the illness has passed. A mild illness, such as a cold, is not usually a reason to delay vaccination.
Fainting, feeling faint or other stress-related reactions can occur as a response to any needle injection.
Tell your doctor or nurse if you have experienced this kind of reaction previously.
Tell your doctor if you or your child have or have had any medical conditions especially the following:
- an immune deficiency condition
- allergies or allergic reaction
- a low blood platelet count
Tell your doctor if you are pregnant or intend to become pregnant soon. Your doctor will discuss the possible risks and benefits of receiving MENVEO vaccine during pregnancy.
Tell your doctor if you are breastfeeding. Your doctor will discuss the possible risks and benefits of receiving MENVEO vaccine during breastfeeding.
Tell your doctor if you receive treatment that blocks the part of the immune system known as complement activation, such as eculizumab. Even if you have been vaccinated with MENVEO you remain at increased risk of disease caused by the Neisseria meningitidis groups A, C, W-135 and Y bacteria.
Important information about some of the ingredients of MENVEO:
Tell your doctor or pharmacist if you /your child) are taking any other medicines, including any that you buy without a prescription from your pharmacy, supermarket or health food shop.
Having other vaccines
Tell your doctor if you/your child have had any vaccines recently. MENVEO may be given at the same time as other vaccinations.
Other injected vaccines must be given into a different arm or leg (children 2-23 months) from the site of the MENVEO injection. These include:
- Tetanus, Reduced Diphtheria and Acellular Pertussis Vaccine,
- Human Papillomavirus Vaccine
Your doctor or pharmacist may have more information on medicines and vaccines that require special precautions or should be avoided during vaccination with MENVEO.
How MENVEO is given
MENVEO is given as an injection, usually into your/your child’s upper arm muscle or for children under 2 years of age into the thigh muscle.
MENVEO should not be injected directly into a blood vessel.
MENVEO is for single use in one patient only.
If you have any concerns about how this vaccine is to be given, talk to your doctor, nurse or pharmacist.
How much is given
The dose is 0.5 mL given by injection.
You should keep a record of you/your child's vaccinations
Side effects
Tell your doctor or pharmacist as soon as possible if you or your child do not feel well after having MENVEO.
After being vaccinated with MENVEO, your doctor or pharmacist may ask you/your child to remain for observation for a few minutes.
MENVEO may have unwanted side effects in a few people. All medicines, including vaccines, can have side effects. Sometimes they are serious. However, most of the time, they are not.
The most common side effects include the following:
- redness, firmness, and/or pain at the injection site
- fever, chills, malaise
- rash
- headaches
- muscle ache, joint ache
- nausea
These side effects are usually mild, self limited, and do not require any specific treatments. Other side effects that have been reported during marketed use include:
- impaired hearing, ear pain, spinning sensation
- injection site itching, inflammation, swelling, including extensive swelling of the injected limb, tiredness
- enlarged lymph nodes near injection site
- allergic reactions, severe allergic reaction that may include swelling of the lips, mouth, throat (which may cause difficulty in swallowing), difficulty breathing with wheezing or coughing, rash and swelling of the hands, feet and ankles, loss of consciousness, very low blood pressure.
- fall, head injury
- increased liver enzymes on blood test results, body temperature increased
- bone pain
- dizziness, balance disorder, faint, seizure, facial nerve paralysis, upper eyelid fall
- sore throat
- raised bumps of fluid on the skin
As with all vaccines given by injection there is a very small risk of serious allergic reaction.
If someone who has just been vaccinated appears to be having a severe allergic reaction to the vaccine, medical help should be obtained immediately. These include difficulty breathing or hives.
Other side effects not listed above may occur in some patients.
Tell your doctor or pharmacist if you notice anything that is making you or your child feel unwell.
Some of the side effects may temporarily affect your ability to drive or use machinery.
Ask your doctor or pharmacist to answer any questions you may have.
Storing MENVEO
- Keep MENVEO in the original pack until it is time for it to be given.
- Keep it in the refrigerator, between 2°C and 8°C.
- Do not freeze MENVEO.
- Following reconstitution, to reduce microbiological hazard the product should be used as soon as practicable after reconstitution. If storage is necessary, hold at 2-8°C for not more than 24 hours.
- Do not use MENVEO after the expiry date which is stated on the carton.
The vials left over after administration of MENVEO must be disposed of as medical waste.
Product description
What it looks like
MENVEO is a powder with liquid for reconstitution.
Each dose of MENVEO is supplied as a:
- Vial containing the MenA lyophilised conjugate component as a white to off-white powder
- Vial containing the MenCWY liquid conjugate component as a clear solution
The contents of the two components (MenA vial and MenCWY vial) are to be mixed prior to vaccination.
The other ingredients in the vial of MenA lyophilised conjugate component are: potassium dihydrogen phosphate and sucrose.
The other ingredients in the vial of MenCWY liquid conjugate component are: sodium chloride, sodium phosphate - monobasic monohydrate; dibasic sodium phosphate dihydrate and water for injections
The MENVEO vial/vial presentation is supplied either as a single dose pack (2 vials) or 5 doses multi-pack (10 vials).
Not all pack sizes may be marketed.
MENVEO does not contain lactose, gluten, thiomersal (organic mercurials), tartrazine or any other azo dyes.
Sponsor (license holder):
GlaxoSmithKline Australia Pty Ltd
Level 4,
436 Johnston Street,
Abbotsford, Victoria, 3067
AUST R 192696 - (vial / vial pack)
Date of preparation:
14 January 2020
Trademarks are owned by or licensed to the GSK group of companies.
© 2019 GSK group of companies or its licensor.
Version: 6.0
Published by MIMS May 2020







 In study V59P14, children 2 months of age at enrolment were administered either 3 doses at 2, 6 and 12 months of age or 4 doses at 2, 4, 6 and 18 months of age, and were assessed for immune response at 7 months of age. Among 284 infants who received doses at 2 and 6 months, 74%, 94%, 99%, 97% had hSBA ≥ 1:8 against serogroups A, C, W-135 and Y, respectively, compared to 89%, 97%, 98%, and 98% of 277 infants who received doses at 2, 4, and 6 months. Pre-specified non-inferiority criteria were met for serogroups C, W-135, and Y.
In study V59P14, children 2 months of age at enrolment were administered either 3 doses at 2, 6 and 12 months of age or 4 doses at 2, 4, 6 and 18 months of age, and were assessed for immune response at 7 months of age. Among 284 infants who received doses at 2 and 6 months, 74%, 94%, 99%, 97% had hSBA ≥ 1:8 against serogroups A, C, W-135 and Y, respectively, compared to 89%, 97%, 98%, and 98% of 277 infants who received doses at 2, 4, and 6 months. Pre-specified non-inferiority criteria were met for serogroups C, W-135, and Y. A 2-dose series was also examined in a study of Latin American children, who received Menveo at 12 and 16 months of age. Among the per protocol population of 106 subjects, the proportions of subjects with hSBA ≥ 1:8 for serogroups A, C, W-135 and Y were 97% (92-99), 100% (96-100), 100% (96-100), and 100% (96-100), respectively.
A 2-dose series was also examined in a study of Latin American children, who received Menveo at 12 and 16 months of age. Among the per protocol population of 106 subjects, the proportions of subjects with hSBA ≥ 1:8 for serogroups A, C, W-135 and Y were 97% (92-99), 100% (96-100), 100% (96-100), and 100% (96-100), respectively. In the same study, a separate group of children, 2 through 5 years of age (N=297) in the per protocol population were immunised with two doses of Menveo, two months apart. The observed seroresponse rates (with 95% CI) at 1 month after the second dose were: 91% (87-94), 98% (95-99), 89% (85-92), and 95% (91-97) for serogroups A, C, W-135 and Y, respectively. The proportion of subjects with hSBA ≥ 1:8 (95% CI) were 91% (88-94), 99% (97-100), 99% (98-100), and 98% (95-99) for serogroups A, C, W-135 and Y, respectively. The hSBA GMTs (95% CI) for this group were 64 (51-81), 144 (118-177), 132 (111-157), and 102 (82-126) for serogroups A, C, W-135 and Y, respectively.
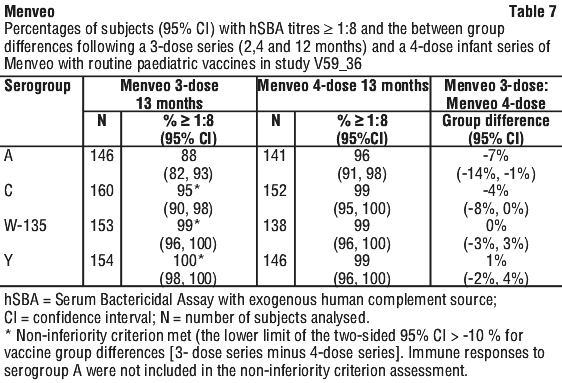
In the same study, a separate group of children, 2 through 5 years of age (N=297) in the per protocol population were immunised with two doses of Menveo, two months apart. The observed seroresponse rates (with 95% CI) at 1 month after the second dose were: 91% (87-94), 98% (95-99), 89% (85-92), and 95% (91-97) for serogroups A, C, W-135 and Y, respectively. The proportion of subjects with hSBA ≥ 1:8 (95% CI) were 91% (88-94), 99% (97-100), 99% (98-100), and 98% (95-99) for serogroups A, C, W-135 and Y, respectively. The hSBA GMTs (95% CI) for this group were 64 (51-81), 144 (118-177), 132 (111-157), and 102 (82-126) for serogroups A, C, W-135 and Y, respectively. Antibody persistence at 5 years after primary vaccination was assessed in the extension study V59P20E1. There was substantial antibody persistence observed against serogroups C, W and Y, with the percentages of subjects with hSBA ≥ 1:8 being 32% and 56% against serogroup C in subjects 2-5 and 6-10 years of age, respectively, 74% and 80% against serogroup W, and 48% and 53% against serogroup Y. GMTs were respectively 6.5 and 12 for serogroup C, 19 and 26 for serogroup W, and 8.13 and 10 for serogroup Y. For serogroup A, 14% and 22% of subjects 2-5 and 6-10 years of age, respectively, had hSBA ≥ 1:8 (GMTs 2.95 and 3.73). Levels for all serogroups were higher than those seen in meningococcal vaccine-naïve children of similar ages. The children also received a booster dose of Menveo, 5 years after a single dose primary vaccination. All subjects in both age groups had hSBA ≥ 1:8 across serogroups, with antibody titres several fold higher than seen after the primary vaccination (Table 11).
Antibody persistence at 5 years after primary vaccination was assessed in the extension study V59P20E1. There was substantial antibody persistence observed against serogroups C, W and Y, with the percentages of subjects with hSBA ≥ 1:8 being 32% and 56% against serogroup C in subjects 2-5 and 6-10 years of age, respectively, 74% and 80% against serogroup W, and 48% and 53% against serogroup Y. GMTs were respectively 6.5 and 12 for serogroup C, 19 and 26 for serogroup W, and 8.13 and 10 for serogroup Y. For serogroup A, 14% and 22% of subjects 2-5 and 6-10 years of age, respectively, had hSBA ≥ 1:8 (GMTs 2.95 and 3.73). Levels for all serogroups were higher than those seen in meningococcal vaccine-naïve children of similar ages. The children also received a booster dose of Menveo, 5 years after a single dose primary vaccination. All subjects in both age groups had hSBA ≥ 1:8 across serogroups, with antibody titres several fold higher than seen after the primary vaccination (Table 11).
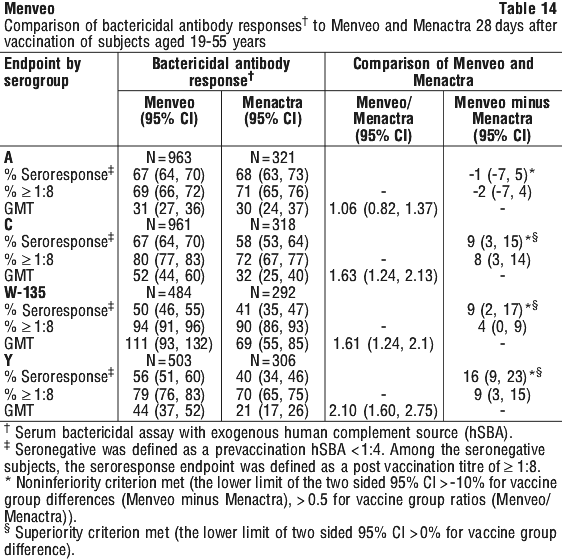
 In the non-inferiority study, V59P6, immunogenicity was assessed among adolescents aged 11-17 years who had been randomised to receive either Menveo or Menomune (Sanofi Pasteur Ltd), a quadrivalent meningococcal polysaccharide vaccine. Responses to Menveo were shown to be non-inferior to those to Menomune for all four serogroups (A, C, W and Y) based on seroresponse, proportions achieving hSBA titres ≥ 1:8, and GMTs. At one-year post vaccination, the percentage of Menveo recipients with hSBA ≥ 1:8 remained significantly higher compared with Menomune recipients for serogroups C, W and Y, and similar between the two study groups for serogroup A (Table 13).
In the non-inferiority study, V59P6, immunogenicity was assessed among adolescents aged 11-17 years who had been randomised to receive either Menveo or Menomune (Sanofi Pasteur Ltd), a quadrivalent meningococcal polysaccharide vaccine. Responses to Menveo were shown to be non-inferior to those to Menomune for all four serogroups (A, C, W and Y) based on seroresponse, proportions achieving hSBA titres ≥ 1:8, and GMTs. At one-year post vaccination, the percentage of Menveo recipients with hSBA ≥ 1:8 remained significantly higher compared with Menomune recipients for serogroups C, W and Y, and similar between the two study groups for serogroup A (Table 13).