1 Name of Medicine
Meropenem (as trihydrate).
2 Qualitative and Quantitative Composition
Meropenem-AFT powder for intravenous injection or infusion contains meropenem trihydrate equivalent to meropenem, 500 mg or 1 g, blended with sodium carbonate.
Excipients with known effects.
Meropenem-AFT powder for intravenous injection or infusion contains 208 mg sodium carbonate for each gram of meropenem. One vial of 500 mg contains 45 mg of sodium and one vial of 1 g contains 90 mg of sodium.
For the full list of excipients, see Section 6.1 List of Excipients.3 Pharmaceutical Form
White to light yellow crystalline powder for injection.
4.1 Therapeutic Indications
Meropenem is indicated for treatment of the following infections, in adults and children (aged 3 months and over), when the causative organisms are known or suspected to be resistant to commonly used antibiotics: community acquired lower respiratory tract infection; hospital acquired lower respiratory tract infection; complicated urinary tract infection; febrile neutropenia; intra-abdominal and gynaecological (polymicrobial) infections; complicated skin and skin structure infections; meningitis; septicaemia.
4.2 Dose and Method of Administration
Adults.
Usual dose. 500 mg to 1 g by intravenous administration every 8 hours depending on type and severity of infection, the known or suspected susceptibility of the pathogen(s) and the condition of the patient.
Exceptions.
1. Febrile episodes in neutropenic patients - the dose should be 1 g every 8 hours.
2. Meningitis - the dose should be 2 g every 8 hours.
As with other antibiotics, caution may be required in using meropenem as monotherapy in critically ill patients with known or suspected Pseudomonas aeruginosa lower respiratory tract infection.
Regular sensitivity testing is recommended when treating Pseudomonas aeruginosa infection.
Meropenem-AFT should be given as an intravenous bolus injection over approximately 5 minutes or by intravenous infusion over approximately 15 to 30 minutes (see Constitution and compatibility). There is limited safety data available to support the administration of a 2 g bolus dose.
Dosage schedule for adults with impaired renal function. Dosage should be reduced in patients with creatinine clearance less than 51 mL/min, as scheduled below in Table 1.
 Meropenem is cleared by haemodialysis. If continued treatment with Meropenem-AFT is necessary, it is recommended that the unit dose (based on the type and severity of infection) is administered at the completion of the haemodialysis procedure to restore therapeutically effective plasma concentrations.
Meropenem is cleared by haemodialysis. If continued treatment with Meropenem-AFT is necessary, it is recommended that the unit dose (based on the type and severity of infection) is administered at the completion of the haemodialysis procedure to restore therapeutically effective plasma concentrations.
There is no experience with peritoneal dialysis.
Use in adults with hepatic insufficiency. No dosage adjustment is necessary in patients with impaired hepatic metabolism.
Elderly patients. No dosage adjustment is required for the elderly with normal renal function or creatinine clearance values above 50 mL/min.
Children.
For infants and children over 3 months and up to 12 years of age the recommended intravenous dose is 10 to 40 mg/kg every 8 hours depending on type and severity of infection, the known or suspected susceptibility of the pathogen(s) and the condition of the patient. In children over 50 kg weight, adult dosage should be used.
Exceptions.
1. Febrile episodes in neutropenic patients - the dose should be 20 mg/kg every 8 hours.
2. Meningitis - the dose should be 40 mg/kg every 8 hours.
Meropenem-AFT should be given as an I.V. bolus over approximately 5 minutes or by intravenous infusion over approximately 15 to 30 minutes. There is limited safety data available to support the administration of a 40 mg/kg bolus dose.
There is no experience in children with renal impairment.
Constitution and compatibility.
Meropenem-AFT to be used for bolus I.V. injection should be constituted with sterile water for injections (10 mL per 500 mg meropenem). This provides an approximate available concentration of 50 mg/mL. Constituted solutions are both clear and colourless to pale yellow.
Meropenem-AFT for I.V. infusion may be directly constituted with either 0.9% sodium chloride solution for infusion or 5% glucose (dextrose) solution for infusion and then further diluted (50 to 200 mL) with the compatible infusion fluid (final concentration of 1 to 20 mg/mL).
Pharmaceutical precautions.
Shake constituted solution before use. All vials are for single use only. Standard aseptic technique should be employed during constitution and administration.
Meropenem-AFT should not be mixed with or physically added to solutions containing other medicines.
Stability after constitution and dilution.
After reconstitution.
The reconstituted solutions for intravenous injection or infusion should be used immediately. The time interval between the beginning of reconstitution and the end of intravenous injection or infusion should not exceed one hour.
Solutions of Meropenem-AFT should not be frozen.4.3 Contraindications
Meropenem is contraindicated in patients who have demonstrated hypersensitivity to this product.
4.4 Special Warnings and Precautions for Use
Identified precautions.
Serious and occasionally fatal hypersensitivity reactions have been reported in patients receiving therapy with β-lactams. These reactions are more likely to occur in persons with a history of sensitivity to multiple allergens. There have been reports of patients with a history of penicillin hypersensitivity who have experienced severe hypersensitivity when treated with another β-lactam. Before initiating treatment with meropenem, careful inquiry should be made concerning previous hypersensitivity reactions to carbapenems, penicillins, or other β-lactam antibiotics. If an allergic reaction occurs to meropenem then discontinue the medicine. Serious hypersensitivity reactions may require adrenaline and other emergency measures.
Rhabdomyolysis has been reported with the use of meropenem. If signs or symptoms of rhabdomyolysis are observed, meropenem should be discontinued and appropriate therapy initiated.
As with other antibiotics, overgrowth of non-susceptible organisms may occur and repeated evaluation of each patient is necessary.
As with other β-lactam antibiotics, strains of Pseudomonas aeruginosa may develop resistance on treatment with meropenem. Development of resistance has been reported in pseudomonal hospital acquired lower respiratory tract infections. In such cases, meropenem should be used with caution and repeat sensitivity testing is recommended.
Rarely, pseudomembranous colitis has been reported with meropenem as with practically all antibiotics and may vary in severity from slight to life-threatening. Therefore, antibiotics should be prescribed with care for individuals with a history of gastrointestinal complaints, particularly colitis. It is important to consider the diagnosis of pseudomembranous colitis in the case of patients who develop diarrhoea when using an antibiotic. Although studies indicate that a toxin produced by Clostridium difficile is one of the main causes of antibiotic-associated colitis, other causes should be considered. Mild cases usually respond to medicine discontinuation alone. However, in moderate to severe cases appropriate therapy such as oral antibacterial agents effective against Clostridium difficile should be considered.
Fluids, electrolytes and protein replacement should be provided when indicated. Medicines which delay peristalsis, e.g. opiates and diphenoxylate with atropine (Lomotil) may prolong and/or worsen the condition and should not be used.
Neurological sequelae were reported following treatment of severe meningitis with meropenem. In clinical trials these adverse events were reported in 23 of 148 patients treated with meropenem and in 17 of 144 patients treated with comparator antibiotics.
The concomitant use of valproic acid/sodium valproate and Meropenem-AFT is not recommended. Meropenem may reduce serum valproic acid levels. Subtherapeutic levels may be reached in some patients (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
Skin and subcutaneous tissue disorders.
Severe cutaneous adverse reactions (SCAR), such as Stevens-Johnson syndrome (SJS), toxic epidermal necrolysis (TEN), drug reaction with eosinophilia and systemic symptoms (DRESS), erythema multiforme (EM) and acute generalised exanthematous pustulosis (AGEP) have been reported in patients receiving meropenem (see Section 4.8 Adverse Effects (Undesirable Effects)). If signs and symptoms suggestive of these reactions appear, meropenem should be withdrawn immediately and an alternative treatment should be considered.
Use in hepatic impairment.
Patients with pre-existing liver disorders should have liver function monitored during treatment with Meropenem-AFT.
Use in renal impairment.
See Section 4.2 Dose and Method of Administration.
Use in the elderly.
See Section 4.2 Dose and Method of Administration; Section 5.2 Pharmacokinetic Properties.
Paediatric use.
Efficacy and tolerability in infants under 3 months of age have not been established; therefore, meropenem is not recommended for use below this age.
Effects on laboratory tests.
A positive or indirect Coombs' test may develop.4.5 Interactions with Other Medicines and Other Forms of Interactions
Meropenem has been administered concomitantly with many other medications without apparent adverse interaction. However, no specific medicine interaction studies other than with probenecid were conducted.
Probenecid.
Probenecid competes with meropenem for active tubular secretion and thus inhibits the renal excretion of meropenem with the effect of increasing the elimination half-life and plasma concentration of meropenem. As the potency and duration of action of meropenem dosed without probenecid are adequate the co-administration of probenecid with meropenem is not recommended. The potential effect of meropenem on the protein binding of other medicines or metabolism has not been studied. However, the protein binding is so low (approximately 2%) that no interactions with other compounds would be expected on the basis of this mechanism.
Valproic acid/ sodium valproate.
Decreases in blood levels of valproic acid have been reported when it is co-administered with carbapenem agents resulting in a 60-100% decrease in valproic acid levels in about two days. Due to the rapid onset and the extent of the decrease, co-administration of Meropenem-AFT in patients stabilised on valproic acid/sodium valproate is not considered to be manageable and therefore should be avoided (see Section 4.4 Special Warnings and Precautions for Use; Section 6.2 Incompatibilities).4.6 Fertility, Pregnancy and Lactation
Effects on fertility.
Fertility was not impaired in rats with exposures (based on AUC) slightly greater than those observed in patients at the recommended intravenous dose.
(Category B2)
Reproduction studies conducted with meropenem in rats have shown no embryotoxicity or teratogenicity at plasma exposures (based on AUC values) approximately equal to those observed in patients at the recommended intravenous dose. In a teratology study in cynomolgus monkeys given daily intravenous injections meropenem showed no evidence of teratogenicity at dose levels up to 360 mg/kg/day.
There are however, no adequate or well controlled trials of meropenem in pregnant women. Because reproduction studies are not always predictive of human response, Meropenem-AFT should not be used in pregnancy unless the potential benefit justifies the potential risk to the fetus.
Meropenem should not be used in breast-feeding women unless the potential benefit justifies the potential risk to the baby. Meropenem has been reported to be excreted in human breast milk.4.7 Effects on Ability to Drive and Use Machines
No studies on the ability to drive and use machines have been performed. However, when driving or operating machines it should be taken into account that headache, paresthesia and convulsions have been reported for meropenem.
4.8 Adverse Effects (Undesirable Effects)
Meropenem is generally well tolerated.
In clinical trials, adverse events lead to cessation of treatment in less than 1% of patients. Serious adverse events are rare. See Table 2.
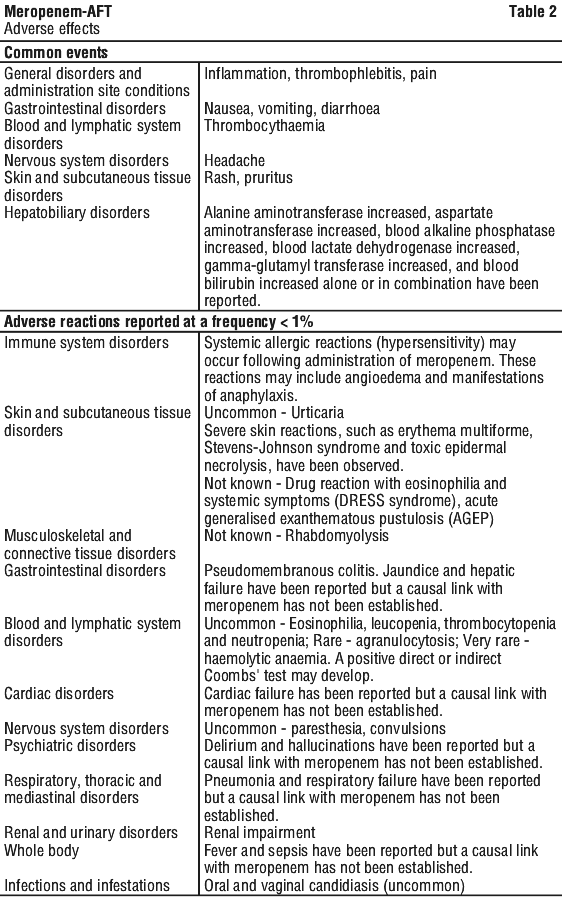
Description of selected adverse reactions.
Kounis syndrome.
Kounis syndrome (acute coronary syndrome associated with an allergic reaction) has been reported with other beta-lactam antibiotics.
Reporting suspected adverse effects.
Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.4.9 Overdose
The pharmacological properties and mode of administration make it unlikely that intentional overdose will occur. Accidental overdosage could occur during therapy, particularly in patients with renal impairment. Treatment of overdosage should be symptomatic. In normal individuals rapid renal elimination will occur. In subjects with renal impairment haemodialysis will remove meropenem and its metabolite.
For information on the management of overdose, contact the Poisons information Centre on 131126 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action.
Meropenem is a carbapenem antibiotic for parenteral use, that is stable to human dehydropeptidase-1 (DHP-1).
Meropenem exerts its bactericidal action by interfering with vital bacterial cell wall synthesis. The ease with which it penetrates bacterial cell walls, its high level of stability to all serine β-lactamases and its marked affinity for multiple penicillin binding proteins (PBPs) explain the potent bactericidal action of meropenem against a broad spectrum of aerobic and anaerobic bacteria. Bactericidal concentrations are commonly the same as the minimum inhibitory concentrations (MICs).
Meropenem is stable in susceptibility tests and these tests can be performed using normal routine methods. In vitro tests show that meropenem acts synergistically with various antibiotics. It has been demonstrated both in vitro and in vivo that meropenem has a post-antibiotic effect.
Mechanisms of resistance.
Localised clusters of infections due to carbapenem-resistant bacteria have been reported in some regions.
The susceptibility to meropenem of a given clinical isolate should be determined by standard methods. Interpretation of test results should be made in accordance with local infectious diseases and clinical microbiology guidelines.
The antibacterial spectrum of meropenem includes the following species, based on clinical experience.
Commonly susceptible species. Gram-positive aerobes.
Enterococcus faecalis (note that E. faecalis can naturally display intermediate susceptibility), Staphylococcus aureus (methicillin-susceptible strains only: methicillin-resistant staphylococci including MRSA are resistant to meropenem), Staphylococcus species including Staphylococcus epidermidis (methicillin-susceptible strains only: methicillin-resistant staphylococci including MRSE are resistant to meropenem), Streptococcus agalactiae (Group B streptococcus), Streptococcus milleri group (S. anginosus, S. constellatus, and S. intermedius), Streptococcus pneumoniae, Streptococcus pyogenes (Group A streptococcus).
Gram-negative aerobes.
Citrobacter freundii, Citrobacter koseri, Enterobacter aerogenes, Enterobacter cloacae, Escherichia coli, Haemophilus influenzae, Klebsiella oxytoca, Klebsiella pneumoniae, Morganella morganii, Neisseria meningitidis, Proteus mirabilis, Serratia marcescens.
Gram-positive anaerobes.
Clostridium perfringens, Peptostreptococcus spp. (including P. micros, P. anaerobius, P. magnus).
Gram-negative anaerobes.
Bacteroides caccae, Bacteroides fragilis.
Species for which acquired resistance may be a problem. Gram-positive aerobes.
Enterococcus faecium (E. faecium can naturally display intermediate susceptibility even without acquired resistance mechanisms).
Gram-negative aerobes.
Acinetobacter species, Burkholderia cepacia, Pseudomonas aeruginosa.
Inherently resistant organisms. Gram-negative aerobes.
Stenotrophomonas (Xanthomonas) maltophilia, Legionella species.
Other inherently resistant organisms.
Chlamydophila pneumoniae, Chlamydophila psittaci, Coxiella burnetii, Mycoplasma pneumoniae.
The published medical microbiology literature describes in vitro meropenem-susceptibilities of many other bacterial species. However, the clinical significance of such in vitro findings is uncertain. Advice on the clinical significance of in vitro findings should be obtained from local infectious diseases and clinical microbiology experts and local professional guidelines.
The prevalence of acquired resistance may vary geographically and with time for selected species and local information on resistance is desirable, particularly when treating severe infections. As necessary, expert advice should be sought when the local prevalence of resistance is such that the utility of the agent in at least some types of infections is questionable.
Clinical trials.
No data available.
5.2 Pharmacokinetic Properties
A 30-minute intravenous infusion of a single dose of meropenem in normal volunteers results in peak plasma levels of approximately 11 microgram/mL for the 250 mg dose, 23 microgram/mL for the 500 mg dose, 49 microgram/mL for the 1 g dose and 115 microgram/mL following the 2 g dose.
A 5-minute intravenous bolus injection of meropenem in normal volunteers results in peak plasma levels of approximately 52 microgram/mL for the 500 mg dose and 112 microgram/mL for the 1 g dose.
Intravenous infusions over 2 minutes, 3 minutes and 5 minutes of a 1 g dose of meropenem were compared in a three-way crossover trial. These durations of infusion resulted in peak plasma levels of 110, 91 and 94 microgram/mL, respectively.
Distribution.
Meropenem penetrates well into most body fluids and tissues including cerebrospinal fluid of patients with bacterial meningitis, achieving concentrations in excess of those required to inhibit most bacteria.
Plasma protein binding of meropenem is approximately 2%.
Metabolism.
The only metabolite of meropenem is microbiologically inactive.
Excretion.
After an intravenous dose of 500 mg, plasma levels of meropenem decline to values of 1 microgram/mL or less, 6 hours after administration.
When multiple doses are administered at 8 hourly intervals to subjects with normal renal function, accumulation of meropenem does not occur.
In subjects with normal renal function, meropenem's elimination half-life is approximately one hour.
Approximately 70% of the intravenous administered dose is recovered as unchanged meropenem in the urine over 12 hours, after which little further urinary excretion is detectable. Urinary concentrations of meropenem in excess of 10 microgram/mL are maintained for up to 5 hours at the 500 mg dose. No accumulation of meropenem in plasma or urine was observed with regimens using 500 mg administered every 8 hours or 1 g administered every 6 hours in volunteers with normal renal function.
Special populations.
Use in hepatic impairment.
Pharmacokinetic studies in patients with liver disease have shown no effects of liver disease on the pharmacokinetics of meropenem.
Use in renal impairment.
Pharmacokinetic studies in patients with renal insufficiency have shown the plasma clearance of meropenem correlates with creatinine clearance. Dosage adjustments are necessary in subjects with renal impairment.
Use in the elderly.
Pharmacokinetic studies in the elderly have shown a reduction in plasma clearance of meropenem which correlated with age-associated reduction in creatinine clearance.
Paediatric use.
Studies in children have shown that the pharmacokinetics of meropenem in children are essentially similar to those in adults. The elimination half-life for meropenem was approximately 1.5 hours in children under the age of 2 years.
The pharmacokinetics are linear over the dose range of 10 to 40 mg/kg.
5.3 Preclinical Safety Data
Genotoxicity.
Meropenem, with and without metabolic activation as appropriate, was not genotoxic in assays for gene mutations (Salmonella typhimurium, E. coli and Chinese hamster ovary cells) and chromosomal damage (mouse micronucleus assay and human lymphocytes in vitro).
Carcinogenicity.
No data available.6 Pharmaceutical Particulars
6.1 List of Excipients
Sodium carbonate.
6.2 Incompatibilities
Meropenem-AFT should not be mixed with or physically added to solutions containing other medicines. Also see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG).
The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 30°C.
6.5 Nature and Contents of Container
Meropenem-AFT 500 mg and 1000 mg are supplied in 20 mL and 30 mL Type I glass vials, respectively covered with brominated butyl rubber stopper. 10 vials are packed in a carton.
6.6 Special Precautions for Disposal
Product is for single use in one patient only. Discard any residue.
In Australia, any unused medicine or waste material should be disposed of in accordance with local requirements.
6.7 Physicochemical Properties
Chemical structure.

CAS number.
119478-56-7.7 Medicine Schedule (Poisons Standard)
S4 - Prescription only medicine.
Summary Table of Changes


 Meropenem is cleared by haemodialysis. If continued treatment with Meropenem-AFT is necessary, it is recommended that the unit dose (based on the type and severity of infection) is administered at the completion of the haemodialysis procedure to restore therapeutically effective plasma concentrations.
Meropenem is cleared by haemodialysis. If continued treatment with Meropenem-AFT is necessary, it is recommended that the unit dose (based on the type and severity of infection) is administered at the completion of the haemodialysis procedure to restore therapeutically effective plasma concentrations.

