1 Name of Medicine
Metoprolol succinate.
2 Qualitative and Quantitative Composition
Metrol-XL 23.75 mg.
Each modified release tablet contains 23.75 mg metoprolol succinate.
Metrol-XL 47.5 mg.
Each modified release tablet contains 47.5 mg metoprolol succinate.
Metrol-XL 95 mg.
Each modified release tablet contains 95 mg metoprolol succinate.
Metrol-XL 190 mg.
Each modified release tablet contains 190 mg metoprolol succinate.
Excipient with known effect.
None.
For the full list of excipients, see Section 6.1 List of Excipients.3 Pharmaceutical Form
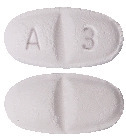
Metrol-XL 23.75 mg.
White to off white, oval shape, biconvex film coated tablet with breakline on one side and debossed with 'A' and '3' on each side of breakline on other side.
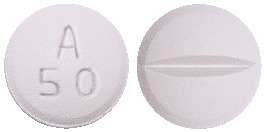
Metrol-XL 47.5 mg.
White to off white, round shape, biconvex film coated tablet with breakline on one side and debossed with 'A50' on other side.
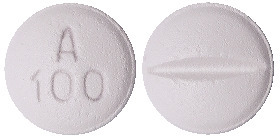
Metrol-XL 95 mg.
White to off white, round shape, biconvex film coated tablet with breakline on one side and debossed with 'A100' on other side.
Metrol-XL 190 mg.
White to off white, oval shape, biconvex film coated tablet with breakline on one side and debossed with 'A200' on other side.4.1 Therapeutic Indications
Stable, chronic heart failure as an adjunct to other heart failure therapy.
4.2 Dose and Method of Administration
Dosage.
Metrol-XL has not been established to be clinically equivalent to immediate release forms of metoprolol, and should not be used for treatment of conditions other than stable, chronic heart failure.
Metrol-XL is recommended for once daily treatment and is preferably taken together with the morning meal. Metrol-XL tablets should be swallowed with liquid and should not be chewed or crushed.
The dose of Metrol-XL should be individually adjusted in patients with chronic heart failure stabilised on other heart failure treatment.
It is recommended that patients be titrated from an initial low dose in accordance with the following titration schedule (see Table 1):
 Metrol-XL 47.5 mg, Metrol-XL mg and Metrol-XL 190 mg tablets may only be broken in half for ease of swallowing and not to divide into equal doses.
Metrol-XL 47.5 mg, Metrol-XL mg and Metrol-XL 190 mg tablets may only be broken in half for ease of swallowing and not to divide into equal doses.
The patient should be carefully evaluated at each dose level with regard to tolerability. If the patient experiences hypotension a decreased dose of concomitant heart failure medication may be necessary. Initial hypotension does not necessarily mean that the dose cannot be tolerated during chronic treatment but the patient should be kept at the lower dose until their blood pressure has stabilised.
Some patients may experience an initial, usually transient, worsening of the symptoms and signs of heart failure when starting treatment with Metrol-XL. If this occurs, the patient should be monitored very closely and the dose of Metrol-XL should be reduced if symptoms continue to worsen. Metrol-XL should not be ceased abruptly due to the risk of rebound hypertension and tachycardia. If treatment is to be discontinued, it should be reduced gradually (see Section 4.4 Special Warnings and Precaution for Use).
Special patient populations. Impaired renal and hepatic function.
Dose adjustment is not needed in patients with impaired renal function.
Dose adjustment is normally not needed in patients suffering from liver cirrhosis because metoprolol is low protein binding (5-10%). When there are signs of serious impairment of liver function (e.g. patients who have had a shunt operation), a dose reduction should be considered.
Elderly.
Dose adjustment is not needed in the elderly.
Children.
There is limited experience with Metrol-XL treatment in children.4.3 Contraindications
Predisposition to bronchospasm. β-adrenergic blockade of the smooth muscle of bronchi and bronchioles may result in an increased airways resistance. These drugs also reduce the effectiveness of asthma treatment. This may be dangerous in susceptible patients.
Therefore, β-blockers are contraindicated in any patient with a history of airways obstruction or a tendency to bronchospasm. Use of cardioselective β-blockers can also result in severe bronchospasm. If such therapy must be used, great caution should be exercised. Alternative therapy should be considered.
Allergic disorders (including allergic rhinitis) which may suggest a predisposition to bronchospasm.
Right ventricular failure secondary to pulmonary hypertension.
Significant right ventricular hypertrophy.
Second and third degree atrioventricular block.
Shock (including cardiogenic and hypovolaemic shock).
Unstable decompensated congestive heart failure (pulmonary oedema, hypoperfusion or hypotension).
Continuous or intermittent inotropic therapy acting through β-receptor agonism.
Clinically relevant sinus bradycardia (less than 45-50 beats/minute).
Non-compensated congestive heart failure (see Section 4.4 Special Warnings and Precaution for Use).
Sick-sinus syndrome (unless a permanent appropriately functioning pacemaker is in place).
Severe peripheral arterial circulatory disorders.
Suspected acute myocardial infarction with a heart rate of < 45 beats/minute, a P-R interval of > 0.24 seconds or a systolic blood pressure of < 100 mmHg, and/or moderate to severe non-compensated heart failure.
Hypotension.
Untreated phaeochromocytoma (see Section 4.4 Special Warnings and Precaution for Use).
Hypersensitivity to any component of Metrol-XL and related derivatives. Cross-sensitivity between β-blockers can occur.
4.4 Special Warnings and Precautions for Use
Symptomatic cardiac failure.
β-blockers should not be used in patients with unstabilised heart failure. This condition should first be stabilised with appropriate treatment (e.g. angiotensin-converting enzyme inhibitors, digoxin, diuretics). If cardiac failure persists, Metrol-XL should be discontinued gradually (see Section 4.4 Special Warnings and Precaution for Use, Abrupt withdrawal).
Bronchospasm.
Where Metrol-XL is prescribed for patients known to be suffering from asthma, an inhaled β2-agonist should be administered. The dosage of β2-agonists may require adjustment (increase), however the risk of Metrol-XL interfering with β2-receptors is less than with conventional tablet formulations of β1-selective blockers.
Concomitant therapy with calcium antagonists.
The concomitant use of calcium antagonists with myocardial suppressant and sinus node activity (e.g. verapamil and to a lesser extent diltiazem) and β-blockers may cause bradycardia, hypotension and asystole. Extreme caution is required if these drugs have to be used together (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
Anti-arrhythmic drugs.
Care should be taken when prescribing β-blockers with antiarrhythmic drugs as they may enhance the negative inotropic and chronotropic effects (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
Diabetes.
Metrol-XL should be used with caution in patients with diabetes mellitus, especially those who are receiving insulin or oral hypoglycaemic agents. Diabetic patients should be warned that β-blockers affect glucose metabolism and may mask some important premonitory signs of acute hypoglycaemia, such as tachycardia.
In patients with insulin or non-insulin dependent diabetes, especially labile diabetes, or with a history of spontaneous hypoglycaemia, β-blockade may result in the loss of diabetic control and delayed recovery from hypoglycaemia. The dose of insulin or oral hypoglycaemic agent may need to be adjusted. Diabetic patients receiving Metrol-XL should be monitored to ensure diabetes control is maintained.
Other metabolic effects.
Beta adrenoreceptors are involved in the regulation of lipid as well as carbohydrate metabolism. Some drugs affect the lipid profile adversely although the long-term clinical significance of this change is unknown and the effect appears to be less for drugs with intrinsic sympathomimetic activity.
Conduction disorders.
Very rarely a pre-existing A-V conduction disorder of moderate degree may become aggravated (possibly leading to A-V block). Metrol-XL should be administered with caution to patients with first degree A-V block (see Section 4.3 Contraindications).
Peripheral vascular disease.
β-blockade may impair the peripheral circulation and exacerbate the symptoms of peripheral vascular disease (see Section 4.3 Contraindications).
Bradycardia.
If patients develop increasing bradycardia, Metrol-XL should be given in lower doses or gradually withdrawn.
Prinzmetal angina.
There is a risk of exacerbating coronary artery spasm if patients with Prinzmetal or variant angina are treated with a β-blocker. If this treatment is essential, it should only be undertaken in a coronary or intensive care unit.
Effects on the thyroid.
The effects of β-blockers on thyroid hormone metabolism may result in elevations of serum free thyroxine (T4) levels. In the absence of any signs or symptoms of hyperthyroidism, additional investigation is necessary before a diagnosis of thyrotoxicosis can be made.
Phaeochromocytoma.
Where Metrol-XL is prescribed for a patient known to be suffering from phaeochromocytoma, an α-blocker should be given concomitantly to avoid exacerbation of hypertension.
Effects on the eye and skin.
Various skin rashes and conjunctival xerosis have been reported with β-blocking agents. Cross-reactions may occur between β-blockers, therefore substitutions within the group may not necessarily preclude occurrence of symptoms.
During long-term treatment with the β-blocking drug practolol a specific rash bearing a superficial resemblance to psoriasis was occasionally described. In a number of the patients affected, this rash was accompanied by adverse effects on the eye (xerophthalmia and/or keratoconjunctivitis) of varying severity. This condition is called the oculomucocutaneous or practolol syndrome. On a few rare occasions, serious otitis media, sclerosing peritonitis and pleurisy have been reported as part of this syndrome.
The oculomucocutaneous syndrome as reported with practolol has not been reported with metoprolol. However, dry eyes and skin rash have been reported with metoprolol. If such symptoms occur, discontinuation of metoprolol should be considered.
Recently, an association between Peyronie's disease (a fibrosing induration of the penis) and various β-blockers has been suggested but is not proven.
General anaesthesia.
Prior to surgery the anaesthetist should be informed that the patient is receiving Metrol-XL because of the potential for interactions with other drugs, resulting in severe bradyarrhythmias and hypotension, decreased reflex ability to compensate for blood loss, hypovolaemia and regional sympathetic blockade, and decreased propensity for vagal-induced bradycardia (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions). It is not recommended to stop β-blocker treatment in patients undergoing surgery. Acute initiation of high-dose metoprolol to patients undergoing non-cardiac surgery should be avoided, since it has been associated with bradycardia, hypotension and stroke including fatal outcome in patients with cardiovascular risk factors.
If it is thought necessary to withdraw β-blocker therapy before surgery, this should be done gradually and completed about 48 hours before surgery. (See Abrupt withdrawal below.)
Abrupt withdrawal.
Abrupt withdrawal of β-blockade is hazardous, especially in high risk patients, and should not be done. If there is a need to discontinue treatment with Metrol-XL, this should be done gradually over at least two weeks with the dose reduced by half in each step, down to a final dose of half a 23.75 mg tablet. The final dose should be taken for at least four days before discontinuation. Close observation of the patient is required during the withdrawal phase. If symptoms occur, a slower withdrawal rate is recommended. Sudden withdrawal of β-blockade may aggravate chronic heart failure and also increase the risk of myocardial infarction and sudden death.
Allergic conditions.
Allergic reactions may be exaggerated by β-blockade (e.g. allergic rhinitis during the pollen season and allergic reactions to bee and wasp stings). β-blockers should be avoided if there is a risk of bronchospasm.
In patients taking β-blockers, anaphylactic shock assumes a more severe form and may be resistant to usual doses of adrenaline. Whenever possible, β-blockers should be avoided in patients who are at increased risk of anaphylaxis.
Hyperthyroidism.
Because β-blockers may mask the clinical signs of developing or continuing hyperthyroidism resulting in symptomatic improvement without any change in thyroid status, special care should be exercised in hyperthyroid patients who are also receiving β-blockers. Where Metrol-XL is administered to patients having, or suspected of developing thyrotoxicosis, both thyroid and cardiac function should be closely monitored.
Effects on the heart rate.
If the patient develops increasing bradycardia (heart rate less than 50 to 55 beats/minute), the dosage of Metrol-XL should be gradually reduced or treatment gradually withdrawn (see Section 4.3 Contraindications).
Use in renal impairment.
In patients with severe renal disease haemodynamic changes following β-blockade may impair renal function further. β-blockers which are excreted mainly by the kidney may require dose adjustment in patients with renal failure.
Use in hepatic impairment.
Metoprolol is mainly eliminated by hepatic metabolism (see Section 5.2 Pharmacokinetic Properties). Therefore, liver cirrhosis may increase the systemic bioavailability of metoprolol and reduce its total clearance, leading to increased plasma levels.
Use in the elderly.
See Section 4.2 Dose and Method of Administration; Section 5.2 Pharmacokinetic Properties.
Paediatric use.
The safety and efficacy of metoprolol in children has not been established.
Effects on laboratory tests.
No data available.4.5 Interactions with Other Medicines and Other Forms of Interactions
CYP2D6 inhibitors.
Coadministration of drugs which inhibit CYP2D6 such as quinidine, fluoxetine and paroxetine may cause increased exposure to metoprolol and consequent increased pharmacological effects.
Concomitant administration of the CYP2D6 inhibitor quinidine has been shown to substantially increase systemic exposure of both enantiomers of metoprolol. In healthy subjects with CYP2D6 extensive metaboliser phenotype, coadministration of quinidine 100 mg and immediate release metoprolol 200 mg tripled the concentration of S-metoprolol and doubled the metoprolol elimination half-life. These increases in plasma concentration are highly likely to be associated with exaggerated pharmacological effects and decrease in the cardioselectivity of metoprolol. Interactions with hydroxychloroquine and diphenhydramine, although smaller, could still be clinically significant.
Other anti-hypertensive agents.
Metoprolol enhances the effects of other antihypertensive drugs. Particular care is required when initiating administration of a β-blocker and prazosin together.
Sympathetic ganglion blocking agents, other β-blockers or monoamine oxidase (MAO) inhibitors.
Patients receiving concomitant treatment with sympathetic ganglion blocking agents, other β-blockers (including eye drops), or monoamine oxidase (MAO) inhibitors should be kept under close surveillance.
Clonidine.
Concurrent use of β-blockers and clonidine should be avoided because of the risk of adverse interaction and severe withdrawal symptoms.
If concomitant treatment with clonidine is to be discontinued, the β-blocker medication should be withdrawn several days before the gradual withdrawal of clonidine. The rebound hypertension associated with clonidine withdrawal can be exacerbated by the presence of a β-blocker. If both drugs are withdrawn simultaneously, a marked rise in blood pressure and/or arrhythmias may result.
If replacing clonidine by β-blocker therapy, the introduction of β-blockers should be delayed for several days after clonidine administration has stopped.
Calcium antagonists.
If Metrol-XL is given with calcium antagonists of the verapamil and diltiazem type the patient should be monitored for possible negative inotropic and chronotropic effects. Calcium antagonists of the phenylalkylamine type (e.g. verapamil) should not be given by intravenous administration to patients treated with metoprolol because there is a risk of cardiac arrest in this situation. Patients taking oral calcium antagonists of this type in combination with metoprolol should be closely monitored.
The combination of β-blockers with dihydropyridine calcium channel blockers with a weak myocardial depressant effect (e.g. felodipine, nifedipine) can be administered together with caution. In case excess hypotension develops, the calcium antagonist should be stopped or the dosage reduced.
Antiarrhythmic agents.
When metoprolol is given together with antiarrhythmic agents, the patients should be monitored for possible negative inotropic and chronotropic effects. The negative inotropic and negative chronotropic effects of antiarrhythmic agents of the quinidine type and amiodarone may be enhanced by β-blockers. Interactions have been reported during concomitant β-blocker therapy with the Class IA agents disopyramide, and less frequently quinidine; class IB agents, tocainide, mexiletine and lignocaine; the Class IC agent flecainide; the Class III agent amiodarone; and the Class IV antiarrhythmic agents (e.g. verapamil).
Anaesthetics.
In patients receiving β-blocker therapy, inhalation anaesthetics enhance the cardiodepressant effect (see Section 4.4 Special Warnings and Precautions for Use). Metoprolol may also reduce the clearance of other drugs (e.g. lignocaine).
Modern inhalational anaesthetic agents are generally well tolerated, although older agents (ether, cyclopropane, methoxyflurane, trichlorethylene) were sometimes associated with severe circulatory depression in the presence of β-blockage.
Liver enzyme effects.
Enzyme-inducing and enzyme-inhibiting substances may exert an influence on the plasma level of metoprolol. The plasma concentration of metoprolol is lowered by rifampicin and may be raised by cimetidine, alcohol, hydralazine, and selective serotonin re-uptake inhibitors (SSRIs) e.g. paroxetine, fluoxetine, and sertraline, quinidine, verapamil and diphenhydramine.
Prostaglandin synthetase inhibiting agents.
Concomitant treatment with indomethacin or other prostaglandin synthetase inhibiting agents may decrease the antihypertensive effect of β-blockers.
Alcohol.
Metoprolol may modify the pharmacokinetic behaviour of alcohol when taken together. The plasma level of metoprolol may be raised by alcohol.
Oral antidiabetic agents.
The dosages of oral antidiabetics may need to be adjusted in patients receiving β-blockers (see Section 4.4 Special Warnings and Precautions for Use).
Warfarin.
A limited number of reports have demonstrated a rise in AUC and concentration of warfarin when taken with another β-blocker. This could potentially increase the anti-coagulant effect of warfarin.
Catecholamine-depleting agents.
Concomitant use of catecholamine-depleting drugs such as reserpine, monoamine oxidase (MAO) inhibitors and guanethidine have an additive effect when given with β-blocking agents. Patients treated with Metrol-XL plus a catecholamine depletor should therefore be closely observed for evidence of hypotension or marked bradycardia, which may produce vertigo, syncope, or postural hypotension, since the added effect of a β-blocker may produce an excessive reduction of the resting sympathetic nervous tone.
Digitalis glycosides.
Digitalis glycosides, in association with β-blockers, may increase atrioventricular conduction time and may induce bradycardia.4.6 Fertility, Pregnancy and Lactation
Effects on fertility.
In rats dosed with 500 mg/kg/day (23 times the MRCD on a mg/m2 basis), there was a slight decrease in insemination rate (75% cf. 95% in untreated controls) with signs of maternal toxicity. There was no evidence of impaired fertility at 50 mg/kg/day (2.3 times the MRCD).
(Category C)
As with most drugs, metoprolol succinate should not be given during pregnancy unless its use is considered essential. As with all antihypertensive agents, β-blockers may cause side effects (e.g. reduced placental perfusion and bradycardia) in the foetus and new-born. Reduced placental perfusion has been associated with growth retardation, intrauterine death, abortion and early labour. It is therefore suggested that appropriate maternofoetal monitoring be performed in pregnant women treated with metoprolol succinate. During the late stages of pregnancy these drugs should only be given after weighing the needs of the mother against the risk to the foetus.
The lowest possible dose should be used and discontinuation of treatment should be considered at least 2 to 3 days before delivery to avoid increased uterine contractility and effects of β-blockade in the newborn (e.g. bradycardia, hypoglycaemia).
Metoprolol succinate was shown to increase foetal loss in rabbits at 25 mg/kg/day PO (2 times the MRCD on a mg/m2 basis), and increase still births and decrease neonatal survival in rats at 500 mg/kg/day PO (23 times the MRCD on a mg/m2 basis). These studies revealed no evidence of teratogenicityr.
As with most drugs, Metrol-XL should not be given during lactation unless its use is considered essential. As with all antihypertensive agents, β-blockers may cause side effects (e.g. bradycardia), in the breast-fed infant. The amount of metoprolol ingested via breast-milk seems to be negligible, in regard to β-blocking effect in the infant, if the mother is treated with metoprolol in doses within the normal therapeutic range.
Postnatal growth was not affected in lactating rats dosed with metoprolol succinate at up to 500 mg/kg/day PO (23 times the MRCD on a mg/m2 basis).4.7 Effects on Ability to Drive and Use Machines
Metrol-XL may occasionally cause dizziness, visual disturbances or fatigue (see Section 4.8 Adverse Effects (Undesirable Effects)) hence patients should know how they react to Metrol-XL before they drive or use machinery, particularly when starting or changing treatment.
4.8 Adverse Effects (Undesirable Effects)
Metrol-XL is well tolerated and adverse reactions have generally been mild and reversible. The following events have been reported as adverse events in clinical trials or reported from routine use, mostly with conventional metoprolol (metoprolol succinate). In many cases a relationship with metoprolol has not been established.
The following definitions of frequency are used: very common ≥ 10%; common 1 - 9.9%; uncommon 0.1 - 0.9%; rare 0.01 - 0.09%; very rare < 0.01%. See Table 2.

Reporting suspected adverse effects.
Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.4.9 Overdose
Symptoms.
Symptoms of overdosage may include severe hypotension, cardiac insufficiency, bradycardia and bradyarrhythmia, cardiac conduction disturbances, cardiogenic shock, cardiac arrest, impairment of consciousness/coma, convulsions and bronchospasm. The main clinical signs of overdosage are cardiovascular and in some cases decompensation may be rapid. Overdosage with Metrol-XL can lead to death.
Cases of overdosage in paediatric patients need to be given extra attention even if the patient appears well on presentation and even if only a small number of tablets have apparently been taken.
Management.
For information on management of overdose, contact the Poisons Information Centre on 131126 (Australia).
Care should be provided at a facility that can provide appropriate supporting measures, monitoring, and supervision.
Activated charcoal may reduce absorption of the medicine if given within one or two hours after ingestion. In patients who are not fully conscious or have impaired gag reflex, consideration should be given to administering activated charcoal via a nasogastric tube, once the airway is protected.
Syrup of ipecac and gastric lavage are no longer considered to be standard therapy for gut decontamination.
Atropine, adreno stimulating drugs or pacemaker to treat bradycardia and conduction disorders.
Hypotension, acute cardiac failure, and shock to be treated with suitable volume expansion, injection of glucagon (if necessary, followed by an intravenous infusion of glucagon), intravenous administration of adreno stimulating drugs such as dobutamine, with α1 receptor agonistic drugs added in presence of vasodilation. Intravenous use of calcium salts (Ca2+) can also be considered.
Bronchospasm can usually be reversed by bronchodilators.5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action.
Metoprolol is a β1-selective β-blocker, i.e. it blocks β1-receptors at doses lower than those needed to block β2-receptors.
Metoprolol is practically devoid of membrane stabilising activity and does not display partial agonist activity (i.e. intrinsic sympathomimetic activity = ISA) at doses required to produce β-blockade. The stimulant effect of catecholamines on the heart is reduced or inhibited by metoprolol. This leads to a decrease in heart rate, cardiac output, cardiac contractility and blood pressure.
Controlled release metoprolol succinate and the immediate release metoprolol succinate are not bioequivalent. Controlled release metoprolol succinate gives an even plasma concentration time profile and effect (β1-blockade) over 24 hours in contrast to conventional tablet formulations of β1-selective blockers including metoprolol succinate formulations.
When given together with a β2-agonist, metoprolol succinate in therapeutic doses interferes less than non-selective β-blockers with β2-mediated bronchodilation (see Section 4.4 Special Warnings and Precaution for Use). When clinically necessary, metoprolol succinate, in combination with a β2-agonist, may be given to patients with symptoms of obstructive pulmonary disease. Metoprolol succinate also interferes less with insulin release than non-selective β-blockers.
Clinical trials.
Three randomised, double-blind, placebo-controlled studies and one randomised, open crossover study had been conducted to establish the efficacy and safety of metoprolol succinate in patients with chronic heart failure.
The pivotal study, MERIT-HF (n=3991), was a survival study in patients with NYHA (New York Heart Association) functional class II-IV and decreased ejection fraction (≤ 0.40) on optimal standard therapy at enrolment. Patients were randomised to receive either metoprolol succinate (n=1990) or placebo (n=2001) once daily. The dose of metoprolol succinate was titrated during 6-8 weeks to the target of 200 mg. Treatment ranged from 0 to 622 days with a mean duration of one year.
Participants were predominantly male (76%), Caucasian (94%), previous (55%) or current (19%) smokers, aged 60-79 years (66%; mean age 64), with heart failure secondary to ischaemia (65%). Most had mild to moderate symptomatic heart failure at baseline: 41% in NYHA Class II, 56% in Class III and 4% in Class IV. The mean ejection fraction was 0.28. About half the patients had a history of hypertension (44%) and/or myocardial infarction (48%). 25% had a history of diabetes mellitus. 10-20% had peripheral oedema, jugular venous distension, pulmonary rales and/or hepatomegaly. 23% had a third heart sound, 34% had a heart murmur, 25% had an irregular heart beat and 16% were in atrial fibrillation. 90% of patients were on one or more diuretics, 89% on an angiotensin converting enzyme inhibitor, 7% on an angiotensin II-blocker, 63% on digitalis, 36% on long-acting nitrates, 54% on aspirin, 37% on an oral anticoagulant and 23% on a statin.
Table 3 summarises the results of the efficacy variables for metoprolol succinate compared to placebo.
 The numbers needed to treat (NNT) to achieve a reduction of 1 case of all cause mortality, cardiac death and non-fatal AMI, mortality from cardiovascular causes and from sudden death are as follows (see Table 4).
The numbers needed to treat (NNT) to achieve a reduction of 1 case of all cause mortality, cardiac death and non-fatal AMI, mortality from cardiovascular causes and from sudden death are as follows (see Table 4).
 Metoprolol succinate was generally well tolerated. Treatment was ceased due to adverse events in 10.3% of patients taking metoprolol succinate compared to 12.3% of those taking placebo. Compared to placebo, the overall rate of treatment withdrawal and withdrawal due to worsening heart failure tended to be less with metoprolol succinate, but the difference did not reach statistical significance.
Metoprolol succinate was generally well tolerated. Treatment was ceased due to adverse events in 10.3% of patients taking metoprolol succinate compared to 12.3% of those taking placebo. Compared to placebo, the overall rate of treatment withdrawal and withdrawal due to worsening heart failure tended to be less with metoprolol succinate, but the difference did not reach statistical significance.
5.2 Pharmacokinetic Properties
Absorption and distribution.
Metrol-XL consists of several hundred beads of metoprolol succinate, each coated with a polymeric membrane which controls the rate of metoprolol release. After rapid disintegration within the gastrointestinal tract, metoprolol is continuously released for approximately 20 hours, and a stable metoprolol plasma concentration is achieved over a dosage interval of 24 hours. Approximately 12% of metoprolol is bound to human serum proteins.
Distribution and excretion.
Metoprolol undergoes oxidative metabolism in the liver, primarily by CYP2D6. Due to polymorphism of CYP2D6, about 5-10% of Caucasians and a lower percentage of Asian and African populations are poor metabolisers of metoprolol. Such people experience higher plasma concentrations of metoprolol for a given dose. Because of these differences between individuals, gradual dose titration is important. Co-administration of drugs which inhibit CYP2D6 may increase plasma concentrations of metoprolol, particularly in extensive metabolisers (the majority of the population). Three main metabolites have been identified, although none have a beta-blocking effect of clinical importance.
Over 95% of an oral dose can be recovered in the urine. Only approximately 5% of the administered dose is excreted unchanged, with this figure rising to 30% in isolated cases.
Pharmacokinetics in the elderly.
The elderly shows no significant differences in the pharmacokinetics of metoprolol as compared with younger persons.
5.3 Preclinical Safety Data
Genotoxicity.
Metoprolol succinate was not mutagenic in a bacterial assay, nor did it induce chromosomal damage in Chinese hamsters (bone marrow micronucleus and chromosome aberration assays) or in mice (dominant lethal assay).
Carcinogenicity.
Long-term studies in animals have been conducted to evaluate the carcinogenic potential of metoprolol succinate. In rats at dietary doses of up to 800 mg/kg/day (36 times the maximum recommended clinical dose (MRCD) on a mg/m2 basis) for 18 months, there was no increase in the incidence of neoplasms. The only histologic changes that appeared to be drug related were an increased incidence of focal accumulation of foamy macrophages in pulmonary alveoli and an increased incidence of biliary hyperplasia.
In a 21-month study in CD-1 mice at dietary doses of up to 750 mg/kg/day (17 times the MRCD on a mg/m2 basis), benign lung tumours (small adenomas) occurred more frequently in female mice receiving the highest dose than in untreated control animals. There was no increase in malignant or total (benign plus malignant) lung tumours, nor in the overall incidence of tumours.6 Pharmaceutical Particulars
6.1 List of Excipients
Each Metrol-XL tablet contains: microcrystalline cellulose, silicon dioxide, povidone, ethylcellulose, methacrylic acid copolymer, triethyl citrate, macrogol 6000, silicified microcrystalline cellulose, croscarmellose sodium, sodium stearylfumarate, purified talc, opadry complete film coating system YS-1R-7006 Clear (ARTG PI No. 13068), opadry complete film coating system 02F280014 (ARTG PI No. 123088).
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 25°C. Protect from moisture.
6.5 Nature and Contents of Container
23.75 mg.
PVC/PVDC/Al blister packs containing 15 tablets.
HDPE bottles with polypropylene child-resistant screw caps containing 15, 30 and 90 tablets.
47.5 mg/95 mg/190 mg.
PVC/PVDC/Al blister packs containing 15 and 30 tablets.
HDPE Bottles with polypropylene child-resistant screw caps containing 15, 30 and 90 tablets.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
Metoprolol succinate is a white, crystalline powder with a melting point of approximately 138°C. It is freely soluble in water, soluble in methanol, sparingly soluble in ethanol, slightly soluble in dichloromethane and 2-propanol, and practically insoluble in ethyl acetate, acetone, diethyl ether and heptane.
Chemical structure.
 Chemical name: di-(±)-1-(isopropylamine)-3-[p-(2-methoxyethyl) phenoxy]-2-propanol succinate.
Chemical name: di-(±)-1-(isopropylamine)-3-[p-(2-methoxyethyl) phenoxy]-2-propanol succinate.
CAS number.
98418-47-4.7 Medicine Schedule (Poisons Standard)
S4 - Prescription only medicine.
Summary Table of Changes



 Metrol-XL 47.5 mg, Metrol-XL mg and Metrol-XL 190 mg tablets may only be broken in half for ease of swallowing and not to divide into equal doses.
Metrol-XL 47.5 mg, Metrol-XL mg and Metrol-XL 190 mg tablets may only be broken in half for ease of swallowing and not to divide into equal doses.
 The numbers needed to treat (NNT) to achieve a reduction of 1 case of all cause mortality, cardiac death and non-fatal AMI, mortality from cardiovascular causes and from sudden death are as follows (see Table 4).
The numbers needed to treat (NNT) to achieve a reduction of 1 case of all cause mortality, cardiac death and non-fatal AMI, mortality from cardiovascular causes and from sudden death are as follows (see Table 4). Metoprolol succinate was generally well tolerated. Treatment was ceased due to adverse events in 10.3% of patients taking metoprolol succinate compared to 12.3% of those taking placebo. Compared to placebo, the overall rate of treatment withdrawal and withdrawal due to worsening heart failure tended to be less with metoprolol succinate, but the difference did not reach statistical significance.
Metoprolol succinate was generally well tolerated. Treatment was ceased due to adverse events in 10.3% of patients taking metoprolol succinate compared to 12.3% of those taking placebo. Compared to placebo, the overall rate of treatment withdrawal and withdrawal due to worsening heart failure tended to be less with metoprolol succinate, but the difference did not reach statistical significance. Chemical name: di-(±)-1-(isopropylamine)-3-[p-(2-methoxyethyl) phenoxy]-2-propanol succinate.
Chemical name: di-(±)-1-(isopropylamine)-3-[p-(2-methoxyethyl) phenoxy]-2-propanol succinate.