What is in this leaflet
Please read this leaflet before you start taking MEZAVANT. This leaflet answers some common questions about MEZAVANT.
It does not contain all available information. It does not take the place of talking to your doctor or pharmacist.
All medicines have risks and benefits. Your doctor has weighed the risk of you using MEZAVANT against the benefit it is expected to have for you.
If you have any concerns about taking MEZAVANT, talk to your doctor or pharmacist.
Keep this leaflet with the medicine. You may want to read it again.
What MEZAVANT is used for
The name of your medicine is MEZAVANT 1.2 g, gastro-resistant, prolonged release tablets. It contains the active ingredient mesalazine (also called 5-aminosalicylic acid or 5-ASA), an anti-inflammatory drug for the treatment of ulcerative colitis.
Ulcerative colitis is a disease of the large intestine (the colon and rectum), where the lining of the intestine becomes red and swollen (inflamed) and ulcers develop. This can cause symptoms of frequent and bloody stools together with stomach cramps.
When MEZAVANT is taken for a flare up of ulcerative colitis, it acts through the entire colon and rectum to treat the inflammation and reduce symptoms. MEZAVANT can also be taken to help prevent re-occurrence of ulcerative colitis.
Before you take MEZAVANT
MEZAVANT 1.2 g gastro-resistant, prolonged release tablets are not suitable for everyone.
When you must not take it
Do not take MEZAVANT:
- If you are allergic (hypersensitive) to a family of drugs known as salicylates (which include aspirin).
- If you are allergic (hypersensitive) to mesalazine or any of the other ingredients of MEZAVANT listed in "Other ingredients" section of this leaflet.
- The expiry date printed on the carton is passed. If you use this medication after the expiry date has passed, it may not work as well.
- The packaging is torn or shows signs of tampering.
If you are not sure whether you should start using MEZAVANT, contact your doctor.
Before you take it
Tell your doctor or pharmacist if you:
- Have any kidney problems
- Have previously had inflammation of the heart (which may be the result of an infection in the heart)
- Have had a previous allergic reaction to MEZAVANT or other medicines containing mesalazine or sulphasalazine (other medicines taken for treatment of ulcerative colitis)
- Have narrowing or blockage of the stomach or the gut
- Have skin conditions such as atopic dermatitis or atopic eczema
- Are pregnant or intend to become pregnant
- Are breastfeeding or wish to breastfeed.
If you have not told your doctor or pharmacist about any of the above, tell them before you take any MEZAVANT.
Taking other medicines
Tell your doctor or pharmacist if you are taking or have recently taken any other medicines that contain:
- Mesalazine or sulphasalazine (taken for treatment of ulcerative colitis)
- Non-steroidal anti-inflammatory drugs (for example medicines containing aspirin, ibuprofen or diclofenac)
- Azathioprine or 6-mercaptopurine (known as 'immunosuppressant' medicines).
- Warfarin (taken to prevent blood clots) or other coumarin-type anticoagulants
Tell your doctor or pharmacist if you are taking or have recently taken other medicines, including any that you buy without a prescription from your pharmacy, supermarket or health food shop.
How to take MEZAVANT
Follow all directions given to you by your pharmacist or doctor carefully as this may differ from the information contained in this leaflet.
If you do not understand the instructions in this leaflet, ask your doctor or pharmacist for help.
How much to take
Adults and the elderly
The usual daily dose for adults and the elderly is 2.4g to 4.8g (two to four tablets) taken once a day for an acute episode of ulcerative colitis.
- If you are taking the highest daily dose of 4.8g/day, you should be evaluated after 8 weeks of treatment
- Once your symptoms have cleared and to help prevent re-occurrence of another episode, your doctor may direct you to take 2.4g (two tablets) once a day.
Children and Adolescents
MEZAVANT is not recommended to be given to children under 18 years of age due to lack of data on safety and efficacy.
How to take it
Take your tablets with or without food at the same time each day. The tablets must be swallowed whole and must not be crushed or chewed.
How long to take it
It is important to take your MEZAVANT tablets every day for as long as your doctor tells you to, even when you don't have any symptoms of ulcerative colitis.
If you forget to take it
If you forget to take your tablets then take them as usual the next day. Do not take a double dose to make up for a forgotten tablet.
If you take too much
Immediately telephone your doctor or the Poisons Information Centre (telephone 13 11 26) for advice, or go to Accident and Emergency at the nearest hospital, if you think that you or anyone else may have used too much MEZAVANT. Do this even if there are no signs of discomfort or poisoning.
If you take too much MEZAVANT you may have one or more of the following symptoms: ringing in ears (tinnitus), dizziness, headache, confusion, drowsiness, shortness of breath, excess loss of water (associated with sweating, diarrhoea and vomiting), low blood sugar (which can cause light-headedness), rapid breathing, and increased body temperature. Blood tests may show changes in your blood chemistry (electrolyte balance).
While you are taking MEZAVANT
Things you must do
- MEZAVANT should be taken with or without food at the same time each day. The tablets should be swallowed whole and must not be crushed or chewed
- Ensure adequate fluid intake during treatment.
- Make sure that all of your doctors and pharmacists know you are taking MEZAVANT. Remind them if any new medicines are about to be started.
Things you must not do
- Do not stop taking your tablets or change the dosage without checking with your doctor
- Do not give to children under 18 years of age.
MEZAVANT is not suitable for children under 18 years of age as there is not enough information on its effects in children. - Do not give MEZAVANT to anyone else, even if they have the same condition as you. It may not be safe for another person to take MEZAVANT
- Do not take MEZAVANT to treat any other complaints unless your doctor tells you to. It may not be safe to use MEZAVANT for another complaint.
Driving or operating machinery
MEZAVANT is unlikely to have any effect on your ability to drive or use machines.
Side effects
Tell your doctor or pharmacist as soon as possible if you do not feel well while you are taking MEZAVANT.
Like all medicines, MEZAVANT can cause some side effects. Sometimes they are serious, most of the time they are mild and temporary.
Do not be alarmed by this list of possible side effects. You may not experience any of them.
Ask your doctor or pharmacist to answer any questions you may have.
Tell your doctor or pharmacist if you notice any of the following and they worry you:
- Headache
- Mild stomach pains
- Bloating or passing wind
- Excessive gas in the stomach or bowel
- Increased number of bowel motions
- Nausea (feeling sick)
- Vomiting (being sick)
- Rash or itchy skin
- Fever
- Loss of strength
- Muscle, joint or back pain
- Increased blood pressure
- Indigestion
- Tiredness
Tell your doctor immediately or go to Accident and Emergency at your nearest hospital if you notice any of the following:
- If you experience symptoms such as cramping, severe stomach pain, bloody and excessive stools (diarrhoea), fever, headache or rash. These symptoms could be a sign of Acute Intolerance Syndrome which can happen during an acute episode of ulcerative colitis. This is a serious condition which occurs rarely, but means your treatment would have to be stopped immediately
- If you develop unexplained bruising (without injury), rash, anaemia (feeling tired, weak and looking pale, especially on lips, nails and inside of eyelids), fever (high temperature), sore throat or unusual bleeding (e.g. nose bleeds)
- If you develop allergic swelling of face, eyes, lips, mouth, tongue or throat which may cause difficulty in swallowing or breathing.
- Widespread rash with blistering of the skin, particularly around the mouth, eyes and genitals
- Allergic skin rash with fever
- Severe pain in the back, side or stomach. Trouble urinating, pain or burning during urination. These could be symptoms of kidney stones
These are very serious side effects. You may need urgent medical attention or hospitalisation. All of these side effects are very rare.
Other rare events which have been reported with mesalazine include:
- Changes in kidney function and inflammation of the kidney, including kidney failure
- Changes in blood test results such as low white blood cell and/or platelet counts
- Changes in liver function tests
- Liver disease with nausea, vomiting, loss of appetite, feeling generally unwell, fever, itching, yellowing of the skin and eyes, and dark coloured urine
- Changes relating to your heart
- Inflammation of the lung, causing difficulty in breathing or wheezing
Tell your doctor or pharmacist if you notice anything that is making you feel unwell.
Other side effects not listed above may also occur in some people.
After taking MEZAVANT
Storage
- Keep out of the reach and sight of children
- Store below 25°C
- Store in the original package
- Do not use MEZAVANT after the expiry date which is stated on the box.
Disposal
Return any unused or out of date medicine to your pharmacist.
Product description
What MEZAVANT looks like
MEZAVANT are oval shaped red-brown tablets which are stamped with S476.
MEZAVANT is supplied as a 60 tablets pack size.
Ingredients
Active substance
The active substance is 1.2 g mesalazine
Other ingredients
Carmellose sodium, carnauba wax, stearic acid, silicon dioxide, sodium starch glycollate, talc-purified, magnesium stearate, methacrylic acid copolymer, triethyl citrate, titanium dioxide (E171), iron oxide red (CI77491), Macrogol 6000.
Sponsor
Shire Australia Pty Ltd
Shire is now part of Takeda
Level 39
225 George Street
Sydney, NSW 2000
Australia
Phone: 1800 012 612
This leaflet was prepared in Aug 2020.
Australian Registration Number: AUST R 155172
Published by MIMS September 2020


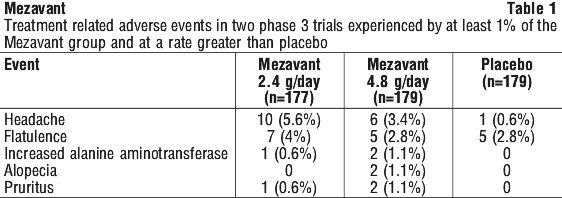 In a pooled safety analysis of patients with active ulcerative colitis who participated in controlled studies, the majority of subjects did not experience treatment emergent adverse events associated with Mezavant. Of the events reported, the majority were transient, and mild or moderate in severity. The three most frequently reported adverse drug reactions within the pooled safety analysis of the ulcerative colitis patient clinical studies were headache, abdominal pain and nausea. The common (≥ 1/100 to < 1/10), uncommon (≥ 1/1,000 to < 1/100) and rare (≥ 1/10,000 to < 1/1,000) treatment related adverse events are listed in Table 2.
In a pooled safety analysis of patients with active ulcerative colitis who participated in controlled studies, the majority of subjects did not experience treatment emergent adverse events associated with Mezavant. Of the events reported, the majority were transient, and mild or moderate in severity. The three most frequently reported adverse drug reactions within the pooled safety analysis of the ulcerative colitis patient clinical studies were headache, abdominal pain and nausea. The common (≥ 1/100 to < 1/10), uncommon (≥ 1/1,000 to < 1/100) and rare (≥ 1/10,000 to < 1/1,000) treatment related adverse events are listed in Table 2. In postmarketing experience, the following adverse reactions have been reported. See Table 3.
In postmarketing experience, the following adverse reactions have been reported. See Table 3.
 A randomised, open label extension study to studies 301 and 302 was designed to assess the long-term safety and tolerability of Mezavant 2.4 g/day administered once daily and in 2 divided doses (1.2 g twice daily) in the Maintenance of UC in remission over 12 months. This study (Study 303) included an 8-week Acute Extension Phase during which Mezavant 4.8 g/day was administered twice daily, and a 12-month Maintenance Phase during which Mezavant 2.4 g/day was administered either [1.2 g] twice daily or once daily. Efficacy was a secondary objective of this uncontrolled extension study.
A randomised, open label extension study to studies 301 and 302 was designed to assess the long-term safety and tolerability of Mezavant 2.4 g/day administered once daily and in 2 divided doses (1.2 g twice daily) in the Maintenance of UC in remission over 12 months. This study (Study 303) included an 8-week Acute Extension Phase during which Mezavant 4.8 g/day was administered twice daily, and a 12-month Maintenance Phase during which Mezavant 2.4 g/day was administered either [1.2 g] twice daily or once daily. Efficacy was a secondary objective of this uncontrolled extension study.

