What is in this leaflet
This leaflet answers some common questions about insulin Penfill®. It does not contain all the available information. It does not take the place of talking to your doctor, diabetes education nurse or pharmacist.
All medicines have risks and benefits. Your doctor has weighed the risks of you using insulin against the benefits they expect it will have for you.
If you have any concerns about using this medicine, ask your doctor, diabetes education nurse or pharmacist.
Keep this leaflet with the medicine. You may need to read it again.
What insulin is used for
Insulin is used to treat diabetes mellitus. Diabetes mellitus is a condition where your pancreas does not produce enough insulin to control your blood sugar (glucose) level. Extra insulin is therefore needed.
There are two types of diabetes mellitus:
- Type 1 diabetes
- Type 2 diabetes
Patients with type 1 diabetes always require insulin to control their blood sugar levels.
Some patients with type 2 diabetes may also require insulin after initial treatment with diet, exercise and tablets.
Insulin lowers your blood sugar level after injection. Each of the following insulins is available in Penfill®, a 3 mL glass cartridge designed to be used with Novo Nordisk insulin delivery devices such as NovoPen®.
Actrapid® is Neutral Insulin Injection, a fast-acting insulin. This means that it will start to lower your blood sugar level about half an hour after you inject it, and the effect will last for approximately 8 hours. Actrapid® is often used in combination with longer-acting insulin products.
Protaphane® is Isophane Insulin Injection, an intermediate-acting insulin. This means that it will start to lower your blood sugar level about one and a half hours after you inject it, and the effect will last for approximately 24 hours. Protaphane® is often used in combination with fast- or rapid-acting insulin products.
Mixtard® 30/70 and Mixtard® 50/50 are Biphasic Isophane Insulin Injection, and each is a mixture of fast- and longer-acting insulins. Mixtard® 30/70 and Mixtard® 50/50 will start to lower your blood sugar level about half an hour after you inject, and the effect will last for approximately 24 hours.
As with all insulins, the duration of action will vary according to the type of insulin, the dose, injection site, blood flow, temperature and level of physical activity.
Actrapid® is also used by doctors to treat some people with diabetes in emergency situations.
Insulin is not addictive.
Insulin is available only with a doctor’s prescription.
Ask your doctor if you have any questions about why insulin Penfill® has been prescribed for you.
Before you use insulin Penfill®
When you must not use it
Do not use Penfill® if:
- you have an allergy to:
- any medicine containing insulin
- any of the ingredients listed in the “Ingredients” section of this leaflet
Some of the symptoms of an allergic reaction may include:
- redness, swelling, rash and itching at the injection site
- rash, itching or hives on the skin
- shortness of breath
- wheezing or difficulty breathing
- swelling of the face, lips, tongue or other parts of the body.
- you are experiencing a low blood sugar level (a “hypo”) when the dose is due.
If you have a lot of hypos discuss appropriate treatment with your doctor.
If you are not sure whether you should start using this medicine, talk to your doctor.
Do not inject Protaphane®, Mixtard® 30/70 or Mixtard® 50/50 into a vein.
Do not use Protaphane®, Mixtard® 30/70 or Mixtard® 50/50 to treat a condition known as diabetic ketoacidotic coma.
Do not use this medicine after the expiry date printed on the label and carton, or if the packaging is torn or shows signs of tampering or does not look quite right. If it has expired or is damaged, return it to your pharmacist for disposal.
Before you start to use it
Tell your doctor if you have allergies to any other medicines, foods, preservatives or dyes.
Tell your doctor if you have or have had any medical conditions, especially the following:
- kidney problems
- liver problems
- adrenal, pituitary or thyroid gland problems.
Tell your doctor if you are pregnant or planning to become pregnant. Pregnancy may make managing your diabetes more difficult. Insulin needs usually decrease during the first three months of pregnancy and increase during the last six months. Your doctor can discuss with you the risks and benefits involved.
Tell your doctor if you are breast-feeding or planning to breast-feed. Your doctor or pharmacist can discuss with you the risks and benefits involved.
Inform your doctor as soon as possible if you experience signs of heart failure such as unusual shortness of breath or rapid increase in weight or localised swelling (oedema). Some patients with long-standing type 2 diabetes mellitus and heart disease or previous stroke who are treated with thiazolidinediones in combination with insulin may develop heart failure.
If you have not told your doctor about any of the above, tell them before you use Penfill®.
Tell your doctor if you notice any skin changes at the injection site. The injection site should be rotated to help prevent changes to the fatty tissue under the skin, such as skin thickening, skin shrinking or lumps under the skin. The insulin may not work very well if you inject into a lumpy, shrunken or thickened area (see How to use it). Tell your doctor if you notice any skin changes at the injection site. Tell your doctor if you are currently injecting into these affected areas before you start injecting in a different area. Your doctor may tell you to check your blood sugar more closely, and to adjust your insulin or your other antidiabetic medications dose.
Taking other medicines
Tell your doctor if you are taking any other medicines, including any that you get without a prescription from your pharmacy, supermarket or health food shop.
Some medicines and insulin may interfere with each other. These include:
- other medicines for treatment of diabetes
- monoamine oxidase inhibitors (MAOI) - used to treat depression
- non-selective beta-blockers - used to treat certain heart conditions and high blood pressure
- alpha-blockers - used to treat high blood pressure and to relieve difficulty in passing urine caused by an enlarged prostate
- angiotensin converting enzyme (ACE) inhibitors - used to treat certain heart conditions, high blood pressure or elevated protein/albumin in the urine
- salicylates e.g. aspirin - used to relieve pain and lower fever
- anabolic steroids – used to promote growth
- glucocorticoids (except when applied locally) – used to treat inflammatory conditions
- oral contraceptives (“the pill”) - used for birth control
- thiazides, frusemide or ethacrynic acid - used to treat high blood pressure or fluid retention (oedema)
- thyroid hormones - used to treat malfunction of the thyroid gland
- sympathomimetics - used to treat asthma
- sulphonamides – used to treat bacterial infections
Or other specific medicines including:
- danazol - used to treat certain female conditions
- oxymetholone – used to treat certain blood disorders
- octreotide - used to treat gastrointestinal endocrine tumours and enlargement of parts of the body (e.g. hands, feet, head) caused by abnormal growth hormone levels
- lanreotide – used to treat enlargement of parts of the body (e.g. hands, feet, head) caused by abnormal growth hormone levels
- diazoxide - used to treat high blood pressure
- nicotinic acid - used to treat high cholesterol levels in the blood
- asparaginase - used to treat leukaemia and lymph gland tumours
- quinine – used to treat malaria and relieve muscle cramps
- quinidine – used to treat heart problems
- growth hormone – used to treat growth disorders
Tell your doctor about any other medicines that you are taking. This is very important. Your doctor will advise you if it is alright to keep taking them or if you should stop taking them.
Your doctor and pharmacist have more information on medicines to be careful with or avoid while using this medicine.
How to use insulin Penfill®
Your doctor, diabetes education nurse or pharmacist will have given you advice on how to use your medicine. Carefully follow all the directions. They may differ from the information contained in this leaflet.
Any change in dose or type of insulin should be made cautiously and only under medical supervision. If you change the type of insulin that you use, you may have to use more or less than before. This may happen with the first dose or over a period of time.
If you do not understand the instructions, ask your doctor, diabetes education nurse or pharmacist for help.
How much to use
Your doctor or diabetes education nurse will tell you how much of this medicine you need to use.
It is very important that you manage your diabetes carefully. Too much or too little insulin can cause serious effects.
When to use it
Your doctor or diabetes education nurse will tell you when and how often to inject the insulin. If you are using Actrapid®, Mixtard® 30/70 or Mixtard® 50/50, it is recommended that you eat a meal or a snack containing carbohydrate within 30 minutes of the injection.
How to use it
- Penfill® cartridges are designed to be used with Novo Nordisk insulin delivery devices (such as NovoPen®), and NovoFine® needles.
- Make sure you are using the correct type and strength of insulin. Carefully follow the instructions on how to use your Penfill® in the Novo Nordisk insulin delivery device.
- If you use more than one type of Penfill® (more than one type of insulin), you must use a separate insulin delivery device for each type of Penfill®.
- Do not use the insulin level indicator on the device to measure your dose of insulin.
- You should inject insulin under the skin (subcutaneous injection) as shown to you by your doctor or diabetes education nurse.
- In an emergency, the insulin contained within Actrapid® Penfill® is suitable for intramuscular administration under medical guidance. In an emergency, the insulin contained within Actrapid® Penfill® is also suitable for intravenous administration, but only if administered by a physician. For emergency use, the insulin must first be withdrawn from Penfill® into a syringe.
Discard your Penfill® after emergency use. - In an emergency, the insulins contained within Protaphane®, Mixtard® 30/70 and Mixtard® 50/50 Penfill® are suitable for intramuscular administration under medical guidance. For emergency use, the resuspended insulin must first be withdrawn from Penfill® into a syringe.
Discard your Penfill® after emergency use. - Do not use Actrapid®, Protaphane®, Mixtard® 30/70 or Mixtard® 50/50 Penfill® in insulin pumps.
- Take care not to drop or knock the device that contains Penfill®.
Checking your insulin Penfill®:
Check your Penfill® before each preparation and injection to make sure you are using the correct type and strength of insulin. Disinfect the rubber membrane with a medicinal swab.
Do not use your Actrapid® Penfill® unless the insulin appears clear and colourless. Do not use it if the insulin appears thickened or has bits in it.
Do not use your Protaphane®, Mixtard® 30/70 or Mixtard® 50/50 Penfill® if the insulin is no longer a cloudy, white suspension after shaking, or if it has bits in it.
Always check your Penfill®, including the rubber plunger (stopper). Do not use your Penfill® if any damage is seen or if there is a gap between the plunger and the white label band. If these observations are noted, return your Penfill® to your pharmacist. See the insulin delivery device user manual for further instructions.
Preparing a dose:
- Wash your hands.
IMPORTANT - If your Penfill® contains cloudy insulin please read and follow points 2-5 carefully:
- On the Penfill® cartridge is a coloured band. Do not start an injection with a Penfill® cartridge you have emptied beyond the coloured band. The glass ball must have adequate space to resuspend the insulin. Resuspending is easier when the insulin has reached room temperature.
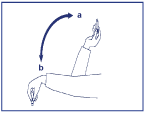
- Before you put Penfill® into the Novo Nordisk insulin delivery device, move it up and down between positions a and b – see picture below - so that the glass ball in the cartridge moves from one end to the other at least 20 times. The movement must always be repeated until the liquid appears uniformly white and cloudy (‘resuspended’).
- If Penfill® is already inside the Novo Nordisk insulin delivery device, turn the delivery device upside-down at least 10 times with the cartridge inside it. Do this again before each subsequent injection.
- Complete the remaining stages of injection without delay.
Checking for insulin flow (priming):
Always check your Novo Nordisk insulin delivery device for insulin flow (priming) before each injection, as described in the device user manual.
The priming procedure may highlight a malfunction with your insulin delivery device. Priming also removes any air bubbles and helps indicate whether or not a needle is broken.
Only dial up your required dose after you see a drop of insulin at the needle tip.
After priming, if you need to put the insulin delivery device down, make sure the needle does not touch anything.
Injecting a dose:
Choose a site for injection.
Inject the medicine into the abdomen, thighs, upper arms or buttocks. Your insulin will work more quickly if you inject into the abdomen.
Change the injection site so that the same position is not used more often than once a month. This reduces the risk of developing lumps or skin pitting.
Pinch the skin between two fingers, push the needle into the raised skin, and inject the full dose of insulin under the skin.
Slowly count to 6 before pulling the needle out.
Keep the push button fully depressed until the needle has been withdrawn. This will ensure correct delivery and limit possible flow of blood into the needle or insulin reservoir.
Apply gentle pressure over the injection site for several seconds.
Do not rub the area.
After injecting:
Dispose of your used needle safely into a yellow plastic sharps container after each injection.
If you are using Actrapid® or Mixtard® insulins, it is recommended that you eat a meal or a snack containing carbohydrate within 30 minutes of the injection.
Do not share needles, insulin cartridges or insulin delivery devices.
How long to use it
Do not stop using insulin unless your doctor tells you to.
If you use too much (overdose) - Hypoglycaemia
Your blood sugar level may become too low (you may experience hypoglycaemia or a “hypo”) if you:
- accidentally use too much of this medicine
- have too much or unexpected exercise
- delay eating meals or snacks
- eat too little food
- are ill.
The first symptoms of mild to moderate hypos can come on suddenly. They may include:
- cold sweat, cool pale skin
- fatigue, drowsiness, unusual tiredness and weakness
- nervousness, anxious feeling, tremor, rapid heart beat
- confusion, difficulty concentrating
- excessive hunger
- vision changes
- headache, nausea.
Always carry some sugary food or fruit juice with you. If you experience any of these symptoms of a hypo, immediately eat some sugary food or have a sugary drink e.g. lollies, biscuits or fruit juice.
Do not inject any insulin if you feel a hypo coming on.
Tell your relatives, friends, close workmates or carers that you have diabetes. It is important that they recognise the signs and symptoms of a hypo. Make sure they know to give you some sugary food or fruit juice for mild to moderate symptoms of a hypo.
If you lose consciousness, make sure they know:
- to turn you on your side and get medical help immediately.
- not to give you anything to eat or drink.
This is because you could choke.
An injection of the hormone glucagon may speed up recovery from unconsciousness. This can be given by a relative, friend, workmate or carer who knows how to give it.
If glucagon is used, eat some sugary food or have a sugary drink as soon as you are conscious again.
If you do not feel better after this, contact your doctor, diabetes education nurse, or the closest hospital.
If you do not respond to glucagon treatment, you will require medical attention.
See your doctor if you keep having hypo reactions, or if you have ever become unconscious after using insulin. Your insulin dose may need to be changed.
If a severe hypo is not treated, it can cause convulsions, brain damage and even death.
If you miss a dose – Hyperglycaemia
If you forget to inject your insulin dose, test your blood sugar level as soon as possible.
If you are not sure what to do, talk to your doctor, diabetes education nurse or pharmacist.
Do not use a double dose of your insulin.
If it is almost time for your next dose, skip the dose you missed and use your next dose when you are meant to.
Otherwise, use it as soon as you remember and then go back to using it as you would normally.
Your blood sugar levels may become high (hyperglycaemia) if you:
- miss doses of insulin or use less insulin than you need
- have uncontrolled diabetes
- exercise less than usual
- eat more carbohydrates than usual
- are ill or stressed.
High blood sugar levels over a long period of time can lead to too much acid in the blood (diabetic ketoacidosis).
Contact your doctor immediately if your blood sugar level is high or you recognise any of the following symptoms.
Symptoms of mild to moderate hyperglycaemia include:
- drowsy feeling
- flushed face
- thirst, loss of appetite
- fruity odour on the breath
- blurred vision
- passing larger amounts of urine than usual
- getting up at night more often than usual to pass urine
- high levels of glucose and acetone in the urine.
Symptoms of severe hyperglycaemia include:
- heavy breathing
- fast pulse
- nausea, vomiting
- dehydration
- loss of consciousness.
Severe hyperglycaemia can lead to unconsciousness and in extreme cases death if untreated.
Discuss any worries you may have about this with your doctor, diabetes education nurse or pharmacist.
While you are using insulin Penfill®
Things you must do
Measure your blood sugar level regularly.
Make sure all friends, relatives, workmates or carers know that you have diabetes. If your child has diabetes it is important to tell their carers.
Keep using your insulin even if you feel well. It helps to control your condition, but does not cure it.
Tell your doctor if you often have hypos (low blood sugar levels). Your doctor may need to adjust your insulin dose.
Always carry some sugary food or fruit juice with you. If you experience any of the symptoms of a hypo, immediately eat some sugary food or have a sugary drink e.g. lollies, biscuits or fruit juice.
Tell your doctor if you have trouble recognising the symptoms of hypos. Under certain conditions, the early warning signs of hypos can be different or less obvious. Your doctor may need to adjust your insulin dose.
Make sure that you tell every doctor, dentist, pharmacist or other health care professional who is treating you that you have diabetes and are using insulin.
Tell your doctor, diabetes education nurse or pharmacist if you are travelling.
Ask them for a letter explaining why you are taking injecting devices with you. Each country you visit will need to see this letter, so you should take several copies.
You may need to inject your insulin and eat your meals at different times because of time differences in and between countries.
You may not be able to get the same type of insulin in the country you are visiting.
Your doctor, diabetes education nurse or pharmacist can provide you with some helpful information.
As a precautionary measure, always carry a spare insulin delivery device in case your Penfill® is lost or damaged.
Things you must not do
Do not stop using your medicine unless your doctor tells you to.
Do not use the medicine if you think it has been frozen or exposed to excessive heat. It will not work as well.
Do not refill your Penfill®.
Do not use this medicine to treat any other complaints unless your doctor tells you to.
Do not give your medicine to anyone else, even if they have the same condition as you.
Things to be careful of
If you drive or use tools or machines, watch out for signs of a hypo. Your ability to concentrate or to react will be less during a hypo. Never drive or use machinery if you feel a hypo coming on. Discuss with your doctor whether you can drive or use machines at all, if you have a lot of hypos or if you find it hard to recognise hypos.
Tell your doctor if you drink alcohol. Alcohol may mask the symptoms of hypos.
Tell your doctor if you are ill. Illness, especially with nausea and vomiting, may cause your insulin needs to change. Even if you are not eating, you still require insulin. You and your doctor should design an insulin plan for those times when you are sick.
Tell your doctor if you are exercising more than usual. Exercise may lower your need for this medicine. Exercise may also speed up the effect of a dose of it, especially if the exercise involves the area of the injection site (e.g. the leg should not be used for injection prior to jogging or running).
Tell your doctor if your diet changes. Changes in diet may cause your insulin needs to change.
Side effects
Tell your doctor, diabetes education nurse or pharmacist as soon as possible if you do not feel well while you are using insulin.
This medicine helps most people for whom it is prescribed, but it may have unwanted side effects in a few people. All medicines can have side effects. Sometimes they are serious, most of the time they are not. You may need medical treatment if you get some of the side effects.
Do not be alarmed by the following lists of side effects. You may not experience any of them.
Ask your doctor, diabetes education nurse or pharmacist to answer any questions you may have.
The most common side effect when using insulin is low blood sugar levels (a hypo).
Tell your doctor if you notice any of the following and they worry you:
- hypos (mild to moderate).
- redness, swelling or itching at the injection site. Usually these symptoms disappear within a few weeks during continued use. If you have serious or continuing reactions, you may need to stop using your current insulin and use another insulin.
- a depression or thickening of the skin around the injection site (lipodystrophy). If you inject insulin at the same place, the fatty tissue may shrink (lipoatrophy) or thicken (lipohypertrophy). Lumps under the skin may also be caused by build-up of a protein called amyloid (cutaneous amyloidosis; how often this occurs is not known). The insulin may not work very well if you inject into a lumpy, shrunken or thickened area. Change the injection site with each injection to help prevent these skin changes.
- when you first start your insulin treatment you may get visual problems or swollen hands and feet.
This list includes the more common side effects of your medicine. They are usually mild and short-lived.
If any of the following happen, tell your doctor immediately or go to Accident and Emergency at your nearest hospital:
More severe symptoms of low blood sugar levels, including:
- disorientation
- seizures, fits or convulsions
- loss of consciousness.
If a severe hypo is not treated, it can cause brain damage and death.
Tell your doctor immediately or go to Accident and Emergency at your nearest hospital if you notice any of the following:
- skin rashes over a large part of the body
- shortness of breath, wheezing
- swelling of the face, lips or tongue
- fast pulse
- sweating.
This list includes very serious side effects. You may need urgent medical attention or hospitalisation. These side effects are very rare.
Tell your doctor if you notice anything that is making you feel unwell.
Other side effects not listed above may also occur in some people.
Ask your doctor, diabetes education nurse or pharmacist to answer any questions you have.
After using insulin Penfill®
Storage
Penfill® that are not being used should be stored between 2°C and 8°C in the refrigerator (away from the cooling element).
Protect the insulin in Penfill® from light by keeping the cartridges in the carton when not in use.
The Penfill® that you are using in your device, or that you are carrying as a spare, does not need to be kept in a refrigerator. You can use it for up to 4 weeks after taking it out of the refrigerator if it is kept below 25ºC.
Discard Penfill® after 4 weeks even if there is still some insulin left in it.
Penfill® must not be frozen, or exposed to excessive heat or light.
Never use insulin after the expiry date printed on the label and carton after ‘Expiry’. The expiry date refers to the last day of that month.
Never use Actrapid® Penfill® if the solution is not clear and colourless.
Never use Protaphane®, Mixtard® 30/70 or Mixtard® 50/50 Penfill® if the suspension does not become white and uniformly cloudy after shaking.
Keep out of the reach of children.
Disposal
Dispose of used needles safely into a yellow plastic sharps container.
If your doctor tells you to stop using this medicine or the expiry date has passed, ask your pharmacist what to do with any medicine that is left over.
Product description
What it looks like
A coloured band is located on the Penfill® cartridge. The colour of the band is specific for the insulin preparation.
Actrapid® is a clear, colourless solution for subcutaneous injection.
Actrapid® Penfill® 3 mL is a glass cartridge.
Protaphane®, Mixtard® 30/70 and Mixtard® 50/50 are white, cloudy suspensions for subcutaneous injection. Protaphane®, Mixtard® 30/70 and Mixtard® 50/50 Penfill® 3 mL are glass cartridges.
Ingredients
Insulin (rys) is characterised by being identical to natural human insulin. The abbreviation “rys” indicates the method of genetic engineering used to manufacture the insulin.
Actrapid® is a neutral solution of insulin (rys) 100 IU/mL. Also contains: glycerol, metacresol, zinc chloride, water for injections. Hydrochloric acid and sodium hydroxide are used to adjust the pH.
Protaphane® is a neutral suspension of isophane insulin (rys) 100 IU/mL. Also contains: glycerol, metacresol, phenol, dibasic sodium phosphate dihydrate, zinc chloride, protamine sulfate (a fish product), water for injections. Hydrochloric acid and sodium hydroxide are used to adjust the pH.
Mixtard® 30/70 is a pre-mixed neutral suspension of insulin (rys) 100 IU/mL, consisting of neutral insulin injection (30%) and isophane insulin injection (70%). Also contains: glycerol, metacresol, phenol, dibasic sodium phosphate dihydrate, zinc chloride, protamine sulfate (a fish product), water for injections. Hydrochloric acid and sodium hydroxide are used to adjust the pH.
Mixtard® 50/50 is a pre-mixed neutral suspension of insulin (rys) 100 IU/mL, consisting of neutral insulin injection (50%) and isophane insulin injection (50%). Also contains: glycerol, metacresol, phenol, dibasic sodium phosphate dihydrate, zinc chloride, protamine sulfate (a fish product), water for injections. Hydrochloric acid and sodium hydroxide are used to adjust the pH.
Sponsor
Penfill® is supplied in Australia by:
Novo Nordisk Pharmaceuticals Pty Ltd
Level 10, 118 Mount Street,
North Sydney NSW 2060,
Australia
This leaflet was prepared in July 2023.
Australian Registration Numbers:
Actrapid® Penfill® 3 mL:
AUST R 169623
Protaphane® Penfill® 3 mL:
AUST R 169635
Mixtard® 30/70 Penfill® 3 mL:
AUST R 169629
Mixtard® 50/50 Penfill® 3 mL:
AUST R 169630
Penfill ®, NovoFine ®, NovoPen ®, Actrapid ®, Protaphane ® and Mixtard ® are registered trademarks of Novo Nordisk A/S.
© 2023
Novo Nordisk A/S
Further information
For further information call Novo Nordisk Customer Care on 1800 668 626.
www.novonordisk.com.au
You can also get more information about diabetes and insulin from Diabetes Australia:
- freecall helpline 1300 136 588
- www.diabetesaustralia.com.au
Published by MIMS February 2026


