1 Name of Medicine
Apomorphine hydrochloride hemihydrate.
2 Qualitative and Quantitative Composition
Each 20 mL cartridge contains 100 mg apomorphine hydrochloride hemihydrate.
Excipients with known effect.
Contains sulfites.
For the full list of excipients, see Section 6.1 List of Excipients.3 Pharmaceutical Form
Solution for injection in cartridge.
Movapo Pod is a clear, colourless to slightly yellow sterile solution, practically free from visible particles.
4.1 Therapeutic Indications
Movapo Pod is indicated to reduce the number and severity of 'off' phases in patients with Parkinson's disease severely disabled by motor fluctuations refractory to conventional therapy. Initiation of therapy with apomorphine should be undertaken in a specialist unit in a hospital setting. Conventional therapy should be continued during 'on' phases.
4.2 Dose and Method of Administration
The optimal dosage of Movapo Pod must be determined on an individual patient basis. Hospital admission under appropriate specialist supervision is advised when establishing a patient's therapeutic regimen.
It is essential that the patient is established on the antiemetic domperidone for at least 48 - 72 hours prior to initiation of therapy.
Patient selection.
For patients in whom conventional therapy has failed, Movapo Pod is only considered to be suitable for Parkinson's disease patients capable of recognising and anticipating 'off' phases in motor performance. Movapo Pod is contraindicated in children and adolescents up to 18 years of age (see Section 4.3 Contraindications).
The elderly are well represented in the population of patients with Parkinson's disease and constitute a high proportion of those studied in clinical trials of apomorphine. The management of elderly patients treated with Movapo Pod is not different from that of younger patients, except for the extra caution on commencing therapy, because of the risk of postural hypotension.
Patients who have shown a good 'on' period response during the initiation stage of apomorphine therapy, but whose overall control remains unsatisfactory using intermittent injections, or who require many and frequent injections, may be commenced on, or transferred to, continuous subcutaneous infusion by minipump and/or syringe driver.
The practical steps described below should be followed when commencing a patient on treatment:
Pretreat with domperidone.
Discontinue all existing antiparkinsonian medication to provoke an 'off' phase in motor performance.
Determine the threshold dose response to apomorphine that produces an unequivocal motor response.
Re-establish other antiparkinsonian agents.
Determine effective treatment regimen for Movapo Pod.
Teach patient and/or carer how and when to administer.
Discharge from clinic or hospital.
Monitor frequently and adjust dosage regimen as appropriate.
Full details are given below.
Pre-treatment.
Domperidone is a peripherally acting dopamine receptor antagonist given by mouth to prevent nausea and vomiting. Domperidone is commenced 48 - 72 hours prior to the first dose of apomorphine. When patients are stabilised with respect to dosage of Movapo Pod, the dose of domperidone is reduced by 10 mg per day every week until mild nausea appears. The maintenance dose of domperidone is the lowest level which completely prevents nausea. Domperidone can usually be withdrawn after several weeks. Before the decision to initiate domperidone and apomorphine treatment, risk factors for QT interval prolongation in the individual patient should be carefully assessed to ensure that the benefit outweighs the risk (see Section 4.4 Special Warnings and Precautions for Use). The cardiovascular assessment should include an ECG and QT measurement. Patients with severe renal insufficiency will require the dosing interval of domperidone to be changed from three times a day to once or twice a day. For further information regarding domperidone refer to the domperidone Product Information.
After provoking an 'off' phase in motor performance.
Determination of the threshold dose.
Movapo injection (10 mg/mL) is recommended.
Initiation of treatment.
Following establishment of an acceptable threshold dose of apomorphine, the patient should be restarted on conventional antiparkinsonian therapy.
See Section 4.2, Patient selection for information on patients who may be suitable for treatment by continuous subcutaneous (SC) infusion.
Continuous subcutaneous infusion of Movapo Pod is administered by infusion pump (Crono APO-Go III or Crono PAR4 20) (see Section 4.2, How to set up the infusion with Movapo Pod). The dose should be titrated to the patient's response. Infusion rates can be commenced at 1 mg/hr, and then increased as necessary. The maximum daily dose should in general not exceed 200 mg/day. In clinical studies the required infusion rate varies between 1.25 and 5.5 mg/hr (equivalent to 0.02 and 0.08 mg/kg/hr), with most patients requiring (a total of) between 2 and 4 mg/hr.
Infusions should be run for waking hours only. Unless the patient is experiencing night-time problems, 24 hour infusions are not advised. Tolerance does not seem to occur unless the overnight period without treatment is less than 4 hours. The infusion site should be changed every 24 hours. Prolonged infusion times are associated with local adverse effects to a more severe degree.
Patients may need to supplement their continuous infusion with intermittent bolus boosts, as necessary, and as directed by their physician.
A reduction in dosage of other dopamine agonists may be considered during continuous infusion.
Do not use if the solution has turned green. The solution should be inspected visually prior to use. Only a clear, colourless to slightly yellow and particle-free solution should be used (also see Section 6.4 Special Precautions for Storage).
Monitoring treatment.
Long term specialist supervision of patients is advised.
There is a high probability of adverse effects to Movapo Pod therapy. The frequency and severity of adverse events should be monitored carefully at regular intervals and a reassessment of the patient carried out if appropriate. Adjustments to the dosage or discontinuation may be necessary.
Method of administration.
Movapo Pod is for subcutaneous use only and must not be administered via intravenous infusion. This product does not require dilution prior to administration.
Movapo Pod is pre-diluted and intended for use as a continuous subcutaneous infusion.
Movapo Pod has been designed to be used with the Crono APO-Go III or Crono PAR4 20 infusion pumps and CronoBell Sleeve (see Section 4.2, How to set up the infusion with Movapo Pod).
Movapo Pod is for single use in one patient only. The solution contains no antimicrobial agent. For microbial reasons, once opened the contents of the cartridge should be used within 48 hours. The infusion site should be changed every 24 hours. Any unused solution should be discarded after 48 hours and a new cartridge used.
Movapo Pod must not be used via the intravenous route.
How to set up the infusion with Movapo Pod.
Wash and dry your hands before handling any infusion equipment.
Make sure you have a "Sharps" bin and these items before you begin:
1 x infusion line;
1 x CronoBell Sleeve;
1 x cartridge (Movapo Pod);
1 x infusion pump (Crono APO-Go III or Crono PAR4 20) with collar attachment;
Tray;
A box of single use sterile Leur caps may also be provided.
Please note that there are different scenarios on how to use and change the cartridge and are shown in the steps below. Please follow the instructions given by your healthcare professional.
Instructions for using the Movapo Pod cartridge for one day. Please follow the steps below if using the Movapo Pod cartridge for one day, without changing the cartridge during the day.
1. Wash and dry your hands.
2. Remove plastic cap from the cartridge and discard.
3. Remove CronoBell Sleeve from its sterile packaging.
4. Connect the infusion line to the top of the CronoBell Sleeve and turn clockwise until tight.
5. Place the cartridge on a flat surface and push the CronoBell Sleeve firmly down onto the cartridge until the rubber bung in the top of the cartridge is pierced.
6. The cartridge, CronoBell Sleeve and infusion line is now ready to be connected to the pump.
7. Slide the CronoBell Sleeve (with cartridge inside) into the collar on the pump aligning the outer lip on the CronoBell Sleeve with the gap collar of the pump.
8. Once inserted, turn the CronoBell Sleeve (with cartridge inside) clockwise until it clicks into place.
9. If instructed by your healthcare professional, the infusion line can be primed. Insert the infusion line, as directed by your healthcare professional.
10. Once the infusion line needle is inserted into the body, the pump can be switched on and the infusion started. At the end of the infusion stop the pump and detach the infusion line from the body/patient. The infusion is generally stopped before sleeping.
11. Ensure the pusher of the pump is fully retracted, once this is done the CronoBell Sleeve (with cartridge inside) and the infusion line can be turned and detached.
12. Dispose of the CronoBell Sleeve (with cartridge inside) and the infusion line in a sharps bin.
Instructions for using the same Movapo Pod cartridge over 2 days. Please use the following steps if using the Movapo Pod cartridge up to a maximum of 48 hours.
1. Wash and dry your hands.
2. Remove the plastic cap from the cartridge and discard.
3. Remove the CronoBell Sleeve from its sterile packaging.
4. Connect the infusion line to the top of the CronoBell Sleeve and turn clockwise until tight.
5. Place the cartridge on a flat surface and push the CronoBell Sleeve firmly down onto the cartridge until the rubber bung in the top of the cartridge is pierced.
6. The CronoBell Sleeve (with cartridge and infusion line) is now ready to be connected to the pump.
7. Slide the CronoBell Sleeve (with cartridge inside) into the collar on the pump aligning the outer lip on the CronoBell Sleeve with the gap in the collar of the pump.
8. Once inserted, turn the CronoBell Sleeve (with cartridge inside) clockwise until it clicks into place.
9. If instructed by your healthcare professional, the infusion line can be primed. Insert the infusion line, as directed by your healthcare professional.
10. Once the infusion line needle is inserted into the body, the pump can be switched on and the infusion started. At the end of the infusion stop the pump. The infusion is generally stopped before sleeping.
How to put the pump with cartridge and CronoBell sleeve away for the night (steps 11-13).
11. Wash and dry your hands as in step 1 and detach the infusion line from the body/patient. Place the pump standing upwards on the tray with the cartridge/sleeve still attached. Detach the infusion line from the pump and dispose of in the sharps bin. Wipe any spillage with the alcohol wipe.
12. Screw the sterile Luer cap onto the CronoBell Sleeve (leaving cartridge inside).
13. Place the tray with the infusion pump in a safe place. Store below 30°C and do not refrigerate. Keep out of reach from children.
How to start the infusion the following morning (steps 14-17).
14. The following day, wash and dry your hands.
15. Unscrew the Luer cap and dispose of in the bin. Clean the top of the CronoBell Sleeve with an alcohol wipe. Immediately, take a new infusion line and connect to the top of the CronoBell Sleeve (with cartridge still inside) by turning clockwise until tight.
16. If instructed by your healthcare professional, the infusion line can be primed. Insert the infusion line, as directed by your healthcare professional.
17. Once the infusion line needle is inserted into the body, the pump can be switched on and the infusion restarted. At the end of the infusion stop the pump and detach the infusion line from the body/patient.
18. Ensure the pusher of the pump is fully retracted, once this is done the CronoBell Sleeve (with cartridge inside) and the infusion line can be turned and detached.
19. Dispose of the CronoBell Sleeve (with cartridge inside) and the infusion line in a sharps bin.
Instructions for changing the Movapo Pod cartridge during the day. If you are using the Movapo Pod cartridge for more than one day, it may be necessary to use a new cartridge when the old one finishes. Please use the following steps when changing the cartridge during the day.
1. Wash and dry your hands.
2. Remove the plastic cap from the new cartridge and discard.
3. Remove the CronoBell Sleeve from its sterile packaging.
4. Place the cartridge on a flat surface and push the CronoBell Sleeve firmly down onto the cartridge until the rubber bung in the top of the cartridge is pierced.
5. The CronoBell Sleeve (with cartridge) is now ready to be connected to the pump. Wipe any spillage with the alcohol wipe.
6. Stop the infusion.
7. Detach the infusion line from the used CronoBell sleeve (with cartridge inside), leaving the infusion line in the skin.
8. Wipe the end of the infusion line with a new alcohol wipe. In order to prevent contamination, do not touch the end of the infusion line except to wipe it.
9. Connect the infusion line to the new CronoBell Sleeve (with cartridge inside).
10. Ensure the pusher of the pump is fully retracted, once this is done the used CronoBell Sleeve (with cartridge inside) can be turned and detached.
11. Dispose of the used CronoBell Sleeve (with cartridge inside) in a sharps bin.
12. Slide the new CronoBell Sleeve (with cartridge inside) into the collar on the pump aligning the outer lip on the CronoBell Sleeve with the gap in the collar of the pump.
13. Once inserted, turn the CronoBell Sleeve (with cartridge inside) clockwise until it clicks into place.
14. The pump can be switched on and the infusion started.
Note.
Different infusion lines require different insertion techniques. The choice of needle is determined by the healthcare professional.
Please refer to user guides for the appropriate pump and CronoBell Sleeve for additional details on setting up Movapo Pod for continuous infusion and additional dosing (boost or bolus dose).
There are differences in the dosing pump of this product and other apomorphine products on the market. Therefore, if switching from or to a different product, patient re-training under the supervision of a healthcare professional is required.
After use, the infusion line, CronoBell Sleeve and cartridge should be discarded and disposed of as per Section 6.6 Special Precautions for Disposal.4.3 Contraindications
Movapo Pod is contraindicated in patients with a known hypersensitivity or allergy to apomorphine, morphine or chemically related products.
Movapo Pod should not be administered to patients with pre-existing neuropsychiatric problems or dementias due to either pathological processes, e.g. Alzheimer's disease, or to patients whose 'on' response to L-dopa is marred by severe dyskinesia, hypotonia or psychotoxicity.
Movapo Pod is also contraindicated in patients with inadequate renal or liver function, unstable coronary vascular disease, cerebrovascular disease, respiratory depression or CNS depression.
Concomitant use with 5HT3 antagonists including antiemetics (for example ondansetron, granisetron, palonosetron) is contraindicated (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
Movapo Pod is contraindicated for children and adolescents under 18 years of age.
Movapo Pod is contraindicated in patients with a known hypersensitivity to sodium metabisulfite.
4.4 Special Warnings and Precautions for Use
For Subcutaneous Use Only (see Section 4.8 Adverse Effects (Undesirable Effects)).
Patients sensitive to morphine or its derivatives may be sensitive to apomorphine. Apomorphine should therefore not be administered to patients with a known hypersensitivity or allergy to apomorphine, morphine or chemically related compounds (see Section 4.3 Contraindications).
Movapo Pod contains sodium metabisulfite which may cause allergic type reactions, including anaphylactic symptoms and life threatening or less severe asthmatic episodes in susceptible people (see Section 4.3 Contraindications).
In patients with cardiac decompensation or cerebrovascular disease, vomiting may cause an increase in blood pressure that may lead to haemorrhage and vascular accidents. Apomorphine is therefore contraindicated in these patients (see Section 4.3 Contraindications).
Caution should be used in administering apomorphine to patients with a predisposition to nausea and vomiting. Apomorphine may cause an increased risk of persistent vomiting. A risk-benefit assessment should be considered in these patients.
Since apomorphine may produce hypotension, even when given with domperidone pre-treatment, care should be exercised in patients with pre-existing cardiac disease or in patients taking vasoactive medicinal products such as anti-hypertensives, and especially in patients with pre-existing postural hypotension.
Since apomorphine, especially at high doses, may have the potential for QT prolongation, caution should be exercised when treating patients at risk for Torsades de pointes arrhythmia.
When used in combination with domperidone, risk factors in the individual patient should be carefully assessed. This should be done before treatment initiation, and during treatment. Important risk factors include serious underlying heart conditions such as congestive cardiac failure, severe hepatic impairment or significant electrolyte disturbance. Also, medication possibly affecting electrolyte balance, CYP3A4 metabolism or QT interval should be assessed.
Monitoring for an effect on the QTc interval is advisable. An ECG should be performed:
prior to treatment with domperidone;
during the treatment initiation phase;
as clinically indicated thereafter.
The patient should be instructed to report possible cardiac symptoms including palpitations, syncope, or near-syncope. They should also report clinical changes that could lead to hypokalaemia, such as gastroenteritis or the initiation of diuretic therapy. At each medical visit, risk factors should be revisited.
Apomorphine is associated with local subcutaneous effects. These can sometimes be reduced by the rotation of injection sites or possibly by the use of ultrasound (if available) in order to avoid areas of nodularity and induration (see Section 4.8 Adverse Effects (Undesirable Effects)).
Haemolytic anaemia and thrombocytopenia have been reported in patients treated with apomorphine. Haematology tests should be undertaken at regular intervals as with levodopa, when given concomitantly with apomorphine.
Caution is advised when combining apomorphine with other medicinal products, especially those with a narrow therapeutic range (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
Neuropsychiatric problems co-exist in many patients with advanced Parkinson's disease. There is evidence that for some patients, neuropsychiatric disturbances may be exacerbated by apomorphine. Special care should be exercised when apomorphine is used in these patients.
The use of apomorphine in conjunction with levodopa treatment may cause Coombs' positive haemolytic anaemia. An initial screen prior to commencement of treatment and at 6 monthly intervals is recommended. In the event of the development of a haemolytic anaemia, a haematological specialist should be consulted. The dose of apomorphine and/or levodopa should be reduced, with careful monitoring of the patient's motor state. It may be necessary to discontinue treatment with levodopa and/or apomorphine in the event that it is not possible to control the anaemia satisfactorily.
Apomorphine has been associated with somnolence, and other dopamine agonists can be associated with sudden sleep onset episodes, particularly in patients with Parkinson's disease.
Patients must be informed of this and advised to exercise caution while driving or operating machines during treatment with apomorphine. Patients who have experienced somnolence, and/or an episode of sudden sleep onset, must refrain from driving or operating machines. Furthermore, a reduction of dosage or termination of therapy may be considered.
Apomorphine should be used with caution in patients with endocrine, renal, pulmonary or cardiovascular disease.
Periodic evaluation of hepatic, haemopoietic, renal and cardiovascular function is advised.
Patients with severe renal insufficiency may require the dosing interval for domperidone to be less frequent (see Section 4.2 Dose and Method of Administration, Pre-treatment).
Neuroleptic malignant syndrome.
Symptoms suggestive of neuroleptic malignant syndrome (characterised by elevated temperature, muscular rigidity, altered consciousness, and autonomic instability) have been reported with abrupt withdrawal of dopaminergic therapy. Therefore, it is recommended to taper treatment (see Section 4.2 Dose and Method of Administration).
Dopamine agonist withdrawal syndrome.
Symptoms suggestive of dopamine agonist withdrawal syndrome (for example, apathy, anxiety, depression, fatigue, sweating, and pain) have been reported with abrupt withdrawal of dopaminergic therapy, therefore, it is recommended to taper treatment (see Section 4.2 Dose and Method of Administration).
Impulse control disorders.
Patients should be regularly monitored for the development of impulse control disorders. Patients and carers should be made aware that behavioural symptoms of impulse control disorders including pathological gambling, increased libido, hypersexuality, compulsive spending or buying, binge eating and compulsive eating can occur in patients treated with dopamine agonists including apomorphine. Dose reduction/tapered discontinuation should be considered if such symptoms develop.
Dopamine Dysregulation Syndrome (DDS) is an addictive disorder resulting in excessive use of apomorphine, seen in some patients treated with apomorphine. Before initiation of treatment, patients and caregivers should be warned of the potential risk of developing DDS.
Use in debilitated patients.
Extra caution is also recommended in debilitated patients, since they may show an increased susceptibility or be more sensitive to the respiratory depressant effects of apomorphine.
Use in the elderly.
Extra caution is also recommended in geriatric patients, since they may show an increased susceptibility or be more sensitive to the respiratory depressant effects of apomorphine. Extra caution is recommended during initiation of therapy in elderly patients because of the risk of postural hypotension.
Paediatric use.
Movapo Pod is contraindicated for children and adolescents under 18 years of age.
Effect on laboratory tests.
Positive Coombs' tests have been reported for patients receiving apomorphine.4.5 Interactions with Other Medicines and Other Forms of Interactions
Patients selected for treatment with apomorphine are almost certain to be taking concomitant medications for their Parkinson's disease. In the initial stages of apomorphine therapy the patient should be monitored for unusual side-effects or signs of potentiation of effect.
Drugs which interfere with central amine mechanisms such as tetrabenazine, metoclopramide, antipsychotic dopamine blocking agents (such as phenothiazines, thioxanthines and butyrophenones), amphetamines and papaverine should be avoided. If their administration is considered essential, extreme care should be taken and the patient monitored for signs of potentiation, antagonism or other interactions and for any unusual adverse effects.
Neuroleptic medicinal products may have an antagonistic effect if used with apomorphine. There is a potential interaction between clozapine and apomorphine.
The possible side effects of apomorphine on the plasma concentrations of other medicinal products have not yet been studied. Therefore, caution is advised when combining apomorphine with other medicinal products, especially those with a narrow therapeutic range.
Antihypertensive and cardiac active medicinal drugs.
Even when co-administered with domperidone, apomorphine may potentiate the antihypertensive effects of antihypertensive and cardiac active medicinal products.
It is recommended to avoid the administration of apomorphine with other drugs known to prolong the QT interval.
Based on reports of profound hypotension and loss of consciousness when apomorphine was administered with ondansetron, the concomitant use of apomorphine with 5HT3 antagonists including antiemetics (for example ondansetron, granisetron, palonosetron) is contraindicated (see Section 4.3 Contraindications).4.6 Fertility, Pregnancy and Lactation
Effects on fertility.
In a fertility study in male rats, fertility was decreased at 2 mg/kg/day SC, one tenth that of the maximum recommended human dose (based on body surface area). Effects on female fertility have not been determined.
(Category B3)
The safety of using apomorphine during pregnancy has not been established in either human or animal studies. Apomorphine should therefore not be used in pregnant women, or those likely to become pregnant.
It is not known whether apomorphine is excreted in breast milk although problems in humans have not been documented. Nevertheless, because many drugs are excreted in human milk and because of the potential for serious adverse drug reactions due to apomorphine in breastfed infants, a decision should be made either to discontinue breastfeeding or the drug, taking into account the benefit of breast-feeding to the child and the benefit of apomorphine to the woman.4.7 Effects on Ability to Drive and Use Machines
Apomorphine has varying degrees of impairment which influences the ability to drive and use machines.
Patients being treated with apomorphine and presenting with somnolence and/or sudden sleep episodes must be informed to refrain from driving or engaging in activities (e.g. operating machines) where impaired alertness may put themselves or others at risk of serious injury of death until such recurrent episodes and somnolence have resolved (see Section 4.4 Special Warnings and Precautions for Use).
4.8 Adverse Effects (Undesirable Effects)
Very common (> 10%).
Itchy nodular lesions at the injection site may be severe in patients on continuous subcutaneous infusions of Movapo Pod. Most patients experience injection site reactions, particularly with continuous use. These may include subcutaneous nodules, induration (see Section 4.4 Special Warnings and Precautions for Use), erythema, tenderness and panniculitis. Various other local reactions (such as irritation, itching, bruising, fibrosis and pain) may also occur (see Section 4.4 Special Warnings and Precautions for Use). Care should be taken to ensure that areas of ulceration do not become infected.
Hallucinations have been reported.
Common (1-10%).
Gastrointestinal side effects including nausea and vomiting appear to be the most prevalent adverse effects, however tolerance to these effects develops rapidly. Pretreatment with domperidone may reduce or prevent these effects (see Section 4.2 Dose and Method of Administration).
Transient sedation with each dose of apomorphine at the start of therapy may occur; this usually resolves over the first few weeks. Apomorphine is associated with somnolence. Drowsiness and sedation occur in most patients on initial treatment but these effects largely subside with repeated dosing, although in some patients these effects may persist. Tachyphylaxis to postural related faintness or syncope also occurs rapidly.
Neuropsychiatric disturbances are common in parkinsonian patients. Apomorphine should be used with special caution in these patients. Neuropsychiatric disturbances (including transient mild confusion and visual hallucinations) have occurred during apomorphine therapy.
Yawning has been reported during apomorphine therapy.
Uncommon (0.1-1%).
Apomorphine may induce dyskinesias during 'on' periods, which can be severe in some cases, and in a few patients may result in cessation of therapy. Apomorphine has been associated with sudden sleep onset episodes (see Section 4.4 Special Warnings and Precautions for Use).
Postural hypotension is seen infrequently and is usually transient (see Section 4.4 Special Warnings and Precautions for Use).
Breathing difficulties have been reported.
Local and generalised rashes have been reported. Injection site necrosis and ulceration have been reported.
Haemolytic anaemia and thrombocytopenia have been reported in patients treated with apomorphine (see Section 4.4 Special Warnings and Precautions for Use).
Rare (0.01-0.1%).
Eosinophilia has rarely occurred during treatment with apomorphine. Peripheral blood eosinophilia, elevated by up to 10%, has occurred in patients on continuous subcutaneous infusion of apomorphine. Blood counts returned to normal in about half of the patients who received treatment over one year.
Due to the presence of sodium metabisulfite, allergic reactions (including anaphylaxis and bronchospasm) may occur.
Not known (cannot be estimated from available data).
Impulse control disorders including pathological gambling, increased libido, hypersexuality, compulsive spending or buying, binge eating and compulsive eating can occur in patients treated with dopamine agonists including apomorphine (see Section 4.4 Special Warnings and Precautions for Use).
Aggression and agitation have also been reported.
Headache has been reported.
Peripheral oedema has been reported.
Dopamine agonist withdrawal syndrome (apathy, anxiety, depression, fatigue, sweating and pain) (see Section 4.4 Special Warnings and Precautions for Use).
Neuroleptic malignant syndrome (see Section 4.4 Special Warnings and Precautions for Use).
Other adverse reactions to apomorphine that have been reported infrequently include stomatitis, confusion, transient rises in serum prolactin, transient metallic taste, rhinorrhoea, increased lacrimation, reduced facial hair growth, loss of libido and spontaneous penile erection.
Thrombus formation and pulmonary embolism have occurred with central venous infusion of apomorphine. Intravenous infusion of the preparation is thus contraindicated.
Reporting suspected adverse effects.
Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.4.9 Overdose
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
Symptoms.
The clinical features of overdose of Movapo Pod are an extension of the pharmacological effects of the drug. They include nausea and persistent vomiting, dyskinesias, hypotension and acute circulatory failure, cardiac arrest, respiratory depression, drowsiness and central nervous system depression or stimulation, euphoria, restlessness and hallucinations and possibly coma and death. Concomitant use of domperidone may exacerbate the clinical features of overdose.
Treatment.
An opioid antagonist such as naloxone may be given to treat excessive vomiting, central nervous system depression and respiratory depression due to Movapo Pod overdose. Excessive vomiting may also be treated with domperidone. Atropine may be also used to treat bradycardia. To treat hypotension, appropriate measures should be taken e.g. raising the foot of the bed.5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action.
Apomorphine is a directly acting dopamine receptor agonist, structurally related to dopamine. Apomorphine has high in vitro binding affinity for the dopamine D4 and D5 receptor (Ki: 4 and 14 nanoM, respectively), moderate affinity (Ki: 26 to 130 nanoM) for the dopamine D2 and D3, adrenergic α1D, α2B, α2C receptors, serotonin 5HT1A, 5HT2A, 5HT2B, and 5HT2C receptors and low affinity for the dopamine D1 receptor (Ki: 370 nanoM). Apomorphine exhibits no affinity for the adrenergic β1 and β2 or histamine H1 receptors.
The effect of apomorphine as an antiparkinsonian agent is believed to be the result of direct stimulation of postsynaptic D2 dopamine receptors, but stimulation of presynaptic D2 dopamine receptors and antagonism of α2 adrenergic receptors may also be important. Apomorphine reduces the tremor, rigidity and bradykinesia in patients receiving levodopa. Apomorphine induces vomiting by direct stimulation of the medullary chemoreceptor trigger zone.
Clinical trials.
No data available.
5.2 Pharmacokinetic Properties
The peripheral pharmacokinetics of apomorphine have been studied following subcutaneous (SC) injection, subcutaneous infusion and intravenous infusion.
Absorption.
Following intramuscular or subcutaneous administration, apomorphine is reported to be well absorbed. Peak plasma concentration occurs as early as three minutes following subcutaneous bolus injection. The rapid and complete absorption from subcutaneous tissues and rapid clearance is believed to correlate with the rapid onset and brief duration of action respectively. Antiparkinsonian effects are observed within five minutes following subcutaneous bolus administration.
Distribution.
The distribution half-life of apomorphine was found to be five minutes. The volume of distribution, plasma clearance and half-life were similar for subcutaneous injection, subcutaneous infusion and intravenous infusion.
Apomorphine reaches a concentration in the brain up to eight times higher than that in plasma, due to high lipid solubility which allows rapid equilibration between blood and tissue compartments.
Metabolism.
Apomorphine is metabolised in the liver. Routes of metabolism in humans include sulfation, N-demethylation, glucuronidation and oxidation to norapomorphine by CYP2B6, CYP2C8 and CYP3A4. The major metabolite in humans after sublingual administration was apomorphine sulfate.
Excretion.
Apomorphine is cleared rapidly. The elimination half-life (t1/2) is about 33 minutes.
5.3 Preclinical Safety Data
Genotoxicity.
In vitro genotoxicity studies demonstrated mutagenic and clastogenic effects, most likely due to products formed by oxidation of apomorphine. Apomorphine was not genotoxic in vivo in a mouse micronucleus test or in a rat unscheduled DNA synthesis test.
Carcinogenicity.
No carcinogenicity studies have been performed.6 Pharmaceutical Particulars
6.1 List of Excipients
Sodium metabisulfite, water for injections, hydrochloric acid.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine. Also see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
To reduce microbiological hazard, use as soon as practicable after opening. Any unused solution should be discarded after 48 hours and a new cartridge used.
6.4 Special Precautions for Storage
Store below 30°C. Do not freeze.
6.5 Nature and Contents of Container
Clear glass (Type 1) siliconized cartridge with a chlorobutyl rubber stopper, aluminum seal (with flip top cap) and siliconized rubber plunger.
Each pack contains 5 x 20 mL (100 mg/20 mL) cartridges in a cardboard tray inside an outer cardboard carton.
The CronoBell sleeve is supplied separately.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of in accordance with local requirements. After use, the infusion line, CronoBell Sleeve and cartridge should also be discarded in accordance with local requirements.
6.7 Physicochemical Properties
The pH of the injection is 3.0 to 4.0.
Chemical structure.
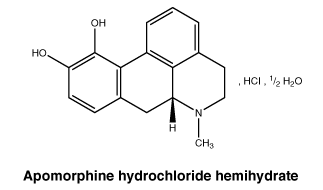
CAS number.
The CAS registry number of apomorphine hydrochloride, hemihydrate is 41372-20-7.7 Medicine Schedule (Poisons Standard)
Schedule 4 - Prescription Only Medicine.
Summary Table of Changes



