Notes
Distributed by Wagner Pharmaceuticals Pty Ltd
1 Name of Medicine
Mupirocin.
2 Qualitative and Quantitative Composition
Each gram of mupirocin ointment contains 20 mg of mupirocin (2% w/w) as the active ingredient.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Mupirocin-WGR ointment is a white, semi-transparent, homogeneous ointment.
4.1 Therapeutic Indications
Mupirocin-WGR is indicated for the topical treatment of mild impetigo.
4.2 Dose and Method of Administration
Mupirocin-WGR is intended for topical application only.
A small amount of mupirocin ointment should be applied to the affected area three times daily. The area treated may be covered with gauze dressing if desired. Usually treatment should not continue for more than 10 days.
Any product remaining at the end of treatment should be discarded.
4.3 Contraindications
Mupirocin-WGR is contraindicated in patients who have demonstrated hypersensitivity to mupirocin, mupirocin calcium, or any components of the formulation.
4.4 Special Warnings and Precautions for Use
If a reaction suggesting sensitivity or chemical irritation should occur with the use of Mupirocin-WGR, treatment should be discontinued, the product should be wiped off and appropriate alternative therapy for the infection instituted.
Mupirocin-WGR is not suitable for ophthalmic use, intranasal use or application to other mucosal surfaces.
Avoid contact with eyes. If contaminated, the eyes should be thoroughly irrigated with water until the residues have been removed.
Mupirocin-WGR is not suitable for application to the site of cannulation or for use in conjunction with cannulae.
As with other antibacterial products, prolonged use may result in overgrowth of nonsusceptible organisms, including fungi. Appropriate measures should be taken if this occurs.
Pseudomembranous colitis has been reported with the use of antibiotics and may range in severity from mild to life threatening. Therefore, it is important to consider its diagnosis in patients who develop diarrhoea during or after antibiotic use. Although this is less likely to occur with topically applied mupirocin, if prolonged or significant diarrhoea occurs or the patient experiences abdominal cramps, treatment should be discontinued immediately and the patient investigated further.
Use in renal impairment.
Polyethylene glycol (macrogol) can be absorbed from open wounds and damaged skin and is excreted by the kidneys. In common with other polyethylene glycol based ointments, Mupirocin-WGR should not be used in conditions where absorption of large quantities of polyethylene glycol is possible, especially if there is evidence of moderate or severe renal impairment.
Use in the elderly.
No restrictions unless there is evidence of moderate or severe renal impairment.
Paediatric use.
The safety and efficacy of mupirocin ointment has not been established in children less than two years of age.
Effects on laboratory tests.
Interactions with laboratory tests have not been established.4.5 Interactions with Other Medicines and Other Forms of Interactions
No drug interactions have been studied with mupirocin.
Mupirocin-WGR should not be combined with other topical preparations as there is a risk of dilution, resulting in a reduction in the antibacterial activity and potential loss of stability of the mupirocin.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility.
Fertility of male and female rats was not affected by mupirocin at subcutaneous doses up to 100 mg/kg/day.
(Category B1)
Reproduction studies have been performed in rats and rabbits at systemic doses up to 160 mg/kg and have revealed no evidence of harm to the foetus due to mupirocin.
There are, however, no adequate and well controlled studies in pregnant women. Because animal studies are not always predictive of human response, mupirocin ointment should be used during pregnancy only if clearly needed.
Adequate human and animal data on use during lactation are not available.
Caution should be exercised when mupirocin ointment is administered to a nursing woman. If a cracked nipple is being treated, the nipple should be thoroughly washed prior to breastfeeding.4.7 Effects on Ability to Drive and Use Machines
The effects of this medicine on a person's ability to drive and use machines were not assessed as part of its registration.
4.8 Adverse Effects (Undesirable Effects)
Systemic allergic reactions such as generalised rash, urticaria and angioedema have been reported with mupirocin ointment.
Local reactions.
Common (approximately 2%): itching, burning, erythema, stinging, pain/ swelling at site of application and dryness. Less than 1% of patients discontinued therapy because of these local reactions.
Uncommon: cutaneous sensitisation reactions to mupirocin or the ointment base.
Gastrointestinal.
One case of nausea has been reported in studies of mupirocin ointment so far.
Mupirocin ointment did not demonstrate any delayed hypersensitivity, cutaneous sensitisation, phototoxicity or photo-contact sensitisation in studies on normal subjects.
Cutaneous sensitisation has been reported rarely in postmarketing surveillance of mupirocin ointment.
Reporting suspected adverse effects.
Reporting suspected adverse reactions after registration of the medical product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at http://www.tga.gov.au/reporting-problems.4.9 Overdose
There is currently limited experience with overdosage of mupirocin ointment.
There is no specific treatment for an overdose of mupirocin. In the event of overdose, the patient should be treated supportively with appropriate monitoring as necessary.
For information on the management of overdose, contact the Poison Information Centre on 131126 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action.
Microbiology.
Mupirocin is a naturally occurring antibiotic, produced by fermentation of the organism Pseudomonas fluorescens.
Mupirocin inhibits bacterial protein synthesis by reversibly and specifically binding to bacterial isoleucyl transfer RNA (tRNA) synthetase. It shows no cross resistance with other commonly used and clinically important antibiotics.
In vitro mupirocin is active mainly against Gram positive aerobes including Staphylococcus aureus (including methicillin resistant Staph. aureus (MRSA) positive strains), Staph. saprophyticus, Staph. epidermidis, Streptococcus pyogenes, Strep. viridans, Strep. agalactiae and Strep. pneumoniae.
Group D Streptococci (including Strep. faecalis and Strep. faecium) are much less sensitive to mupirocin. Most Gram negative organisms (except for Haemophilus influenzae, Neisseria and Branhamella) and anaerobes (including Propionibacterium acnes) are not sensitive to mupirocin.
When mupirocin resistance does occur, it appears to result from the production of a modified isoleucyl tRNA synthetase. High level plasmid mediated resistance (MIC > 1024 microgram/mL) has been reported in some strains of Staph. aureus and coagulase negative staphylococci.
Clinical trials.
No data available.
5.2 Pharmacokinetic Properties
Absorption.
Mupirocin is poorly absorbed through intact human skin; less than 0.24% of a 0.5 g dose being available systemically following the topical application of mupirocin in the ointment base.
Distribution.
Application of 14C-labelled mupirocin ointment to the lower arm of normal male subjects followed by occlusion for 24 hours showed no measurable systemic absorption. Measurable radioactivity was present in the stratum corneum of these subjects 72 hours after application.
Metabolism.
If mupirocin is absorbed through broken skin or is given systemically, it is metabolised to the inactive metabolite, monic acid. The mean plasma half-lives of mupirocin and monic acid are 19 and 77 minutes, respectively.
Excretion.
The major elimination pathway is via the kidney (90%).
5.3 Preclinical Safety Data
Genotoxicity.
Results of the following studies performed with mupirocin calcium or mupirocin sodium in vitro and in vivo did not indicate a potential for mutagenicity: rat primary hepatocyte unscheduled DNA synthesis, sediment analysis for DNA strand breaks, metaphase analysis of human lymphocytes, mouse lymphoma assay and bone marrow micronuclei assay in mice.
Carcinogenicity.
The carcinogenic potential of mupirocin has not been assessed in long-term animal studies.6 Pharmaceutical Particulars
6.1 List of Excipients
The ointment contains the following inactive ingredients: macrogol 400 and macrogol 3350.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 25°C.
6.5 Nature and Contents of Container
Aluminium tube with polyethylene cap containing 15 g of ointment (AUST R 433092).
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
Chemical structure.
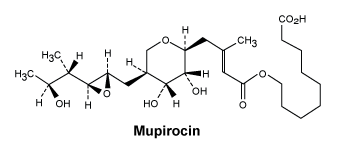 Chemical Name: 9-[[(2E)-4-[(2S,3R,4R,5S)-3,4-Dihydroxy-5-[[(2S,3S)-3- [(1S,2S)-2-hydroxyl-1-methylpropyl]oxiranyl]methyl]tetrahydro-2H-pyran-2-yl]-3-methylbut-2-enoyl]oxy]nonanoic acid.
Chemical Name: 9-[[(2E)-4-[(2S,3R,4R,5S)-3,4-Dihydroxy-5-[[(2S,3S)-3- [(1S,2S)-2-hydroxyl-1-methylpropyl]oxiranyl]methyl]tetrahydro-2H-pyran-2-yl]-3-methylbut-2-enoyl]oxy]nonanoic acid.
Molecular Formula: C26H44O9.
Molecular Weight: 500.622 g/mol.
CAS number.
CAS registry number: 12650-69-0.
Mupirocin is slightly soluble in water and freely soluble in acetone, methylene chloride, chloroform and methanol.7 Medicine Schedule (Poisons Standard)
S4 (Prescription Only Medicine).
Summary Table of Changes
