What is in this leaflet
This leaflet answers some common questions about Nicotinell Chewing Gum.
It does not contain all the available information. It does not take the place of talking to your pharmacist or doctor.
All medicines have risks and benefits. Your pharmacist or doctor will be able to advise you about the risks and benefits of using Nicotinell Chewing Gum.
If you have any concerns about using this medicine, ask your pharmacist or doctor.
Keep this leaflet with the medicine. You may need to read it again.
What is Nicotinell Chewing Gum used for?
Nicotinell Chewing Gum can help you stop smoking over a few months. You can also use it to help you stop smoking gradually over a longer period of time, if you cannot stop smoking immediately.
Whilst giving up isn’t going to happen immediately, it is a habit you can break. But it’s important to understand that there are two sides to quitting smoking.
Firstly there is the psychological dependence on cigarettes. The smoking habit may have become an important part of your life, especially if you have smoked for many years.
Secondly, there is the physical addiction to nicotine to consider. Cigarettes contain nicotine and your body has become dependent on nicotine.
Nicotinell Chewing Gum works on reducing your desire to smoke by providing some of the nicotine that you previously inhaled from cigarettes. This is what helps you to resist smoking.
When chewed, nicotine is released slowly from the gum and absorbed through the lining of the mouth. The amount of nicotine in the gum can also reduce some of the unpleasant effects that often happen when giving up smoking, such as feeling ill or irritable. Nicotinell Chewing Gum does not act as quickly as smoking.
Nicotinell Chewing Gum works most effectively when you have a strong personal commitment to stop smoking. You cannot rely on Nicotinell alone to break the habit. You are more likely to quit smoking when using the product with help from your pharmacist, doctor, a trained counsellor or a support program. Counselling is available from various groups and to get the best out of Nicotinell, we encourage you to enrol in a supportive group.
Before you use Nicotinell Chewing Gum
When you must not use it
Do not use Nicotinell Chewing Gum if:
- Your mouth or throat is sore
- You are under 12 years old
- You are a non-smoker or an occasional smoker
- You are allergic or hypersensitive to nicotine or any of the other ingredients in the gums
- The expiry date printed on the carton or blister platform has passed
- The packaging is torn or shows signs of tampering
There are no health benefits to smoking. It is always better to give up smoking and using Nicotinell can help. In general, any possible side effects associated with nicotine replacement therapy (NRT) are far outweighed by the well established dangers of continuing to smoke.
If you are in hospital because of a heart attack, severe heart rhythm disorders or a stroke, you should try to quit smoking without using NRT unless your doctor tells you to use it. Once you are discharged from the hospital, you may use NRT in consultation with your doctor.
If you have had allergic reactions that involve swelling of the lips, face and throat (angioedema) or itchy skin rash (urticaria), using NRT can sometimes trigger this type of reaction.
Before you start to use it
Talk to your doctor and ask for his/her advice before using Nicotinell Chewing Gum if:
- you have had a recent heart attack or stroke
- you have angina (chest pain) that is not well controlled or is getting worse
- you have severe arrhythmia (irregular heartbeat)
Talk to your doctor or pharmacist and ask for his/her advice before using Nicotinell Chewing Gum if:
- you have any type of heart disease, including angina
- you have had a stroke at any time in the past
- you have high blood pressure or any circulation disorder
- you have kidney or liver problems
- you have a stomach ulcer or persistent stomach upset
- you have hyperthyroidism (a disorder of the thyroid gland)
- you have diabetes, as you should monitor your blood sugar levels more often than usual when starting Nicotinell chewing gum, you may find your insulin or other medication requirements alter
- you have phaeochromocytoma (a tumour of the adrenal gland)
- you have an inability to tolerate fructose (as the gums contain sorbitol, which is a source of fructose)
- you are pregnant or breastfeeding
- you are aged 12 to 17 years
Tell your doctor or pharmacist if you are taking any other medicines, including medicines that you buy without a prescription. Your doctor or pharmacist will be able to advise you if stopping smoking may affect the way these medicines work.
Be careful if you have dentures, dental caps or partial bridges. As with other gums, Nicotinell Chewing Gum may stick to your dentures, dental caps or partial bridges and may damage them. You should stop using the gum if it sticks to your dental work and discuss further use with your dentist.
Driving or operating machinery whilst using Nicotinell Chewing Gum. There is no evidence of any risk associated with driving or operating machinery if Nicotinell Chewing Gum is taken according to the recommended dose but remember that smoking cessation can cause behavioural changes.
Do not drink coffee or soft drinks for 15 minutes before chewing the gum. They may keep the nicotine in the gum from being absorbed properly.
If you are pregnant. Ideally you should not use this program while you are pregnant, and you should stop smoking without using nicotine replacement therapy. Nicotine in any form may cause harm to your unborn baby. However, if you are unable to quit without the use of nicotine replacement therapy, seek advice from your pharmacist or doctor before starting a program. The decision to use NRT should be made as early on in your pregnancy as possible and you should aim to use it for only 2-3 months. Products that are taken intermittently, such as gum, are preferable to nicotine patches. However, patches may be preferred if you have nausea or sickness.
If you are breast-feeding. Nicotine is excreted in breast milk in quantities that may affect the child even in therapeutic doses. Like smoking, nicotine replacement therapy should be avoided during breast-feeding. However, if you are unable to quit without the use of nicotine replacement therapy, seek advice from your pharmacist or doctor before starting a program. You are also recommended to use Nicotinell nicotine chewing gums immediately after breast-feeding, to ensure that the baby gets the smallest amount of nicotine possible.
How to use Nicotinell Chewing Gum
How to get started
Nicotinell Chewing Gum is not like ordinary chewing gum. You must stop smoking completely while you are using it.
It is probably best to nominate a specific day that you will stop smoking.
It may be helpful for you to:
- Try to choose a day when you will not be where others are smoking.
- Choose a day with as little stress as possible.
- Choose a day not too far in the future.
Tell your family and friends that you have set this target “quit day”.
This is the day you take control of your habit and become a non-smoker.
Prepare to stop smoking by trying to reduce the number of cigarettes you smoke each day.
How to use Nicotinell Chewing Gum
At the beginning of the “quit day”, start to use Nicotinell Chewing Gum before any cigarettes have been smoked.
The appropriate dose will depend on your previous smoking habits.
Use 2 mg gum if:
- you were smoking less than 20 cigarettes a day
If, while using the 2 mg gum, your withdrawal symptoms remain so strong as to threaten relapse, then you should use the 4 mg gum.
Use 4 mg gum if:
- you were smoking more than 20 cigarettes a day
- you have previously failed to stop smoking with 2 mg gum
Chew one piece of gum when you feel the urge to smoke.
Follow these 4 easy steps:
- Chew one piece of gum slowly until the taste becomes strong.
- Rest the piece of chewing gum between your gum and cheek.
- Chew again when the taste has faded.
- Repeat the chewing routine for about 30 minutes.
Chew only one piece of gum at a time.
Chew slowly. The nicotine in Nicotinell Chewing Gum is released a little at a time and absorbed through the lining of your mouth into your bloodstream. If you chew too quickly or for too long, you will swallow the nicotine with your saliva. It will be broken down in your stomach and wasted.
Do not use more than 20 pieces of the 2 mg gum or 10 pieces of the 4 mg gum in a day.Most people use 8-12 pieces of the 2mg gum or 4-6 pieces of the 4mg gum in a day.
Usage Program for adults
There are two usage programs you can follow:
Program A. If you would like to stop smoking completely before starting the program:
- After about 3 months, gradually cut down the amount of gum you chew each day.
- When you are down to 1-2 pieces a day, you can stop using Nicotinell Gum.
- You should be able to stop completely within 6 months from the start of the treatment.
Program B. If you cannot stop smoking completely before starting the program:
- Gradually increase gum use, while cutting down smoking.
- When you have cut down the number of cigarettes you smoke to a level you feel you can quit completely, follow Program A.
- If you have not cut down the number of cigarettes you smoke each day after 6 weeks, see you doctor or pharmacist.
Combination therapy
If you have relapsed in the past or if you experience cravings while using a single form of nicotine replacement therapy (NRT), you can combine the use of Nicotinell patch with Nicotinell chewing gum 2 mg.
The combination is more effective than either product alone in people who have been unable to quit smoking using a single NRT method, increasing your chances of successfully quitting.
When using Nicotinell Step 1 patch, chew one piece of Nicotinell chewing gum 2 mg if you get a craving. Use at least 4 pieces of gum and not more than 12 pieces in a day. Continue for 12 weeks.
After 12 weeks, you can wean yourself off therapy by either of the following methods:
- Stop use of Nicotinell patch and gradually reduce the number of Nicotinell chewing gum 2 mg you use until you no longer need them.
- a. Use Nicotinell Step 2 patch for 3-4 weeks, while using the same number of pieces of Nicotinell chewing gum 2 mg in a day that you have routinely used.
b. Then use Nicotinell Step 3 for a further 3-4 weeks, while using the same number of pieces of Nicotinell chewing gum 2 mg in a day that you have routinely used.
c. When patch use is no longer needed, gradually reduce the number of gums you use until you no longer need them.
Children 12 to 17 years old
Do not use for longer than 12 weeks. If you think you need to use for longer than 12 weeks, talk to your doctor or pharmacist.
Chew one piece of gum when you feel the urge to smoke. Follow the directions under ‘How to use Nicotinell Chewing Gum’. Use every 2 to 3 hours. Do not use more than 20 pieces of the 2 mg gum, or 10 pieces of the 4 mg gum in one day.
After about 8 weeks, gradually reduce to 1 to 2 pieces a day and then stop completely.
Do not use for children under 12 years.
While you are using Nicotinell Chewing Gum
Things you must do
Use Nicotinell Chewing Gum as instructed.If you follow these recommendations you should get the full benefit of the Nicotinell Program.
Tell all of the doctors, dentists and pharmacists who are treating you that you are using Nicotinell Chewing Gum.
Things you must not do
Do not stop using Nicotinell Chewing Gum suddenly.You may get side effects similar to those you would get if you stopped smoking suddenly.
Side effects
Tell your pharmacist as soon as possible if you do not feel well while you are using Nicotinell Chewing Gum.
Nicotine, like all other medicines, may cause unwanted side effects in some people.
Some effects, such as headache, irritability, sleep disturbances and light-headedness or dizziness, may be withdrawal symptoms due to low nicotine levels.
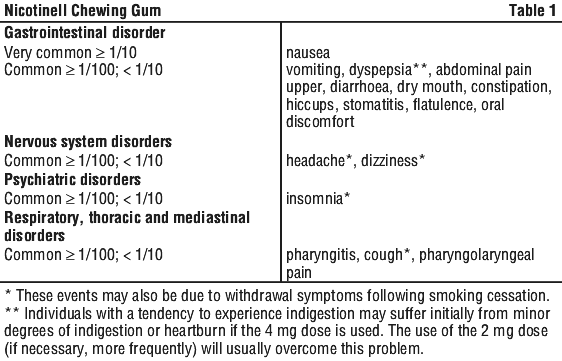
Most of the undesirable effects reported by users of nicotine gums occur during the first few weeks after beginning treatment and may be due to chewing incorrectly or to the effects of nicotine. Such effects include dizziness, headache, stomach trouble such as nausea, vomiting, indigestion or heartburn, increased saliva, hiccups, jaw muscle ache and a sore mouth or throat.
Some less common side effects of nicotine are rapid or irregular heartbeat, palpitations, tingling in the fingers or toes, redness or rash of the skin, hives (urticaria) and allergic reactions.
If you get chest pain, irregular heartbeat or bad indigestion, do not smoke or use Nicotinell or any other nicotine medication. See your pharmacist or doctor as soon as possible.
Other side effects not listed above may also occur in some people. Tell your pharmacist or doctor if these or any other side effects do not go away, or if you notice anything else that is making you feel unwell.
Do not be alarmed by this list of possible side effects. You may not experience any of them.
If you use too much (overdose)
If you smoke or use any other nicotine containing product while you are using Nicotinell Chewing Gum, you may suffer an overdose of nicotine. However if used correctly, nicotine overdose is unlikely.
The general symptoms of nicotine overdosage include: pallor, sweating, burning throat, nausea, salivation, vomiting, stomach upset or abdominal pain, diarrhoea, headache, dizziness, hearing and vision disturbances, tremor, confusion, weakness, seizures (fits), fast or irregular heartbeat, circulatory problems, fainting and breathing difficulties.
The risk of overdose is small as nausea and vomiting usually occurs at an early stage with excessive nicotine intake.
Nicotinell Liquorice Chewing Gum: Individuals who are liquorice-sensitive, along with those who chew more Nicotinell Liquorice Chewing Gum than is recommended, may experience symptoms of water retention, headache, vertigo, increased urination, increased blood pressure and general weakness. If you experience such symptoms, stop using the gums and consult a doctor.
Children: It is very important to keep Nicotinell Chewing Gum out of the reach and sight of children, as doses of nicotine that are tolerated by adult smokers can cause severe poisoning in small children and can be fatal.
In the event of overdose or suspected overdose, seek immediate medical advice or contact a Poisons Information Centre (telephone: Australia 13 11 26; New Zealand 0800 764 766).
After use
Storage
Keep the gum in the blister pack inside the carton until you are ready to use it. If you take the gum out of its blister pack, it may not keep well.
Store the gums in a cool dry place where the temperature stays below 25°C.
Do not store it or any other medicine in the bathroom or near a sink.
Do not leave it in the car or on windowsills. Heat and dampness can destroy some medicines. Keep it where children cannot reach it.A locked cupboard at least one-and-a-half metres above the ground is a good place to store medicines.
Disposal
If your pharmacist or doctor tells you to stop using the gum or you find that it has passed the expiry date, ask your pharmacist what to do with any gum that is left over.
Product description
Nicotinell Chewing Gum is available in two nicotine dosage strengths - Nicotinell 2 mg gum and Nicotinell 4 mg gum.
You can choose from four great flavours in both dosage strengths: Classic, Fruit, Mint and Liquorice.
All Nicotinell Chewing Gums contain nicotine, chewing gum base (containing butylated hydroxytoluene), calcium carbonate, carnauba wax, gelatin, glycerol, mannitol, menthol, polacrilin, sodium bicarbonate, sodium carbonate anhydrous, sorbitol, talc, titanium dioxide, water – purified, xylitol.
Classic Nicotinell Chewing Gum contains maltitol liquid.
Fruit, Mint and Liquorice Nicotinell Chewing Gums contain saccharin, saccharin sodium and acesulfamine potassium.
Flavours : Fruit - fruit flavour. Mint - eucalyptus oil, peppermint oil. Liquorice - eucalyptus oil, anise oil, liquorice root extract. Classic - mild fruit flavour.
All Nicotinell Chewing Gums are sugar-free.
Each gum contains sorbitol, xylitol and mannitol with a combined total of 0.4g per piece. For the 2mg strength, this is equivalent to 8g per maximum dose of 20 pieces. For the 4mg strength, this is equivalent to 4g per maximum dose of 10 pieces. Please note that products containing these ingredients may have a laxative effect or cause diarrhoea.
Each piece of gum also contains 11.5 mg (0.5 mmol) sodium which should be taken into account by those on a low sodium diet. For the 2 mg strength, this is equivalent to 230 mg (10 mmol) sodium per maximum dose of 20 pieces. For the 4 mg strength, this is equivalent to 115 mg (5mmol) sodium per maximum dose of 10 pieces.
For more information
Ask your pharmacist or doctor if you have any questions, or if you experience any difficulties before, during or after using Nicotinell Chewing Gum as they have more complete information.
Nicotinell Chewing Gum is supplied by:
Novartis Consumer Health Australasia Pty Ltd
ABN 46 004 535 513
327 - 333 Police Road
Mulgrave VIC 3170, Australia
NZ Office: Auckland, New Zealand
Toll-free phone:
Australia: 1800 069 643
New Zealand: 0800 700 222
Nicotinell
Try these other flavours in the Nicotinell range
- Classic
- Fruit
- Mint
- Liquorice
Visit our website: www.nicotinell.com.au
This leaflet was prepared in April 2009.
Published by MIMS December 2025


 Nicotine from chewing gum may sometimes cause slight irritation of the mouth and throat and increase salivation at the start of treatment. Excessive swallowing of dissolved nicotine may, at first, cause hiccupping. Those people with a tendency to indigestion may suffer initially from minor indigestion or heartburn. Slower chewing will usually overcome this problem. Excessive consumption of chewing gum by people who have not been in the habit of inhaling tobacco smoke could possibly lead to nausea, faintness or headaches. Less common side effects include palpitations, redness and rashes.
Nicotine from chewing gum may sometimes cause slight irritation of the mouth and throat and increase salivation at the start of treatment. Excessive swallowing of dissolved nicotine may, at first, cause hiccupping. Those people with a tendency to indigestion may suffer initially from minor indigestion or heartburn. Slower chewing will usually overcome this problem. Excessive consumption of chewing gum by people who have not been in the habit of inhaling tobacco smoke could possibly lead to nausea, faintness or headaches. Less common side effects include palpitations, redness and rashes.
