What is in this leaflet
This leaflet answers some common questions about OCTAGAM® 5% [50 mg/ml]. It does not contain complete information about OCTAGAM® 5% [50 mg/ml]. It does not take the place of talking to your doctor. If you have any concerns about using this product, ask your doctor. Follow your doctor's advice even if it is different from what this leaflet says.
Please read this leaflet carefully and keep it for future reference. However, the information in this leaflet is subject to change. Please check with your doctor whether there is any new information about this product that you should know since you were last treated with this product.
What OCTAGAM® 5% [50 mg/ml] is used for
OCTAGAM® 5% [50 mg/ml] is prepared from blood obtained from donors. OCTAGAM® 5% [50 mg/ml] is used for replacement therapy in patients with primary immunodeficiency syndromes, myeloma or chronic lymphocytic leukaemia with severe secondary hypogammaglobulinaemia and recurrent infections or children with congenital Acquired Immune Deficiency Syndrom (AIDS) who have repeated bacterial infections. OCTAGAM® 5% [50 mg/ml] is also used in diseases when the immune system is overactive, these are called autoimmune disorders. OCTAGAM® 5% [50 mg/ml] is used to have an immunomodulatory effect in patients with idiopathic thrombocytopenic purpura, in adults or children with a high risk of bleeding or prior to surgery to correct the platelet count, in Guillain Barré syndrome or Kawasaki disease. OCTAGAM® 5% [50 mg/ml] is also used in allogeneic bone marrow transplantation.
Ask your doctor if you have any questions about why OCTAGAM® 5% [50 mg/ml] has been prescribed for you. Your doctor will have assessed the risks and benefits for you associated with the use of this product.
Before your doctor gives you OCTAGAM® 5% [50 mg/ml]
This product is made from human plasma obtained from donors. When medicines are made from human blood or plasma, certain measures are put in place to prevent infections being passed on to patients.
These include:
- careful selection of blood and plasma donors to make sure those at risk of carrying infections are excluded
- testing of each donation and pools of plasma for signs of virus/infections
- the inclusion of steps in the processing of the blood or plasma that can inactivate or remove viruses.
Despite these measures, when medicines prepared from human blood or plasma are administered, the possibility of passing on infection cannot be totally excluded. This also applies to any unknown or emerging viruses or other types of infections.
The measures taken are considered effective for enveloped viruses such as human immunodeficiency virus (HIV), hepatitis B virus (HBV) and hepatitis C virus (HCV).
The measures taken may be of limited value against the non-enveloped virus hepatitis A and human parvovirus B19.
Immunoglobulins have not been associated with hepatitis A or parvovirus B19 infections possibly because the antibodies against these infections, which are contained in the product, are protective.
Vaccines are available against some of these viruses and your doctor will be able to help you decide whether it is worthwhile having any of those vaccines.
It is strongly recommended that every time you receive a dose of OCTAGAM® 5% [50 mg/ml] the name and batch number of the medicine are recorded in order to maintain a record of the batches used.
Please discuss the risks and benefits of this product with your doctor.
OCTAGAM® 5% [50 mg/ml] must not be used if you have a history of allergy to human immunoglobulin products. Tell your doctor if you have allergies to any other medicines, or if you have ever had an allergic reaction to an injection.
Tell your doctor also if you:
- have previously been advised that you have Immunoglobulin A (IgA) deficiency
- have previously been advised that you have kidney disease
- have previously been advised that you have diabetes
- have or have ever had cerebrovascular disease (such as stroke) or cardiovascular disease (such as a heart attack or angina), including high blood pressure and narrowing or hardening of the arteries
- have a heart condition
- smoke
- have previously had a blood clot in your legs (deep vein thrombosis), lungs (pulmonary embolism) or other parts of your body
- have immediate family members who have had blood clots in the legs, a heart attack, a stroke or high cholesterol
- are taking the contraceptive pill or hormone replacement treatment
- are taking or using any other medicines. These include medicines bought from pharmacies, supermarkets and health food stores
- have any other medical conditions
- have a history of suffering from severe (migraine) headaches
- are pregnant or breast-feeding
- become pregnant during your treatment
- have had any vaccination within the last two weeks or intend to receive one in the next three months
- you are dehydrated
If you want further information, consult your doctor.
How OCTAGAM® 5% [50 mg/ml] is given
Your doctor will determine the dose(s) of OCTAGAM® 5% [50 mg/ml] that you are to receive. Your doctor will give you OCTAGAM® 5% [50 mg/ml] as an infusion, that is, an injection given slowly into the vein.
Unwanted effects
Along with their intended effects, blood products occasionally cause unwanted effects, some of which are serious. Individuals may react differently to similar doses. Most minor effects are related to the rate of infusion and disappear when the rate is slowed down or the infusion stopped.
Unwanted Effects Associated with OCTAGAM® 5% [50 mg/ml]
Unwanted effects which may occur include chills, headache, dizziness, fever, vomiting, diarrhoea, allergic reactions, nausea, joint or muscle pain, changes in blood pressure, chest pain, skin rash, fatigue, flushing, sweating, (rapid) beating of the heart, and moderate low back pain. In addition, human normal immunoglobulins may cause a fall in blood pressure and, in isolated cases, sudden signs of allergy such as rash, itching or hives on the skin, swelling of the face, lips, tongue or other parts of the body, shortness of breath, wheezing or trouble with breathing.
A condition called aseptic meningitis syndrome has been reported to occur infrequently in association with infusions of human normal immunoglobulins. It usually begins within several hours to two days following treatment.
The signs include severe headache (migraine-like), neck stiffness, drowsiness, fever, inability to stand bright light, painful eye movements, and nausea and vomiting. The condition reverses without ill effects when treatment is stopped. You may be more susceptible to this syndrome if you suffer from migraine headaches.
There have been reports that the kidneys may be affected by human normal immunoglobulins.
There have also been reports that OCTAGAM® 5% [50 mg/ml] may lead to the formation of blood clots in some circumstances. This side effect is rare. Very occasionally blood clots may cause serious permanent disabilities, or may even be fatal. Blood clots can form in the veins or the arteries and may result in conditions related to the site of the blood clot, such as heart attack or stroke. The following are the most common symptoms of blood clots:
- unusual pain or swelling in the legs
- sudden chest pain or a feeling of heaviness on your chest which may radiate to the left arm
- sudden shortness of breath or difficulty in breathing
- any unusual, severe or long-lasting headache
- any sudden changes in eyesight (such as loss of vision or blurred vision)
- slurred speech or any other difficulties affecting speech
- dizziness, fainting or fits
- sudden weakness or numbness in one side or part of the body
If you experience any of these symptoms, contact your doctor immediately or go to your nearest hospital emergency department.
If you experience any of the mentioned effects or any other abnormal signs after treatment, contact your doctor immediately.
OCTAGAM® 5% [50 mg/ml] can interfere with some live vaccines (eg. measles, mumps, rubella and polio), even up to three months later. Advise your doctor if you are to receive other vaccines within three months of receiving OCTAGAM® 5% [50 mg/ml].
The maltose in OCTAGAM® 5% [50 mg/ml] can also interfere with blood and urinary glucose tests. Advise your doctor if you are to be tested after receiving OCTAGAM® 5% [50 mg/ml].
Overdose
As OCTAGAM® 5% [50 mg/ml] is given to you under the supervision of your doctor or trained medical professional, it is very unlikely that you will receive an overdose. If you experience several side-effects, tell your doctor immediately and your doctor will know what to do.
Administering a larger than recommended dose may lead to thickening of the blood and expansion of the blood volume, particularly in elderly patients and patients with kidney problems.
How to store OCTAGAM® 5% [50 mg/ml]
Store below 25°C. Do not freeze. Protect from light.
Once opened OCTAGAM® 5% [50 mg/ml] should be used immediately. Any remaining contents should be discarded.
Do not use after the expiry date.
Further information
OCTAGAM® 5% [50 mg/ml] can only be obtained on a doctor's prescription. This leaflet does not contain the complete information about OCTAGAM® 5% [50 mg/ml]. If you require further information about OCTAGAM® 5% [50 mg/ml] and your treatment generally, or if you have any questions or are not sure about something in this leaflet, consult your doctor.
Product Description
Ingredients
In each vial of OCTAGAM® 5% [50 mg/ml] is a sterile solution containing blood proteins of which at least 95% is immunoglobulins. It also contains maltose (a sugar).
Sponsor
OCTAGAM® 5% [50 mg/ml] is supplied by:
Octapharma Australia Pty. Ltd.
Jones Bay Wharf
42/26-32 Pirrama Road
Pyrmont NSW 2009
Australia
Australian Registration Number:
20 mL AUST R 113925
50 mL AUST R 113926
100 mL AUST R 113927
200 mL AUST R 113928
This leaflet was prepared in October 2016.
Published by MIMS May 2017

 Cases of reversible aseptic meningitis, isolated cases of reversible haemolytic anaemia/ haemolysis and rare cases of transient cutaneous reactions, have been observed with human normal immunoglobulin.
Cases of reversible aseptic meningitis, isolated cases of reversible haemolytic anaemia/ haemolysis and rare cases of transient cutaneous reactions, have been observed with human normal immunoglobulin. For information on viral safety, see Section 4.4 Special Warnings and Precautions for Use.
For information on viral safety, see Section 4.4 Special Warnings and Precautions for Use.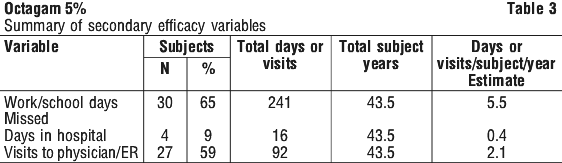 In an open-labelled single-centre study, 17 adolescent patients with PID received Octagam 5% individualised doses of 0.2 to 0.4 g/kg every 3 weeks for a 6 months period. All patients were previously treated with IVIg preparations.
In an open-labelled single-centre study, 17 adolescent patients with PID received Octagam 5% individualised doses of 0.2 to 0.4 g/kg every 3 weeks for a 6 months period. All patients were previously treated with IVIg preparations.