What is in this leaflet
This leaflet answers some common questions about OLMERTAN.
It does not contain all the available information.
It does not take the place of talking to your doctor or pharmacist.
All medicines have risks and benefits. Your doctor has weighed the risks of you taking OLMERTAN against the benefits he/she expects it will have for you.
If you have any concerns about taking this medicine, ask your doctor or pharmacist.
Keep this leaflet with your OLMERTAN tablets. You may need to read it again.
What OLMERTAN is used for
OLMERTAN is used to treat high blood pressure which is sometimes called hypertension.
Everyone has blood pressure. This pressure helps push blood all around your body. Your blood pressure changes at different times of the day, depending on how busy or worried you are. You have hypertension (high blood pressure) when your blood pressure stays higher than is needed, even when you are calm and relaxed.
There are usually no symptoms of hypertension. The only way of knowing that you have hypertension is to have your blood pressure checked on a regular basis. If high blood pressure is not treated it can lead to serious health problems, including stroke, heart disease and kidney failure.
How OLMERTAN works
OLMERTAN belongs to a group of medicines known as angiotensin-II receptor antagonists. Angiotensin-II is a substance produced in the body which causes blood vessels to tighten. OLMERTAN blocks the action of angiotensin-II and therefore relaxes your blood vessels. This helps lower your blood pressure.
Your doctor may have prescribed OLMERTAN for another reason. Ask your doctor if you have any questions about why OLMERTAN has been prescribed for you.
OLMERTAN is not addictive.
This medicine is available only with a doctor's prescription.
Before you take OLMERTAN
When you must not take it
Do not take OLMERTAN if:
- you have serious problems with your kidneys
- you have serious problems with your liver
- you have diabetes and are taking a medicine called aliskiren to reduce blood pressure
- you have an allergy to OLMERTAN or any of the ingredients listed at the end of this leaflet
Symptoms of an allergic reaction to OLMERTAN may include skin rash, itchiness, shortness of breath, swelling of the face, lips or tongue, muscle pain or tenderness or joint pain.
Do not take OLMERTAN if you are pregnant or breastfeeding. OLMERTAN may enter your womb or it may pass into the breast milk and there is the possibility that your baby may be affected.
If pregnancy is discovered OLMERTAN should be discontinued as soon as possible.
Do not take OLMERTAN after the expiry date printed on the pack or if the packaging is torn or shows signs of tampering.
If you are not sure whether you should start taking OLMERTAN, talk to your doctor.
Before you start to take it
Tell your doctor if you have allergies to:
- any other medicines
- any other substances, such as foods, preservatives or dyes.
Tell your doctor if you have or have had any medical conditions, especially the following:
- kidney problems
- liver problems
- heart problems
- excessive vomiting or diarrhea recently
- high levels of potassium in your blood
- problems with your adrenal glands (small glands above the kidneys).
You must also tell your doctor if you:
- are following a very low salt diet
- you are or intend to become pregnant or plan to breastfeed.
- you are taking potassium supplements, potassium-sparing agents, potassium-containing salt substitutes or other medicines that may increase serum potassium (e.g., trimethoprim-containing products)
If you have not told your doctor about any of the above, tell him/ her before you start taking OLMERTAN.
Taking other medicines
Tell your doctor or pharmacist if you are taking any other medicines, including any that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines and OLMERTAN may interfere with each other. These include:
- other medicines used to treat high blood pressure
- other diuretics, also known as fluid or water tablets
- non-steroidal anti-inflammatory drugs (NSAIDs) or COX-2 inhibitors, medicines used to relieve pain, swelling and other symptoms of inflammation, including arthritis
- potassium supplements or potassium-containing salt substitutes
- other medicines that may increase serum potassium (e.g., trimethoprim-containing products)
- lithium, a medicine used to treat mood swings and some types of depression
- any medicines that contain aliskiren
- any medicines that contain colesevelam.
These medicines may be affected by OLMERTAN, or may affect how well it works. You may need different amounts of your medicine, or you may need to take different medicines. Your doctor will advise you.
In general, OLMERTAN has been shown not to interfere with other medicines. However, it is important to tell your doctor and pharmacist about any medicines you are taking or plan to take.
Use in children
The safety and effectiveness of OLMERTAN in children aged less than 1 year has not been established.
How to take OLMERTAN
Follow all directions given to you by your doctor or pharmacist carefully. They may differ from the information contained in this leaflet.
Your doctor or pharmacist will tell you which OLMERTAN tablet you will need to take each day. This depends on your condition and whether or not you are taking any other medicines.
If you do not understand the instructions on the box, ask your doctor or pharmacist for help.
How much to take
Tablets:
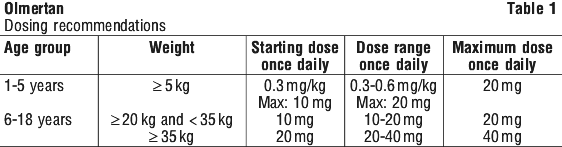
For most adult patients, the usual starting dose of OLMERTAN is 20 mg once a day. The dose may need to be increased to OLMERTAN 40 mg once a day if necessary.
For children 6-18 years (weighing 35 kg or more):
One OLMERTAN 20 mg tablet once daily.
For children 1-5 years (weighing 5 kg or more):
The dose will vary depending on your child's weight.
Note: For children who cannot swallow tablets, the equivalent dose may be given as an extemporaneous suspension. This brand of product has not been approved for use as an extemporaneous suspension. An alternative brand approved for use to compound an extemporaneous suspension is to be used if treatment is required in the suspension form.
How to take it
Tablets:
Swallow OLMERTAN whole with a full glass of water. Do not chew the tablets.
When to take it
Take OLMERTAN at about the same time each day, with or without food. Taking OLMERTAN at the same time each day will have the best effect. It will also help you remember when to take OLMERTAN. It does not matter whether you take it with or without food.
How long to take it
OLMERTAN helps control your condition, but does not cure it. Therefore you must take OLMERTAN every day. Continue taking your medicine for as long as your doctor tells you.
If you forget to take it
If it is almost time for your next dose, skip the dose you missed and take your next dose when you are meant to.
Otherwise, take it as soon as you remember and then go back to taking your medicine as you would normally.
Do not take a double dose to make up for the dose that you missed. This may increase the chance of you getting an unwanted side effect.
If you are not sure what to do, ask your doctor or pharmacist.
If you have trouble remembering to take your medicine, ask your pharmacist for some hints.
If you take too much (overdose)
Immediately telephone your doctor or the Australian Poisons Information Centre (telephone 13 11 26) or go to Accident and Emergency at the nearest hospital, if you think that you or anyone else may have taken too much OLMERTAN. Do this even if there are no signs of discomfort or poisoning.
Keep telephone numbers for these places handy.
If you take too much OLMERTAN, you may feel light-headed, dizzy or you may faint. You may also have a fast heartbeat.
While you are using OLMERTAN
Things you must do
If you are about to be started on any new medicine, tell your doctor, dentist or pharmacist that you are taking OLMERTAN.
Make sure you drink enough water during exercise and hot weather when you are taking OLMERTAN, especially if you sweat a lot. If you do not drink enough water while taking OLMERTAN, you may feel faint, light-headed or sick. This is because your blood pressure is dropping suddenly. If you continue to feel unwell, tell your doctor.
If you have excess vomiting and/or diarrhoea while taking OLMERTAN, tell your doctor. You may lose too much water and salt and your blood pressure may drop too much.
If you feel light-headed or dizzy after taking your first dose of OLMERTAN, or when your dose is increased, tell your doctor immediately.
If you plan to have surgery that needs a general anaesthetic, tell your doctor or dentist that you are taking OLMERTAN. Your blood pressure may drop suddenly.
If you become pregnant while taking OLMERTAN, tell your doctor immediately.
If you are about to have any blood tests, tell your doctor that you are taking OLMERTAN. OLMERTAN may interfere with the results of some tests.
Have your blood pressure checked when your doctor says, to make sure OLMERTAN is working.
Go to your doctor regularly for a check-up. Your doctor may occasionally do a blood test to check your potassium levels and see how your kidneys are working.
Things you must not do
Do not give OLMERTAN to anyone else, even if they have the same condition as you.
Do not take OLMERTAN to treat any other complaints unless your doctor or pharmacist tells you to.
Do not stop taking OLMERTAN, or lower the dosage, without checking with your doctor.
Things to be careful of
If you feel light-headed, dizzy or faint when getting out of bed or standing up, get up slowly. Standing up slowly, especially when you get up from bed or chairs, will help your body get used to the change in position and blood pressure. If this problem continues or gets worse, talk to your doctor.
Be careful driving or operating machinery until you know how OLMERTAN affects you. As with other medicines in this class, OLMERTAN may cause dizziness, light-headedness or tiredness in some people. Make sure you know how you react to OLMERTAN before you drive a car, operate machinery, or do anything else that could be dangerous if you are dizzy or light-headed. If this occurs do not drive. If you drink alcohol, dizziness or light-headedness may be worse.
Things that would be helpful for your blood pressure
Some self help measures suggested below may help your condition. Talk to your doctor or pharmacist about these measures and for more information.
- Alcohol - your doctor may advise you to limit your alcohol intake.
- Weight - your doctor may suggest losing some weight to help lower your blood pressure and help lessen the amount of work your heart has to do. Some people may need a dietician's help to lose weight.
- Diet - eat a healthy diet which includes plenty of fresh vegetables, fruit, bread, cereals and fish.
Also eat less fat and sugar (especially saturated fat) which includes sausages, fatty meats, full cream dairy products, biscuits, cakes, pastries, chocolates, chips and coconut. Monounsaturated and polyunsaturated fats from olive oil, canola oil, avocado and nuts are beneficial in small quantities. - Salt - your doctor may advise you to watch the amount of salt in your diet. To reduce your salt intake you should avoid using salt in cooking or at the table and avoid cooked or processed foods containing high sodium (salt) levels.
- Exercise - regular exercise, maintained over the long term, helps to reduce blood pressure and helps get the heart fitter. Regular exercise also improves your blood cholesterol levels, helps reduce your weight and stress levels, and improves your sleep, mood and ability to concentrate. However, it is important not to overdo it. Walking is good exercise, but try to find a route that is reasonably flat.
Before starting any exercise, ask your doctor about the best kind of programme for you. - Smoking - your doctor may advise you to stop smoking or at least cut down. There are enormous benefits to be gained from giving up smoking. There are many professionals, organisations and strategies to help you quit. Ask your doctor or pharmacist for further information and advice.
Side effects
Tell your doctor or pharmacist as soon as possible if you do not feel well while you are taking OLMERTAN.
OLMERTAN helps most people with high blood pressure, but it may have unwanted side effects in a few people. All medicines can have side effects. Sometimes they are serious, most of the time they are not. You may need medical treatment if you get some of the side effects.
If you are over 65 years of age you may have an increased chance of getting side effects.
Ask your doctor or pharmacist to answer any questions you may have.
Following is a list of possible side effects. Do not be alarmed by this list. You may not experience any of them.
Tell your doctor or pharmacist if you notice any of the following and they worry you:
- feeling light-headed, dizzy or faint
- cough
- headache
- feeling sick (nausea) or vomiting
- stomach pain
- diarrhea
- an uncomfortable feeling in the stomach
- unusual tiredness or weakness, fatigue
- 'flu-like' symptoms
- runny or blocked nose, or sneezing
- bronchitis
- sore throat and discomfort when swallowing (pharyngitis)
- swelling of the hands, feet or ankles
- back pain
- urinary tract infection.
These are common side effects. They are generally mild.
Tell your doctor as soon as possible if you notice any of the following:
- skin rash or itchiness
- aching, tender or weak muscles not caused by exercise
- painful joints
- fast heart beat
- shortness of breath or tightness in the chest
- swelling of the hands, feet or ankles
- symptoms that may indicate high potassium levels in the blood, such as nausea, diarrhoea, muscle weakness, change in heart rhythm.
These may be serious side effects. You may need medical attention. Serious side effects are rare.
If any of the following happen, stop taking OLMERTAN and either tell your doctor immediately or go to Accident and Emergency at your nearest hospital:
- swelling of the face, lips, mouth, tongue or throat which may cause difficulty in swallowing or breathing
- chest pain.
These are very serious side effects. You may need urgent medical attention or hospitalisation. These side effects are very rare.
Other side effects not listed above may occur in some patients. Tell your doctor or pharmacist if you notice anything that is making you feel unwell.
After using OLMERTAN
Storage
Tablets:
Keep your tablets in the box until it is time to take them. If you take the tablets out of the box they will not keep well.
Store below 25°C in a dry place. Do not store OLMERTAN or any other medicine in the bathroom or near a sink.
Do not leave it on a windowsill or in the car on hot days. Heat and dampness can destroy some medicines.
Keep it where children cannot reach it. A locked cupboard at least one-and-a-half metres above the ground is a good place to store medicines.
Disposal
If your doctor or pharmacist tells you to stop taking OLMERTAN or the tablets have passed their expiry date, ask your pharmacist what to do with any that are left over.
Product description
What it looks like
OLMERTAN tablets come in two strengths and each has a different appearance:
- OLMERTAN 20 mg- White to off white coloured, round shape film coated tablets debossed with OLM on one side & 20 on other side.
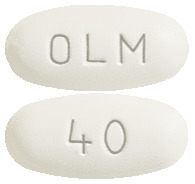
- OLMERTAN 40 mg- White to off white coloured, oval shape film coated tablets debossed with OLM on one side & 40 on other side.
A box of OLMERTAN contains 30 tablets.
Ingredients
Active ingredients:
- OLMERTAN 20 mg tablet - 20 mg olmesartan medoxomil per tablet
- OLMERTAN 40 mg tablet - 40 mg olmesartan medoxomil per tablet.
Other ingredients:
OLMERTAN tablets:
- Lactose monohydrate,
- Microcrystalline cellulose,
- Hydroxypropyl cellulose,
- Low-substituted hydroxypropyl cellulose,
- Magnesium stearate,
- Stearic acid
- OPADRY white (03A28718) that contains titanium dioxide (E171), talc (E553b), hypromellose (E464).
OLMERTAN tablets do not contain sucrose, tartrazine or any other azo dyes.
Sponsor
OLMERTAN is sponsored in Australia by:
Pharmacor Pty Ltd.
CHATSWOOD, NSW, 2067
Australia
Distributor
Arrotex Pharmaceuticals Pty Ltd
15-17 Chapel Street
Cremorne VIC 3121
Australia
Australian Registration Numbers
- OLMERTAN 20 mg tablets – AUST R 235830
- OLMERTAN 40 mg tablets – AUST R 235831
This leaflet was prepared in December 2022
Published by MIMS February 2023


 Other adverse events of potential clinical relevance reported in the clinical trials are listed below. Adverse events reported across all clinical trials with olmesartan medoxomil (including trials with active as well as placebo control), irrespective of causality or incidence relative to placebo, included those events listed below. Frequencies are defined as: common (≥ 1/100, < 1/10); uncommon (≥ 1/1,000, < 1/100); rare (≥ 1/10,000, < 1/1,000), very rare (< 1/10,000).
Other adverse events of potential clinical relevance reported in the clinical trials are listed below. Adverse events reported across all clinical trials with olmesartan medoxomil (including trials with active as well as placebo control), irrespective of causality or incidence relative to placebo, included those events listed below. Frequencies are defined as: common (≥ 1/100, < 1/10); uncommon (≥ 1/1,000, < 1/100); rare (≥ 1/10,000, < 1/1,000), very rare (< 1/10,000). The most common adverse events considered to be treatment related in elderly patients were headache (1.5%) and dizziness (1.1%) on 40 mg olmesartan medoxomil.
The most common adverse events considered to be treatment related in elderly patients were headache (1.5%) and dizziness (1.1%) on 40 mg olmesartan medoxomil.
 Data above from seven placebo-controlled studies also confirm that the blood pressure lowering effect was maintained throughout the 24-hour period with olmesartan medoxomil once daily, with trough-to-peak ratios for systolic and diastolic response between 60 and 80%.
Data above from seven placebo-controlled studies also confirm that the blood pressure lowering effect was maintained throughout the 24-hour period with olmesartan medoxomil once daily, with trough-to-peak ratios for systolic and diastolic response between 60 and 80%. The blood pressure lowering effect of olmesartan medoxomil, with and without hydrochlorothiazide, was maintained in patients treated for up to 1-year. There was no evidence of tachyphylaxis during long-term treatment with olmesartan medoxomil or rebound effect following abrupt withdrawal of olmesartan medoxomil after 1-year of treatment.
The blood pressure lowering effect of olmesartan medoxomil, with and without hydrochlorothiazide, was maintained in patients treated for up to 1-year. There was no evidence of tachyphylaxis during long-term treatment with olmesartan medoxomil or rebound effect following abrupt withdrawal of olmesartan medoxomil after 1-year of treatment. The study included three periods: a 3 week double-blind, randomised, dose-response period for patients aged 6-16 years or, for patients aged 1-5 years, an open-label dose period; up to 2 week double-blind, randomised, placebo-controlled withdrawal period; and a 46 week open-label safety and efficacy period. The primary endpoints were the dose response in systolic blood pressure or in diastolic blood pressure for subjects 6 to 16 years of age at the end of this period. This study was not a clinical outcome study.
The study included three periods: a 3 week double-blind, randomised, dose-response period for patients aged 6-16 years or, for patients aged 1-5 years, an open-label dose period; up to 2 week double-blind, randomised, placebo-controlled withdrawal period; and a 46 week open-label safety and efficacy period. The primary endpoints were the dose response in systolic blood pressure or in diastolic blood pressure for subjects 6 to 16 years of age at the end of this period. This study was not a clinical outcome study. At the end of the open-label efficacy and safety period, compared to baseline, the mean systolic and diastolic blood pressure were reduced at all visits for all patient age groups. However data in children 1-5 years are limited due to small numbers of patients enrolled in the clinical studies. Overall the clinical trials were unable to demonstrate that olmesartan medoxomil was significantly better than placebo in reducing blood pressure in children 1-5 years of age.
At the end of the open-label efficacy and safety period, compared to baseline, the mean systolic and diastolic blood pressure were reduced at all visits for all patient age groups. However data in children 1-5 years are limited due to small numbers of patients enrolled in the clinical studies. Overall the clinical trials were unable to demonstrate that olmesartan medoxomil was significantly better than placebo in reducing blood pressure in children 1-5 years of age.
 Chemical name: 2,3-dihydroxy-2-butenyl 4-(1-hydroxy-1-methylethyl)-2-propyl-1-[p-(o-1H-tetrazol-5-ylphenyl) benzyl] imidazole-5-carboxylate, cyclic 2,3-carbonate.
Chemical name: 2,3-dihydroxy-2-butenyl 4-(1-hydroxy-1-methylethyl)-2-propyl-1-[p-(o-1H-tetrazol-5-ylphenyl) benzyl] imidazole-5-carboxylate, cyclic 2,3-carbonate.