What is in this leaflet
This leaflet answers some common questions about OPSUMIT.
It does not contain all the available information. It does not take the place of talking to your doctor or pharmacist.
All medicines have risks and benefits. Your doctor has weighed the risks of you using this medicine against the benefits they expect it will have for you.
If you have any concerns about using this medicine, speak to your doctor or pharmacist.
Keep this leaflet with the medicine. You may need to read it again.
What OPSUMIT is used for
OPSUMIT tablets contain macitentan which belongs to the class of medicines called "endothelin receptor antagonists".
OPSUMIT is used for the treatment of pulmonary arterial hypertension (PAH), it can be used on its own or with other drugs to treat PAH. PAH is high blood pressure in the blood vessels (the pulmonary arteries) that carry blood from the heart to the lungs. In people with PAH, these arteries get narrower, so the heart has to work harder to pump blood through them. This causes people to feel tired, dizzy, and short of breath.
OPSUMIT widens the pulmonary arteries, making it easier for the heart to pump blood through them. This lowers the blood pressure and relieves the symptoms and improves the course of the disease.
Your doctor however, may prescribe OPSUMIT for another purpose.
Ask your doctor if you have any questions about why it has been prescribed for you.
Before you take OPSUMIT
When you must not take it
DO NOT take OPSUMIT if you are pregnant or if you are planning to become pregnant, or if you could become pregnant because you are not using reliable birth control (contraception). Talk to your doctor about what birth control methods are reliable whilst taking OPSUMIT.
Do not take OPSUMIT if you have ever had an allergic reaction to macitentan, soya or any of the ingredients listed at the end of this leaflet.
Symptoms of an allergic reaction may be mild or severe. They usually include some or all of the following:
- wheezing, swelling of the lips/mouth, difficulty in breathing, hay fever, lumpy rash (hives) or fainting.
Do not take OPSUMIT if you have liver disease or if you have very high levels of liver enzymes in your blood.
Talk to your doctor, who will decide if this medicine is suitable for you.
Do not use OPSUMIT after the expiry date [EXP.] printed on the pack. If you use it after the expiry date has passed, it may not work as well.
Do not use OPSUMIT if the packaging is torn or shows signs of tampering.
If you're not sure whether you should be using OPSUMIT, talk to your doctor.
The safety and efficacy of OPSUMIT has not been proven in patients under 12 years of age.
Before you start to take it
You must tell your doctor if:
- you are allergic to foods, dyes, preservatives or any other medicines,
- you have an intolerance to lactose or any other sugars. OPSUMIT contains small amounts of a sugar called lactose,
- you are pregnant, trying or become pregnant,
- you are breast-feeding,
- you have liver problems,
- you have kidney problems,
- you have low blood pressure,
- you have anaemia or low red blood cells.
- you are 75 years or older
Blood Tests
Your doctor will order some blood tests before you start treatment with OPSUMIT and also during treatment with OPSUMIT. These are to test:
- whether you have anaemia (a reduced number of red blood cells)
- whether your liver is working properly.
Signs that your liver may not be working properly include:
- nausea (urge to vomit)
- vomiting
- fever (high temperature)
- pain in your stomach (abdomen)
- jaundice (yellowing of your skin or the whites of your eyes)
- dark-coloured urine
- itching of your skin
- lethargy or fatigue (unusual tiredness or exhaustion)
- flu-like syndrome (joint and muscle pain with fever)
If you notice any of these signs, tell your doctor immediately.
Pregnancy and Breast-feeding
DO NOT take OPSUMIT if you or your partner are pregnant or planning to become pregnant as it may harm unborn babies conceived before, during or soon after treatment.
You must not become pregnant for at least 3 months after stopping OPSUMIT.
If you are a woman who could become pregnant, your doctor will ask you to take a pregnancy test before you take OPSUMIT and regularly (once a month) while you are taking OPSUMIT.
If it's possible you could become pregnant, use at least two reliable forms of birth control (contraception) while you're taking OPSUMIT. You must continue to use contraception for at least 3 months after stopping OPSUMIT. Talk to your doctor about this.
If you do become pregnant, talk to your doctor immediately.
DO NOT breast feed while you are taking OPSUMIT. You are advised to stop breastfeeding if OPSUMIT is prescribed for you, because it is not known whether this medicine passes into human breast milk.
Male fertility
If you are a male you should avoid exposing your partner to your semen by use of appropriate contraception.
OPSUMIT may lower your sperm count. Talk to your doctor if you have any questions or concerns about this.
Taking other medicines
Tell your doctor if you are taking any other medicines, including medicines you buy without a prescription from a pharmacy, supermarket or health food shop.
You must tell you doctor if you are taking:
- rifampicin, clarithromycin, ciprofloxacin, erythromycin (to treat infections)
- phenytoin (to treat seizures)
- carbamazepine (to treat depression and epilepsy)
- St. John’s Wort (a herbal preparation to support healthy mood balance)
- ritonavir, saquinavir (to treat HIV infections)
- ketoconazole (except shampoo), itraconazole, voriconazole, fluconazole, miconazole (to treat fungal infections)
- amiodarone (to treat irregular heartbeat)
- ciclosporin (for immune systems)
- diltiazem, verapamil (for heart disease)
- piperine (a herbal preparation)
Some medicines may affect the way other medicines work. Your doctor or pharmacist will be able to tell you what to do when using OPSUMIT with other medicines.
How to take OPSUMIT
Always take this medicine exactly as your doctor has told you. Check with your doctor if you are not sure.
How much to take
The recommended dose of OPSUMIT for adults and children over 12 years of age and weighing more than 40 kg is one 10 mg tablet, once a day.
Swallow the whole tablet, with a glass of water. Do not chew or break the tablet.
You can take OPSUMIT with or without food.
How to take it
Use OPSUMIT for as long as your doctor advises you to. OPSUMIT is generally used over a prolonged period of time, possibly years. It should not be stopped suddenly.
If you forget to take it
If you forget to take OPSUMIT, take a dose as soon as you remember, then continue to take your tablets at the usual times. Do not take a double dose to make up for a forgotten tablet.
If you take too much (overdose)
Immediately telephone your doctor, or the Poisons Information Centre or go to accident and emergency at you nearest hospital if you think you or anyone else may have taken too much OPSUMIT. Do this even if there are no signs of discomfort or poisoning.
You may need urgent medical attention or advice on management of overdose.
Poison Information Centre telephone numbers:
- Australia: 13 11 26
- New Zealand: 0800 POISON or 0800 764 766
While you are using OPSUMIT
Things you must do
Tell your doctor or pharmacist that you are taking OPSUMIT if you are about to start on any new medicines.
Tell your doctor if you become pregnant or are trying to become pregnant.
Tell your doctor if, for any reason, you have not used your medicine exactly as prescribed.
Things you must not do
OPSUMIT is a treatment that you will need to keep on taking to control your PAH.
Do not stop taking OPSUMIT unless you have agreed this with your doctor.
If you have any further questions on the use of OPSUMIT, ask your doctor or pharmacist.
Do not give this medicine to anyone else, even if their symptoms seem similar to yours.
Do not use OPSUMIT to treat any other complaints unless your doctor says to.
Things to be careful of
As with many other medicines, OPSUMIT may cause headaches in some people.
Be careful driving or operating machinery until you know how OPSUMIT affects you. If you are affected, do not drive or operate machinery.
Side effects
Check with your doctor as soon as possible if you have any problems while taking OPSUMIT, even if you do not think the problems are connected with the medicine or are not listed in this leaflet.
Like all medicines, OPSUMIT can cause side effects in some people.
Tell your doctor at once if you experience any of the following while you are receiving OPSUMIT:
Very common side effects
- Anaemia (low number of red blood cells) or haemoglobin decreases
- Headache
- Bronchitis (inflammation of the airways)
- Nasopharyngitis (inflammation of the throat & nasal passages)
- Oedema and peripheral oedema/fluid retention (swelling especially in the ankles and feet)
- Upper respiratory tract infection
Common side effects
- Diarrhoea
- Abdominal pain
- Gastroenteritis (inflammation of stomach and gut)
- Irritable bowel syndrome (stomach pain and bloating with diarrhoea or constipation)
- Urinary tract infection (bladder infection)
- Fever
- Tonsillitis
- Pharyngitis (inflammation of the throat)
- Influenza (flu)
- Viral infection of nose, throat or chest
- Sinusitis (sinus infection)
- Lower respiratory tract infection
- Inflammation of respiratory tract
- Itchy, runny or blocked nose
- Increased sensitivity of the airways in the lungs, causing narrowing and difficulty breathing
- Infection of the windpipe
- Hypotension (low blood pressure)
- Pruritus (severe itching of the skin)
- Eczema (itchy skin rash)
- Thrombocytopenia (low number of platelets (cells that help blood to clot))
- Haematocrit decreased (decreased proportion of red blood cells in the blood)
- Hypokalaemia (low level of potassium in the blood)
- Hyperkalaemia (high level of potassium in the blood)
- Increased level of urea in the blood
- Difficulty sleeping
- Ulcer or sore on the skin
- Conjunctivitis (inflammation of the eye)
- Haemorrhoids
- Gallstones
- Problem with the immune system that causes a build up of scar tissue in the skin and internal organs (systemic scleroderma)
- Inflamed tissue (cartilage) in the rib cage
- Bleeding between monthly menstrual periods
- Long or heavy menstrual periods
- Cyst in the ovary
- Enlarged breasts in men
- Flushing
- Joint pain
- Muscle pain
- Depression
- Migraine
If you get any side effects, talk to your doctor or pharmacist. This includes any possible side effects not listed in this leaflet.
Do not be alarmed by this list of possible side effects. You may not experience any of them.
This is not a complete list of all possible side-effects. Others may occur in some people and there may be some side-effects not yet known.
Tell your doctor if you notice anything else that is making you feel unwell, even if it is not on this list.
Ask your doctor if you don't understand anything in this list.
Storing OPSUMIT
Storage
Keep this medicine where young children cannot reach it.
A locked cupboard at least one-and-a half metres above the ground is a good place to store medicines.
Do not leave them in the car or on window sills.
Do not use this medicine after the expiry date which is stated on the carton after EXP. The expiry date refers to the last day of that month.
Do not store above 30°C.
Disposal
Medicines should not be disposed of in wastewater or household waste.
Ask your pharmacist how to dispose of medicines you no longer require. These measures will help to protect the environment.
Product description
What it looks like
OPSUMIT 10 mg tablets are white to off white, biconvex, round, film-coated tablets with "10" on both sides.
Pack size
OPSUMIT is supplied as 10 mg film-coated tablets in blister packs of, 9 or 30 tablets.
Ingredients
Active ingredient: Each tablet of OPSUMIT contains 10 mg of macitentan.
Inactive ingredients:
- lactose monohydrate
- microcrystalline cellulose
- povidone
- sodium starch glycollate type A
- magnesium stearate
- polysorbate 80
- polyvinyl alcohol
- titanium dioxide
- talc
- soya lecithin
- xanthan gum.
Sponsor
JANSSEN-CILAG Pty Ltd
1-5 Khartoum Road
Macquarie Park NSW 2113 Australia
Telephone: 1800 226 334
NZ Office: Auckland New Zealand
Telephone: 0800 800 806
This leaflet was prepared on 17 October 2022.
OPSUMIT 10 mg tablets packs:
- blister packs of 9 tablets AUST R 205624
- blister packs of 30 tablets AUST R 205624
Published by MIMS December 2022






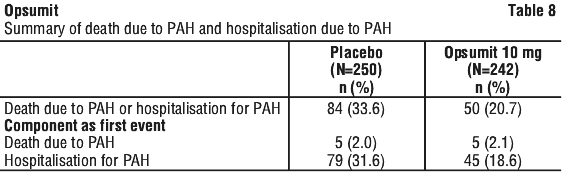
 In SERAPHIN, a double-blind study in patients with PAH, discontinuations for hepatic AEs were 3.3% in the Opsumit 10 mg group vs. 1.6% for placebo.
In SERAPHIN, a double-blind study in patients with PAH, discontinuations for hepatic AEs were 3.3% in the Opsumit 10 mg group vs. 1.6% for placebo.

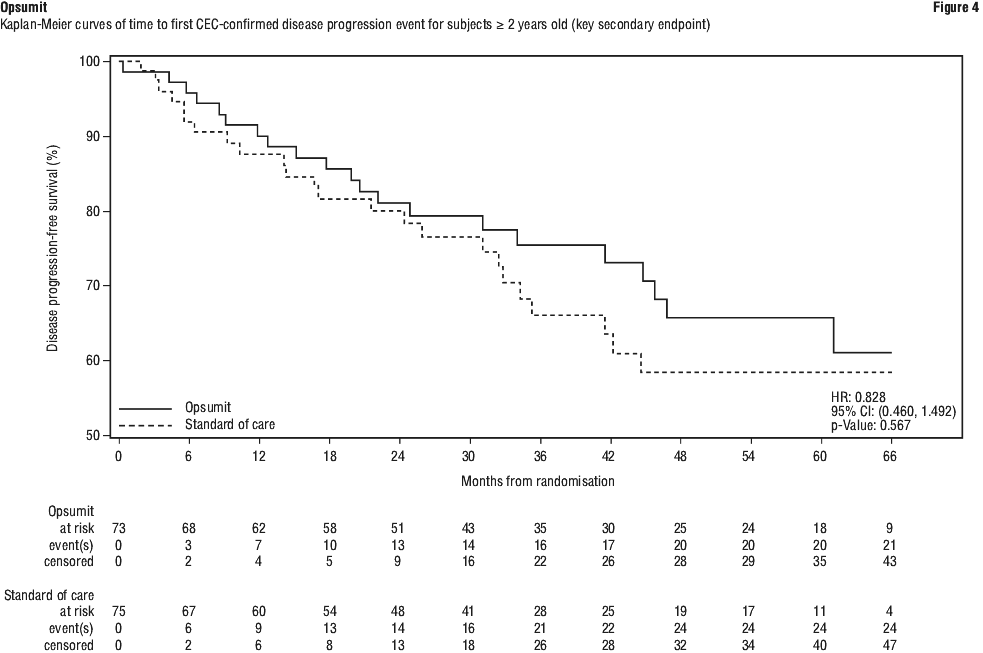
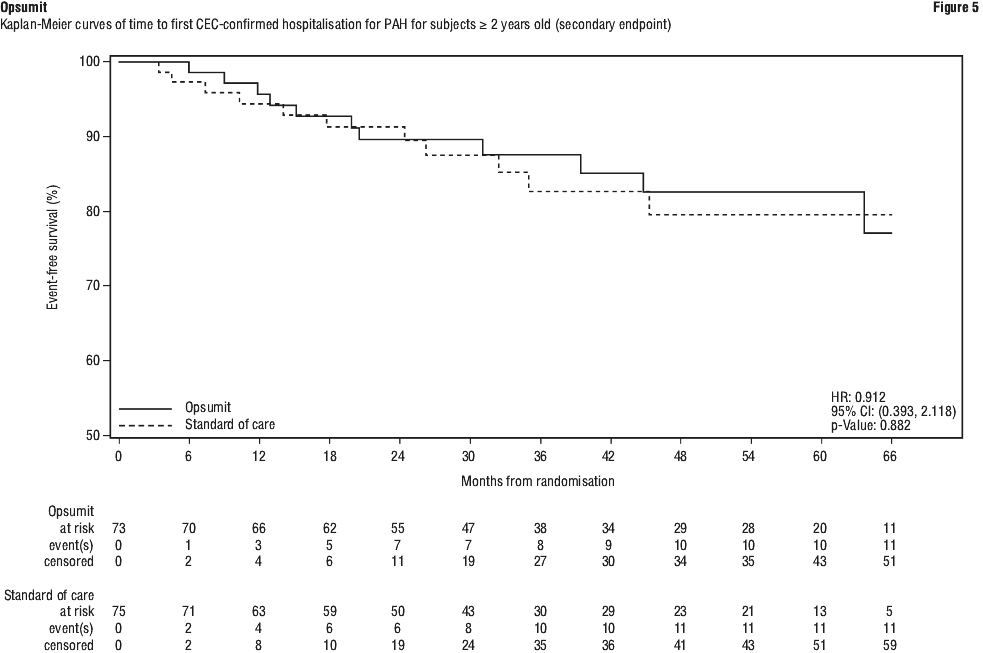
 Subgroup analyses were performed to examine their influence on outcome as shown in Figure 2. Consistent efficacy of Opsumit 10 mg on the primary endpoint was seen across subgroups of age, sex, race, aetiology, by monotherapy or in combination with another PAH therapy, baseline 6MWD, and baseline WHO FC.
Subgroup analyses were performed to examine their influence on outcome as shown in Figure 2. Consistent efficacy of Opsumit 10 mg on the primary endpoint was seen across subgroups of age, sex, race, aetiology, by monotherapy or in combination with another PAH therapy, baseline 6MWD, and baseline WHO FC. The SERAPHIN study was not powered to assess the effect on mortality. Treatment with Opsumit 10 mg resulted in a statistically non-significant 36% relative risk reduction (HR 0.64, 97.5% CI: 0.29-1.42; logrank p = 0.2037) in the occurrence of death of all causes up to EOT regardless of prior worsening. The number of deaths of all causes up to EOS on macitentan 10 mg was 35 versus 44 on placebo (HR 0.77; 97.5% CI: 0.46 to 1.28; logrank p = 0.2509).
The SERAPHIN study was not powered to assess the effect on mortality. Treatment with Opsumit 10 mg resulted in a statistically non-significant 36% relative risk reduction (HR 0.64, 97.5% CI: 0.29-1.42; logrank p = 0.2037) in the occurrence of death of all causes up to EOT regardless of prior worsening. The number of deaths of all causes up to EOS on macitentan 10 mg was 35 versus 44 on placebo (HR 0.77; 97.5% CI: 0.46 to 1.28; logrank p = 0.2509).