What is in this leaflet
This leaflet answers some common questions about OPTIRAY. It does not contain all the available information. It does not take the place of talking to your doctor.
All medicines have risks and benefits. Your doctor has weighed the risks of using OPTIRAY against the benefits it is expected to have for you.
If you have any concerns about receiving OPTIRAY, ask your doctor.
Keep this leaflet. You may need to read it again.
1. What is OPTIRAY and what it is used for
OPTIRAY is an injectable contrast medium containing iodine. The active ingredient is ioversol. It is used to make clearer diagnostic images of the brain and body in adults and children. The iodine blocks the X-rays, allowing vessels and the inner organs supplied with blood to be seen. As a result, it helps to clearly show abnormalities in the brain or body. OPTIRAY is for diagnostic examinations only.
2. Before you are given OPTIRAY
When you must not use it
Do not use OPTIRAY if you:
- you are allergic (hypersensitive) to the active ingredient ioversol, or to any of the other ingredients in OPTIRAY
- you have an overactive thyroid gland or high levels of thyroid hormones you are unable to pass urine or pass very little urine.
Before you start to use it
Tell your doctor if you:
- suffer from asthma or allergies (e.g. medicinal products, seafood, hay fever, hives)
- had previous reactions to injections of a contrast agent, including a previous history of reaction to iodine-based agents
- have kidney or liver disease
- have diabetes
- have heart failure, high blood pressure, circulation disorders, or had a stroke, and if you are elderly
- have brain disorders
- have problems with bone marrow, such as blood cancers known as paraproteinaemia or multiple myeloma
- have a red blood cell abnormality, known as sickle cell anaemia
- have a tumour of the adrenal gland which affects your blood pressure, known as phaeochromocytoma
- have a child who is under the age or 12 months and OPTIRAY is planned for the examination.
- are pregnant, intend to become pregnant. If you receive this medicine whilst pregnant, your newborn should be tested to ensure they are producing the correct amount of thyroid hormone.
- are breast-feeding or plan to breast-feed
- are feeling thirst and/or you have only had small quantities or nothing to drink before the examination
- have a cardiac pacemaker or any ferromagnetic implant (vascular clips, etc.) or a metallic stent in your body
- are taking a antihypertensive medicine, known as a beta-blocker
- you are or your child is on a controlled sodium diet
If any of these apply to you, your doctor will decide whether the intended examination is suitable for you.
Using other medicines
Please tell your doctor if you are taking or have recently taken any other medicines including medicines obtained without a prescription from your pharmacy, supermarket or health food shop.
Tell your doctor if you are taking any of the following medicines:
- metformin, used to treat diabetes
- interleukins, used to treat certain tumours
- vasopressors, used to increase blood pressure
- diuretics, used to help eliminate salt and water from your body.
Pregnancy and breast feeding
It is not known whether OPTIRAY is excreted in human milk. Tell your doctor if you are pregnant and ask your doctor for advice before you are given OPTIRAY.
Driving and using machines
Driving or operating machines is not advisable for up to 1 hour after injection. Symptoms such as dizziness, drowsiness, fatigue and visceral disturbances have been reported. If this affects you, do not attempt any activities which require concentration and the ability to react appropriately.
Children should be careful when riding bicycles or climbing trees.
3. How OPTIRAY is given
Diagnostic examinations involving the use of contrast agents should be conducted under the supervision of a doctor with the prerequisite training and a thorough knowledge of the examination to be performed.
Dosage
Your doctor will decide how much you will be given. The dose depends on your condition, the specific examination you are having and other factors, such as your health and age. The lowest possible dose will be given to produce adequate X-ray images.
You should be well hydrated prior to and following the administration of OPTIRAY.
If you are given more OPTIRAY than you should have been (overdose)
Overdosages are potentially life-threatening and may affect breathing, heart, and circulation system. Inform your doctor immediately if you notice any of these symptoms after receiving OPTIRAY.
4. Possible side effects
Like all medicines, this medicine can cause side effects, but not everybody will get them. Side effects with OPTIRAY in most cases are mild or moderate, they are very rarely serious or life-threatening.
Tell your doctor immediately if you develop any of the following signs of serious side effects:
- heart or breathing irregular
- heart vessel spasms or blood clots
- stroke, blue lips, fainting
- loss of memory
- speech disorders
- sudden movements
- temporary blindness
- acute kidney failure
- skin rash, redness or blisters which may develop into life-threatening skin reactions including extensive peeling of the skin (toxic epidermal necrolysis), or a drug reaction which causes rash, fever, inflammation of internal organs, haemtological abnormalities and systemic illness (DRESS)
- signs of allergic reactions, such as allergic shock, tightened airways, swelling of the throat, tongue, beathing difficulties, cough, sneezing, reddening and/or swelling of the face and eyes, itching, rash and hives.
Immediately tell the doctor or other healthcare professional who is giving you the injection, if you feel unwell, especially if you feel any tightness, pain or discomfort in your chest, face or throat, or you have difficulty breathing.
If you feel lightheaded, dizzy or faint when getting out of bed or standing up, get up slowly.
Standing up slowly, especially when you get up from bed or chairs, will help your body get used to the change in position and blood pressure. If this problem continues or gets worse, talk to your doctor.
Side-effects in Children
Some paediatric patients have a higher risk of adverse reactions to contrast media. Such patients may include those with sensitivity to allergens, including other medicines, those with asthma, congestive heart failure, a serum creatinine 1.5 mg/dL, or ages under 12 months.
If you have any further questions or concerns on the use of this medicine, ask your doctor.
5. How to store OPTIRAY
As this is being given to you by your doctor, it is extremely unlikely that you will be expected to look after the injection. However, in the case that you may have to transport it from the pharmacy to your doctor, keep the container in the outer carton to protect from light. Do not store below 25°C.
Do not use this medicine if the packaging is torn or shows signs of tampering or after the expiry date which is stated on the label.
Do not use the solution if it is discoloured or particulate material is present. This medicine should only be used once. Discard unused contents.
Keep all medicines in a safe place where children cannot reach them.
6. Product Description
What it looks like and packs
OPTIRAY is a clear, colourless to pale yellow solution, available in glass vials or plastic pre-filled syringes.
OPTIRAY contains the active ingredient, ioversol, 509 to 791 mg/mL, depending on the concertation being administered. It also contains sodium calcium edetate, trometamol, trometamol hydrochloride, sodium hydroxide, and water for injections.
This medicine does not contain lactose, sucrose, gluten, tartrazine or any other azo dyes.
The following OPTIRAY packs are registered
The Australian Registration number is AUST R 34371.
OPTIRAY 240
- 30 mL (AUST R 15326) and 50 mL (AUST R 49410) injection vials
- 50 mL (AUST R 46639) and 125 mL (AUST R 46640) Ultraject® injection syringes
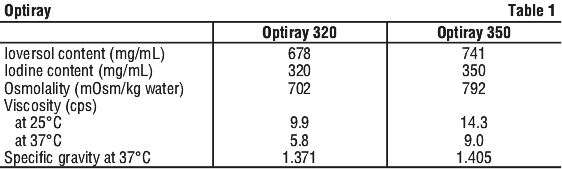
OPTIRAY 320
- 20 mL (AUST R 20034), 30 mL (AUST R 49421), 50 mL (AUST R 49422), 100 mL (AUST R 49423), 150 mL (AUST R49424) and 200mL (AUST R 49425) injection vials
- 50 mL (AUST R 46641), 75 mL (AUST R 73580) and 125 mL (AUST R 46642) Ultraject® injection syringes
OPTIRAY 350
- 20 mL (AUST R 49610), 30 mL (AUST R 47856), 50 mL (AUST R 47996),75 mL (AUST R 47997), 100 mL (AUST R 47998), 150 mL (AUST R 49611),200 mL (AUST R 49612) and 500 mL (AUST R 320145) injection vials
- 30 mL (AUST R51795), 50 mL (AUST R 51796), 75 mL (AUST R 70059), 100 mL (AUST R 61983) and 125 mL (AUST R 61984) Ultraject® injection syringes.
OPTIRAY vials are available in packs of 10 or 25 units.
OPTIRAY 350 in 500 mL vials are available in packs of 5, 6 or 10 units.
OPTIRAY prefilled syringes are available in packs of 10 or 20 units.
Note: Not all strengths and pack sizes may be available.
Sponsor
Guerbet Australia Pty Ltd
166 Epping Road, Level 2
Lane Cove, NSW, 2066
Australia
Telephone: 1800 859 436
Customer Service Email: [email protected]
This leaflet was revised in February 2023.
Published by MIMS May 2023

 For the full list of excipients, see Section 6.1 List of Excipients.
For the full list of excipients, see Section 6.1 List of Excipients.

 Chemical formula: C18H24I3N3O9.
Chemical formula: C18H24I3N3O9.