What is in this leaflet
This leaflet answers some common questions about the OXYTROL® transdermal drug delivery system (patch). It does not contain all the available information. It does not take the place of talking to your doctor or pharmacist.
All medicines have risks and benefits. Your doctor has weighed the risks of you being given OXYTROL® against the benefits they expect it will have for you.
If you have any concerns about being given this medicine, ask your doctor or pharmacist.
Keep this leaflet. You may need to read it again.
What OXYTROL® is used for
OXYTROL® is a transdermal patch that is a treatment for overactive bladder. It delivers the active ingredient, oxybutynin, through your skin and into your bloodstream.
Overactive bladder makes it hard to control when you urinate (pass water). Overactive bladder can make you urinate more often (increased frequency) or make you feel the need to urinate often (urgency). Overactive bladder can also lead to accidental urine loss (leaking or wetting oneself).
The active ingredient in OXYTROL®, oxybutynin, is dissolved in the thin layer of adhesive that sticks the patch to your skin. OXYTROL® delivers the medicine slowly and constantly through your skin and into your bloodstream for the three or four days that you wear the patch.
Ask your doctor if you have any questions about why this medicine has been prescribed for you. Your doctor may have prescribed it for another reason.
This medicine is not addictive.
This medicine is only available with a doctor's prescription.
Care should be taken when driving or operating machinery as this medicine may cause sleepiness or blurred vision.
There is not enough information to recommend the use of this medicine for children under the age of 18 years.
Before you are given OXYTROL®
When you must not be given it
Do not use OXYTROL® if you have an allergy to:
- any medicine containing oxybutynin
- any of the ingredients listed at the end of this leaflet.
Some of the symptoms of an allergic reaction may include shortness of breath, wheezing or difficulty breathing; swelling of the face, lips, tongue or other parts of the body; rash, itching or hives on the skin.
This medicine must not be used by a child under the age of 18 years. Safety and effectiveness in children younger than 18 years have not been established.
You must not use this medicine if you have (or have had) any of the following medical conditions:
- Urinary retention (your bladder does not empty or does not empty completely when you urinate)
- Gastric retention (Your stomach empties slowly or incompletely after a meal)
- Toxic megacolon (a very inflated large intestine)
- Uncontrolled narrow-angle glaucoma (high pressure in your eye)
- Severe ulcerative colitis (inflamed bowel)
- Myasthenia gravis (nerve weakness).
You must not use this medicine after the expiry date printed on the pack or if the packaging is torn or shows signs of tampering. If it has expired or is damaged, return it to your pharmacist for disposal.
If you are not sure whether you should be given this medicine, talk to your doctor.
Before you are given it
Tell your doctor if you have (or have had) any of the following medical conditions:
- Liver disease
- Kidney disease
- Diarrhoea (especially in patients with ileostomy or colostomy)
- Bladder obstruction (blockage)
- Gastrointestinal obstruction (blockage in the digestive system)
- Non-severe ulcerative colitis (inflamed bowel)
- Prostatic hypertrophy (enlargement of the prostate gland)
- Gastric reflux disease or oesophagitis (inflamed oesophageus, the tube between your mouth and stomach)
- Hyperthyroidism,
- Cardiovascular disease (including coronary heart disease, congestive heart failure, cardiac arrhythmias, tachycardia and hypertension )
- A neurological disorder (including autonomic neuropathy, cognitive impairment or Parkinson's disease)
Tell your doctor if you are pregnant or plan to become pregnant or are breast-feeding. Your doctor can discuss with you the risks and benefits involved.
Tell your doctor if you have allergies to any other medicines, foods, preservatives or dyes.
If you have not told your doctor about any of the above, tell him/her before using OXYTROL®.
Taking other medicines
Tell your doctor or pharmacist if you are taking any other medicines, including any that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines and OXYTROL® may interfere with each other.
These include:
- Other anticholinergic drugs (used to treat incontinence)
- Bisphosphonates (used to strengthen bone)
These medicines may be affected by OXYTROL® or may affect how well it works. You may need different amounts of your medicines, or you may need to take different medicines.
Your doctor and pharmacist have more information on medicines to be careful with or avoid while being treated with this medicine.
How to use OXYTROL®
Follow all directions given to you by your doctor or pharmacist carefully. They may differ from the information contained in this leaflet.
If you do not understand the instructions on the label, ask your doctor or pharmacist for help.
How much to apply
One new OXYTROL® patch should be applied two times a week (every three to four days) according to your doctor's instructions. Wear only one patch of OXYTROL® at a time.
When to apply it
Try to change the patch on the same two days each week. Your box of OXYTROL® has a calendar checklist printed inside to help you remember your schedule. Mark the schedule you plan to follow. Always change OXYTROL® on the two days of the week you mark on your calendar.
Where to apply it
Put the OXYTROL® patch on a clean, dry and smooth (fold-free) area of skin on your abdomen (stomach area), hips or buttocks as shown in the picture on the package instructions.
Avoid your waist line area, since tight clothing may rub against the patch. The areas you choose should not be oily, damaged (cut or scraped), irritated (rashes) or have any other skin problems.
Do not put OXYTROL® on areas that have been treated with oils, lotions, or powders that could keep the patch from sticking well to your skin.
Do not expose the patch to sunlight. Therefore, wear it under clothing.
When you put on a new patch, use a different area of skin from the most recent patch site. Do not use the same area for the patch for at least one week. You may wish to try different locations when using OXYTROL® to find the locations that are most comfortable for you and where clothing will not rub against it.
How to apply it
Each patch is sealed in its own protective pouch. When you are ready to put on the OXYTROL® patch, tear open the pouch and remove the patch. Apply the patch to your skin right away. Do not keep or store the patch outside the sealed pouch.
The sticky adhesive side of the patch is covered by two strips of overlapping protective liner. Remove the first piece of the protective liner and place the patch, adhesive face down, firmly onto the skin.
Bend the patch in half and gently roll the remaining part onto your skin using the tip of your fingers. As you roll the patch in place, the second piece of the protective liner should move off the patch. Apply firm pressure over the surface of the patch with your fingers to make sure the patch stays on. When putting on the patch, avoid touching the sticky adhesive side. Touching the adhesive may cause the patch to fall off early. Throw away the protective liners.
Contact with water when you are bathing, swimming, showering, or exercising will not change the way OXYTROL® works. However try to avoid rubbing the patch during these activities.
If the patch partly or completely falls off, press it back in place and continue to follow your application schedule. If the patch does not stay on, throw it away. You should then put on a new patch in a different area, but continue to follow your original application schedule.
How to remove it
When changing OXYTROL®, remove the old patch slowly and carefully, to avoid damaging the skin. Once off, fold the patch in half with the sticky sides together.
Since the patch will contain some oxybutynin, throw it away so that it cannot be accidentally worn or swallowed by another person, especially a child, or a pet.
Gently washing the application site with warm water and mild soap should remove any adhesive that stays on your skin after removing the patch. A small amount of baby oil may also be used to remove any excess residue. If the adhesive becomes dirty you may require a medical adhesive removal pad that you can get from your pharmacist. Alcohol or other dissolving liquids (nail polish remover or other solvents) may cause skin irritation and should not be used.
If you miss a dose
If you forget to change your patch after three or four days, remove the old patch, put on a new patch in a different area and continue to follow your original application schedule.
If you receive too much (overdose)
If you think that you or anyone else may have used too much OXYTROL®, remove the patch and immediately telephone your doctor or the Poisons Information Centre for advice (In Australia, call 13 11 26. In New Zealand, call 0800 764 766), or go to Accident and Emergency at your nearest hospital. Do this even if there are no signs of discomfort or poisoning.
Overdosage with oxybutynin has been associated with anticholinergic effects including:
- Excitation of the nervous system
- Flushing and fever
- Dehydration
- Irregular heart beat (Cardiac arrhythmia)
- Vomiting
- Difficulty passing Urine (urinary retention)
As OXYTROL® is given to you as a single patch, it is very unlikely that you will receive an overdose. However, if you experience severe side effects, tell your doctor or nurse immediately.
While you are being given OXYTROL®
Things you must do
If you are about to be started on any new medicine, remind your doctor and pharmacist you are using OXYTROL®.
Tell any other doctors, dentists, and pharmacists who treat you that you are using this medicine.
If you become pregnant while you are being treated with this medicine, tell your doctor immediately.
If you are about to have any blood tests, tell your doctor that you are being given this medicine. It may interfere with the results of some tests.
Keep all of your doctor's appointments so that your progress can be checked. Your doctor may do some tests from time to time to make sure the medicine is working and to prevent unwanted side effects.
Things to be careful of
Be careful driving or operating machinery until you know how OXYTROL® affects you. This medicine may cause tiredness and drowsiness in some people. If you have any of these symptoms, do not drive, operate machinery or do anything else that could be dangerous.
Drinking alcohol may increase sleepiness.
If you feel light-headed, dizzy or faint when getting out of bed or standing up, get up slowly. Standing up slowly, especially when you get up from bed or chairs, will help your body get used to the change in position and blood pressure. If this problem continues or gets worse, talk to your doctor.
Oxybutynin treatment may decrease sweating.
You may overheat or have fever or heat stroke if you are in warm or hot temperatures.
Side effects
Tell your doctor or nurse as soon as possible if you do not feel well while you are using OXYTROL®.
This medicine helps most people with overactive bladder, but it may have unwanted side effects in a few people. All medicines can have side effects. Sometimes they are serious, most of the time they are not. You may need medical attention if you get some of the side effects.
If you are over 65 years of age you may have an increased chance of getting side effects.
Do not be alarmed by the following lists of side effects. You may not experience any of them.
Ask your doctor or pharmacist to answer any questions you may have.
Tell your doctor, nurse or pharmacist if you notice any of the following and they worry you:
- Application site reactions (skin irritation including rash or slight redness and itching of the skin where the patch has been. This redness should disappear within several hours after removing the patch)
- Constipation
- Diarrhoea
- Dysuria (difficulty urinating)
- Dry Mouth
- Nausea
- Reduced sweating
- Flatulence
- Fatigue or tiredness
- Sleepiness or Drowsiness
- Headache
- Abdominal pain
- Back pain
- Blurred vision
These side effects are usually mild and short lived. The most common side effects of OXYTROL® are skin reactions where the patch is put on. These reactions include redness and itching. If uncomfortable irritation or excessive itchiness continues, tell your pharmacist or doctor.
If you take other medicines that cause dry mouth, constipation, or sleepiness, OXYTROL® can increase these effects.
If the following happens, tell your doctor or nurse immediately (or go to Accident and Emergency at your nearest hospital):
If you notice any symptoms of an allergic reaction such as shortness of breath, wheezing or troubled breathing; swelling of the face, lips, tongue or other parts of the body; rash, itching or hives on the skin.
You may need urgent medical attention (or hospitalisation).
Tell your doctor, nurse or pharmacist if you notice anything that is making you feel unwell.
Other side effects not listed above may also occur in some people. For a complete list of side effects, please ask your doctor or pharmacist.
After using OXYTROL®
Storage
Keep the patches in the original pack until it is time to use them. If you take the patches out of the pouch they will not keep well.
Keep the pack in a cool dry place where the temperature stays below 25 degrees Celsius.
Do not store OXYTROL® in the refrigerator or the freezer.
Do not store OXYTROL® or any other medicine in the bathroom or near a sink. Do not leave it on a window sill or in the car. Heat and dampness can destroy some medicines.
Keep it where children cannot reach it. A locked cupboard at least one-and-a-half metres above the ground is a good place to store medicines.
Disposal
If your doctor tells you to stop using the medicine or the expiry date has passed, ask your pharmacist what to do with any patches that are left over.
Product description
What it looks like
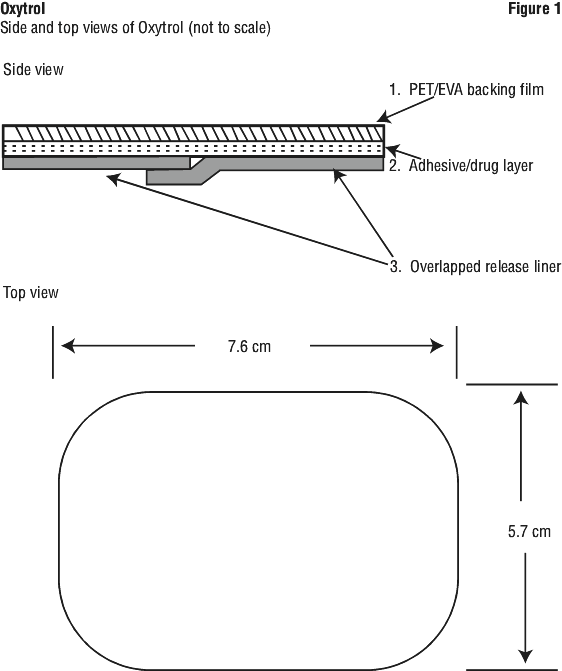
Each OXYTROL® patch is rectangular shape, slightly opaque, and is 39 cm2. Printed on each patch are the words "OXYTROL® 3.9 mg/day".
The patch is composed of three layers. Layer 1 is a backing film which protects the adhesive/drug layer. Layer 2 is the adhesive/drug layer. Layer 3 is a release liner made up of 2 polyester strips that should be peeled off and discarded prior to applying the patch.
Each box of OXYTROL® contains 8 patches (one month's treatment).
Ingredients
Each OXYTROL® transdermal patch contains 36 mg of oxybutynin as the active ingredient and delivers approximately 3.9 mg of oxybutynin per day.
The other ingredients are:
- glycerol triacetate,
- acrylic adhesive solution (containing 2-ethylhexyl acrylate, N-vinyl pyrrolidone and hexamethyleneglycol dimethacrylate polymer domains),
- clear backing film of polyester/ethylene vinyl acetate (PET/EVA), and
- siliconised polyester release liner (PET).
This medicine does not contain lactose, sucrose, gluten, tartrazine or any other azo dyes.
Supplier
Australia
Theramex Australia Pty Ltd
Level 34, 60 Margaret Street,
Sydney NSW 2000
1800 THERAMEX or 1800 843 726
This leaflet was revised in February 2019
- AUST R 125489
Published by MIMS July 2019



 Other adverse events reported by > 1% of Oxytrol-treated patients, and judged by the investigator to be possibly, probably or definitely related to treatment include: abdominal pain, nausea, flatulence, fatigue, somnolence, headache, flushing, rash, application site burning and back pain. Treatment-related adverse events with an incidence of < 1% were abdominal pain, dysuria, somnolence, nausea, back pain, urinary tract infections, inflected injury, rhinitis, palpitations and hot flushes.
Other adverse events reported by > 1% of Oxytrol-treated patients, and judged by the investigator to be possibly, probably or definitely related to treatment include: abdominal pain, nausea, flatulence, fatigue, somnolence, headache, flushing, rash, application site burning and back pain. Treatment-related adverse events with an incidence of < 1% were abdominal pain, dysuria, somnolence, nausea, back pain, urinary tract infections, inflected injury, rhinitis, palpitations and hot flushes. Study 2 was a randomised, double-blind, double-dummy, study of Oxytrol 3.9 mg/day versus active and placebo controls conducted in 361 patients. The 12-week double-blind treatment included an Oxytrol dose of 3.9 mg/day, an active comparator, and placebo. The study was to test superiority over placebo and equivalence with tolterodine. Equivalence to tolterodine was determined by calculating the 95% confidence interval (CI) for the difference in least squares adjusted means from the ANCOVA model for change in number of incontinence episodes per day, i.e. -1.5 to +1.5. The majority of patients were Caucasian (95%) and female (93%) with a mean age of 64 years (range, 18 to 89 years). Entry criteria required that all patients have urge or mixed incontinence (with a predominance of urge) and had achieved a beneficial response from the anticholinergic treatment they were using at the time of study entry. The average duration of prior pharmacological treatment was greater than 2 years. The patient's medical history and a urinary diary during the treatment-free baseline period confirmed the diagnosis of urge incontinence. Reductions in daily incontinence episodes, urinary frequency, and urinary void volume between placebo and active treatment groups are summarised in Table 4.
Study 2 was a randomised, double-blind, double-dummy, study of Oxytrol 3.9 mg/day versus active and placebo controls conducted in 361 patients. The 12-week double-blind treatment included an Oxytrol dose of 3.9 mg/day, an active comparator, and placebo. The study was to test superiority over placebo and equivalence with tolterodine. Equivalence to tolterodine was determined by calculating the 95% confidence interval (CI) for the difference in least squares adjusted means from the ANCOVA model for change in number of incontinence episodes per day, i.e. -1.5 to +1.5. The majority of patients were Caucasian (95%) and female (93%) with a mean age of 64 years (range, 18 to 89 years). Entry criteria required that all patients have urge or mixed incontinence (with a predominance of urge) and had achieved a beneficial response from the anticholinergic treatment they were using at the time of study entry. The average duration of prior pharmacological treatment was greater than 2 years. The patient's medical history and a urinary diary during the treatment-free baseline period confirmed the diagnosis of urge incontinence. Reductions in daily incontinence episodes, urinary frequency, and urinary void volume between placebo and active treatment groups are summarised in Table 4.
 Steady-state conditions are reached during the second Oxytrol application. Average steady-state plasma concentrations were 3.1 nanogram/mL for oxybutynin and 3.8 nanogram/mL for N-desethyloxybutynin (Figure 3). Table 5 provides a summary of pharmacokinetic parameters of oxybutynin in healthy volunteers after single and multiple applications of Oxytrol.
Steady-state conditions are reached during the second Oxytrol application. Average steady-state plasma concentrations were 3.1 nanogram/mL for oxybutynin and 3.8 nanogram/mL for N-desethyloxybutynin (Figure 3). Table 5 provides a summary of pharmacokinetic parameters of oxybutynin in healthy volunteers after single and multiple applications of Oxytrol.

 Following intravenous administration, the elimination half-life of oxybutynin is approximately 2 hours. Following removal of Oxytrol, plasma concentrations of oxybutynin and N-desethyloxybutynin decline with an apparent half-life of approximately 7 to 8 hours.
Following intravenous administration, the elimination half-life of oxybutynin is approximately 2 hours. Following removal of Oxytrol, plasma concentrations of oxybutynin and N-desethyloxybutynin decline with an apparent half-life of approximately 7 to 8 hours. The molecular weight of the compound is 357.49.
The molecular weight of the compound is 357.49.