What is in this leaflet
This leaflet answers some common questions about Paclitaxel Accord. It does not contain all the available information. It does not take the place of talking to your doctor.
All medicines have risks and benefits. Your doctor has weighed the risks of you taking this medicine against the benefits they expect it will have for you.
If you have any concerns about taking this medicine, ask your doctor or pharmacist.
Keep this leaflet. You may need to read it again.
What Paclitaxel Accord is used for
Paclitaxel Accord is used to treat cancer of the ovary, the breast, and non small cell cancer of the lung. Paclitaxel Accord may be used alone or in combination with other anticancer agents.
Paclitaxel Accord is not recommended for use in children as its safety and effectiveness in patients under 18 years of age has not been established.
Ask your doctor if you have any questions about why Paclitaxel Accord was prescribed for you.
How Paclitaxel Accord works
Paclitaxel Accord belongs to a class of anticancer agents known as taxanes. These agents prevent the division of cells, particularly cancer cells.
The use of Paclitaxel Accord to treat your cancer can lead to side-effects, which are discussed below.
Before Receiving Paclitaxel Accord
When Paclitaxel Accord must not be given
You have an allergy to:
- Any medicine containing paclitaxel or other medicines similar to paclitaxel called taxanes
- Any of the ingredients listed at the end of this leaflet
- Any medicines containing PEG-35 castor oil such as cyclosporin injection or teniposide injection.
Some of the symptoms of an allergic reaction may include:
- shortness of breath
- wheezing or difficulty breathing
- swelling of the face, lips, tongue or other parts of the body
- rash, itching or hives on the skin
You must not be given this medicine if you have a very low white blood cell count.
Tell your doctor if you have an infection or high temperature. Your doctor may decide to delay your treatment until the infection has gone. A mild illness, such as a cold, is not usually a reason to delay treatment.
You must not be given this medicine if you are pregnant or plan to become pregnant. Like most cytotoxic medicines paclitaxel is not recommended for use during pregnancy. If there is any need to consider this medicine during your pregnancy, your doctor will discuss with you the benefits and risks of using it.
Males: tell your doctor if your partner plans to become pregnant while you are being treated with this medicine or shortly after you have stopped treatment. Paclitaxel may cause birth defects if either the male or female is being treated with it at the time of conception. It is recommended that you use some kind of birth control while you are being treated with paclitaxel and for at least 12 weeks after you stop using it. Your doctor will discuss this with you.
Do not breast-feed if you are taking this medicine. Paclitaxel passes into breast milk and there is a possibility that your baby may be affected.
If you take this medicine after the expiry date has passed, it may not work as well (or it may make you feel sick).
Before you receive Paclitaxel Accord
Before you receive Paclitaxel Accord for the first time you should tell your doctor if:
- If you are taking any other medicines or treatment
- If you have ever been anaemic or suffered from other problems with your blood
- If you have had kidney or liver problems
- If you have received radiation therapy
- If you have high or low blood pressure
- If you have a heart problem
- Lowered immunity due to diseases such as HIV/AIDS
- Lowered immunity due to treatment with medicines such as cyclosporine, or other medicines used to treat cancer
- If you have ever suffered from neuropathy (numbness, tingling and pain in feet or hands)
- If you have had a previous serious reaction to a similar drug to paclitaxel (called taxanes).
Tell your doctor if you are pregnant or plan to become pregnant or are breast-feeding. Your doctor can discuss with you the risks and benefits involved.
If you have not told your doctor about any of the above, tell him/her before you start taking Paclitaxel Accord.
Taking other medicines
Tell your doctor or pharmacist if you are taking any other medicines, including any that you get without a prescription from your pharmacy, supermarket or health food shop.
Some medicines and paclitaxel may interfere with each other. These include:
- cisplatin and doxorubicin, medicines used to treat cancer
- erythromycin, trimethoprim and rifampicin, antibiotics used to treat some bacterial infections
- gemfibrozil, a medicine used to lower high cholesterol levels
- deferasirox, a medicine used to treat iron overload
- filgrastim, a medicine used for white blood cell disorders
- fluoxetine, a medicine used to treat depression
- carbamazepine, phenytoin and phenobarbital (phenobarbitone), medicines used for epilepsy
- efavirenz and nevirapine, medicines used to treat HIV (Human Immunodeficiency Virus) infection
- herbal medicines containing St John's wort.
These medicines may be affected by paclitaxel or may affect how well it works. You may need different amounts of your medicines, or you may need to take different medicines.
Your doctor and pharmacist have more information on medicines to be careful with or avoid while taking this medicine.
How Paclitaxel Accord is given
How much is given
Your doctor will decide what dose of Paclitaxel Accord you will receive. This depends on your condition and other factors, such as your weight, kidney function and other chemotherapy medicines you are being given.
Before you are given Paclitaxel Accord, you must take some other medicines to prevent allergic reactions occurring during your treatment.
You will need to take dexamethasone tablets 12 hours and 6 hours before your treatment, which your doctor will prescribe for you. You will also be given 2 different injections 30 to 60 minutes prior to receiving Paclitaxel Accord. This will minimize the risk of allergic reactions occurring.
Paclitaxel Accord may be given alone or in combination with other drugs.
Several courses of Paclitaxel Accord therapy may be needed depending on your response to treatment.
Additional treatment may not be repeated until your blood cell numbers return to acceptable levels and any uncontrolled effects have been controlled.
Ask your doctor if you want to know more about the dose of Paclitaxel Accord you receive.
How it is given
Paclitaxel Accord is usually given as an infusion (drip) into a vein over 3 hours.
How long it will be given for
Paclitaxel Accord is usually given once every three weeks. Each infusion is called one ‘cycle’ of chemotherapy. Your doctor will decide how many of these cycles you will need.
If you are given too much (overdose)
As Paclitaxel Accord is given to you under the supervision of your doctor, it is very unlikely that you will receive too much. However, if you experience severe side effects after being given this medicine, tell your doctor or nurse immediately.
You may need urgent medical attention.
Symptoms of a Paclitaxel Accord overdose include the side effects listed below in the ‘Side Effects’ section, but are usually of a more severe nature.
If you experience severe side effects tell your doctor immediately, telephone your doctor or the Poisons Information Centre (telephone 13 11 26) for advice or go to Accident and Emergency at the nearest hospital. You may need urgent medical attention.
Receiving Paclitaxel Accord
Things you must do
If you are about to be started on any new medicine, tell your doctor and pharmacist that you are receiving Paclitaxel Accord.
If you are going to have surgery, tell the surgeon or anaesthetist that you are being treated with this medicine. It may affect other medicines used during surgery.
Tell all of the doctors, dentists and pharmacists who are treating you that you are receiving treatment with Paclitaxel Accord.
You must tell your doctor immediately if you become pregnant while receiving Paclitaxel Accord treatment.
Keep follow-up appointments with your doctor. It is important to have your follow-up cycles of Paclitaxel Accord at the appropriate times to get the best effects from your treatments.
This medicine can lower the number of white blood cells and platelets in your blood. This means that you have an increased chance of getting an infection or bleeding.
The following precautions should be taken to reduce your risk of infection or bleeding:
- Avoid people who have infections. Check with your doctor immediately if you think you may be getting an infection, or if you get a fever, chills, cough, hoarse throat, lower back or side pain or find it painful or difficult to urinate.
- Be careful when using a toothbrush, toothpick or dental floss. Your doctor, dentist, nurse or pharmacist may recommend other ways to clean your teeth and gums. Check with your doctor before having any dental work.
- Be careful not to cut yourself when you are using sharp objects such as a razor or nail cutters.
- Avoid contact sports or other situations where you may bruise or get injured.
Your body breaks down Paclitaxel Accord and uses it to fight cancer. The breakdown products may be excreted in body fluids and waste, including blood, urine, faeces, vomitus and semen.
In general, precautions to protect other people should be taken while you are receiving chemotherapy and for one week after the treatment period by:
- Flushing the toilet twice to dispose of any body fluids and waste.
- Wearing gloves to clean any spill of body fluid or waste. Use paper towels or old rags, a strong solution of nonbleaching detergent and large amounts of water to mop up the spill. Discard the towels or rags into a separate waste bag and dispose of fluids in the toilet.
- Wash linen or clothing that is heavily contaminated by body fluids or waste separately from other items. Use a strong solution of non-bleaching detergent and large amounts of water.
- Place soiled disposable nappies and other pads in a plastic bag, seal and dispose into the garbage.
- For sexual intercourse, use a barrier method such as a condom.
Things to be careful of
Paclitaxel Accord contains absolute ethanol (alcohol). You should not drive, operate machinery, or do anything else that could be dangerous after receiving treatment with Paclitaxel Accord.
You should wear sun protection on your hands and feet after receiving treatment.
Side Effects
Tell your doctor as soon as possible if you do not feel well while you are receiving Paclitaxel Accord.
All medicines can have side effects. Sometimes they are serious, most of the time they are not. You may need medical attention if you get some of the side effects.
Common side effects
The most common side effects include:
- Infections, including of the lungs, upper respiratory tract, inner lining of the abdomen, blood and urinary tract
- flushing and rash
- itchiness
- bone marrow suppression (primarily neutropenia)
- thrombocytopenia (reduced numbers of the white blood cells that are responsible for blood clotting)
- leucopenia (low numbers of white blood cells)
- bleeding, blood clots
- fever
- anaemia (reduced numbers of red blood cells)
- hypotension (low blood pressure)
- disturbances of heart rate and rhythm
- heart problems which can cause shortness of breath or ankle swelling
- chest pain
- increased sweating
- nausea and vomiting
- diarrhoea
- mucositis (inflammation on the lining of the mouth or throat)
- arthralgia (pain in the joints) and myalgia (muscle pain)
- peripheral neuropathy (numbness, tingling and pain in feet and hands)
- abnormal liver blood tests
- alopecia (hair loss)
- seizures and convulsions
- headache
- visual disturbances
- hearing loss
- weight loss
- nail loss
- reactions at the injection site including localised swelling, pain, redness, hardening of the skin and skin discolouration.
Tell your doctor immediately, or go to accident and emergency at your nearest hospital if you notice any of the following:
- swelling to the face, lips, tongue or throat which may cause difficulty in swallowing or breathing
- severe and sudden onset of pinkish, itchy swellings
- gastrointestinal bleeding (blood in your stool)
- blistering of the skin, mouth, eyes and genitals.
Tell your doctor immediately if you notice any of the following:
- fever
- chest pain
- seizures (fits)
- disturbances of heart rate and rhythm
- numbness, tingling and pain in feet and hands
- unexpected bleeding
- abdominal pain.
The use of Paclitaxel Accord is also associated with a reduction in certain types of blood cells in the body:
- a reduction in red blood cells, which may result in fatigue or dizziness
- a reduction in white blood cells, which are used to fight infection
- a reduction in cells that help your blood to clot after injury.
These are serious side effects. You may need urgent medical attention or hospitalisation.
Other side effects not listed above may also occur in some patients. Tell your doctor if you notice anything else that is making you feel unwell.
Storage
Paclitaxel Accord should be stored below 25°C. Protected from light.
Product Description
What it looks like
Paclitaxel Accord is a clear to slightly yellow solution for injection.
Ingredients
Active Ingredient: paclitaxel
Inactive Ingredients: PEG-35 castor oil and absolute ethanol.
Name and Address of the Sponsor
Accord Healthcare Pty Ltd
Level 24, 570 Bourke Street
Melbourne, VIC, 3000
Australia
Australian Register numbers
30 mg/5 mL: AUST R 219554
100 mg/16.7 mL: AUST R 219555
300 mg/50 mL: AUST R 219556
Date of Preparation
This leaflet was prepared on 11 December 2020.
Published by MIMS February 2021



 The safety and efficacy of paclitaxel were studied in a randomised controlled multinational study of chemotherapy alone and in combination with Herceptin (trastuzumab). Patients with previously untreated metastatic breast cancer were treated with an anthracycline (doxorubicin 60 mg/m2 or epirubicin 75 mg/m2) plus cyclophosphamide (600 mg/m2) with (H+AC) or without (AC alone) Herceptin or paclitaxel (175 mg/m2 infused over three hours every three weeks) with (H+P) or without (P alone) Herceptin. Patients were treated with paclitaxel for six cycles, and could be treated with Herceptin until progression of disease. Patients who had previously received anthracycline based adjuvant therapy were treated with paclitaxel whereas those who were anthracycline naive were treated with an anthracycline plus cyclophosphamide. Patients in the Herceptin treatment groups received a 4 mg/kg intravenous loading dose of Herceptin on day 0. From day 7, patients received weekly infusions of Herceptin 2 mg/kg, which they could continue to receive until evidence of disease progression. Patients in both treatment groups were eligible to receive Herceptin in an open label study following disease progression.
The safety and efficacy of paclitaxel were studied in a randomised controlled multinational study of chemotherapy alone and in combination with Herceptin (trastuzumab). Patients with previously untreated metastatic breast cancer were treated with an anthracycline (doxorubicin 60 mg/m2 or epirubicin 75 mg/m2) plus cyclophosphamide (600 mg/m2) with (H+AC) or without (AC alone) Herceptin or paclitaxel (175 mg/m2 infused over three hours every three weeks) with (H+P) or without (P alone) Herceptin. Patients were treated with paclitaxel for six cycles, and could be treated with Herceptin until progression of disease. Patients who had previously received anthracycline based adjuvant therapy were treated with paclitaxel whereas those who were anthracycline naive were treated with an anthracycline plus cyclophosphamide. Patients in the Herceptin treatment groups received a 4 mg/kg intravenous loading dose of Herceptin on day 0. From day 7, patients received weekly infusions of Herceptin 2 mg/kg, which they could continue to receive until evidence of disease progression. Patients in both treatment groups were eligible to receive Herceptin in an open label study following disease progression.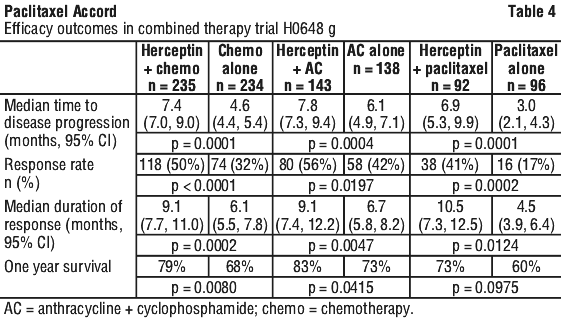 One year survival rates (the prospectively defined survival endpoint) were significantly better for chemotherapy + Herceptin versus chemotherapy arms (79 versus 68%; p=0.008). With a median follow-up of approximately two years, overall survival is improved for patients initially treated with chemotherapy and Herceptin compared with those receiving chemotherapy alone (25.4 versus 20.3 months; p=0.025) with a relative risk of death of 0.769 (95% CI 0.607 to 0.973; p=0.028).
One year survival rates (the prospectively defined survival endpoint) were significantly better for chemotherapy + Herceptin versus chemotherapy arms (79 versus 68%; p=0.008). With a median follow-up of approximately two years, overall survival is improved for patients initially treated with chemotherapy and Herceptin compared with those receiving chemotherapy alone (25.4 versus 20.3 months; p=0.025) with a relative risk of death of 0.769 (95% CI 0.607 to 0.973; p=0.028). Molecular Formula: C47H51NO14.
Molecular Formula: C47H51NO14.