What is in this leaflet
This leaflet answers some common questions about Panamax tablets, elixir and 240 elixir. It does not contain all the available information.
It does not take the place of talking to your pharmacist or doctor.
All medicines have benefits and risks. In deciding to give you Panamax, your pharmacist or doctor has weighed the risks of taking Panamax against the benefits it will have for you.
If you have concerns about taking this medicine, ask your pharmacist or doctor.
Keep this information with the medicine. You may need to read it again.
What is Panamax used for
Panamax is used to relieve pain and fever in adults (including arthritis, headache and muscular pain) and children (including teething, earache and immunisation).
Panamax works to stop the pain messages from getting through to the brain. Panamax also acts in the brain to reduce fever.
Ask your pharmacist or doctor if you have any questions about this medicine.
Your pharmacist or doctor may have prescribed this medicine for another use. If you want more information, ask your pharmacist or doctor.
Before you take it
When you must not take it
You should not take Panamax if you are allergic to paracetamol or any of the ingredients listed under "Product Description".
The symptoms of an allergic reaction may include:
- shortness of breath
- wheezing or difficulty breathing
- swelling of the face, lips, tongue or other parts of the body
- rash, itching or hives on the skin
Do not take this medicine if you have liver or kidney problems.
Do not take Panamax if you have glucose-6-phosphate-dehydrogenase deficiency (an enzyme deficiency).
Do not use Panamax after the expiry date (EXP) printed on the pack or bottle.
Do not use Panamax if the packaging is torn or shows signs of tampering. If it has expired or is damaged, return it to your pharmacist for disposal.
If you are not sure whether you should start taking this medicine, talk to your pharmacist or doctor.
Before you start to take it
You must tell your pharmacist or doctor if you have or have had any of the following medical conditions:
- You have allergies to any ingredients listed under "Product Description" at the end of this leaflet
- You have allergies to aspirin or any other NSAID medicine
- You have liver or kidney problems
- You have low glutathione reserves
- You have Gilbert's syndrome
- Acute breathing difficulties such as bronchitis, unstable asthma or emphysema
- You drink large quantities of alcohol
Tell your pharmacist or doctor about taking paracetamol if you are pregnant or plan to become pregnant, or breastfeeding. Panamax may be used during pregnancy and if you are breastfeeding. However, it is recommended that non-drug therapy such as rest and massage be tried first. If Panamax is needed during pregnancy, it should be used at the lowest effective dose for the shortest possible time and at the lowest possible frequency.
Taking other medicines
You should tell your pharmacist or doctor if you are taking any other medicines, including any that you get without a prescription from your pharmacy, supermarket or health food shop.
Some medicine and Panamax may interfere with each other. These include:
- Any medicines which thin the blood, for example warfarin
- Medicines to treat epilepsy
- Metoclopramide or domperidone, medicines used to control nausea and vomiting
- Propantheline, a drug used to treat stomach ulcers
- Other pain relief medication
- Alcohol and medicines containing alcohol (ethanol) eg some cough syrups
- Medicines used to treat depression
- Medicines used to aid sleep
- Chloramphenicol, an antibiotic used to treat ear and eye infections
- Flucloxacillin, zidovudine and rifampicin, drugs used to treat infections
- Probenecid, a drug used to treat high uric acid levels in the blood associated with gout
- Cholestyramine, a drug used to reduce blood cholesterol
- Chelating resin
These medicines may be affected by Panamax or may affect how well Panamax works.
Your pharmacist or doctor can tell you what to do if you are taking any of these medicines.
If you have not told your pharmacist or doctor about any of these things, tell him/her before you take Panamax.
How to take Panamax
The label on the pack or bottle will tell you how to take your medicine and how often. If you are unsure about the directions ask your pharmacist or doctor.
There are three different Panamax products:
- Panamax Tablets,
- Panamax Elixir and
- Panamax 240 Elixir.
The dose will be different depending upon which product you are taking.
Panamax Tablets
The usual dose is:
Adults:
1 to 2 tablets
This dosage may be repeated every 4 to 6 hours if necessary.
You should not take more than 8 tablets in 24 hours.
Children (7-12 years):
Half (1/2) to 1 tablet.
This dosage may be repeated every 4 to 6 hours if necessary.
Children should not take more than 4 tablets in 24 hours.
Panamax tablets are not recommended for children under 7 years.
Swallow tablets whole with a little water or other liquid.
Panamax Elixir
The usual dose is
Infants:
1 to 3 months: (4 - 6 kg)
2 to 4 mL;
3 to 6 months: (6 - 8 kg)
4 to 5 mL;
6 to 12 months: (8 - 10 kg)
5 to 6 mL.
Children:
1 to 2 years: (10 - 12 kg)
6 to 8 mL;
2 to 4 years: (12 - 16 kg)
8 to 10 mL;
4 to 6 years: (16 - 20 kg)
10 to 13 mL;
6 to 8 years: (20 - 25 kg)
13 to 16 mL;
8 to 10 years: (25 - 32 kg)
16 to 20 mL;
10 to 12 years: (32 - 41 kg)
20 to 26 mL.
This dosage may be repeated every 4 to 6 hours if necessary.
Do not give more than 4 doses in 24 hours.
Panamax Elixir is not recommended for infants under 1 month.
It can be given in water.
Panamax 240 Elixir
Children:
5 to 6 years: (18 - 20 kg)
6 mL;
6 to 8 years: (20 to 25 kg)
6 to 8 mL;
8 to 10 years: (25 - 32 kg)
8 to 10 mL;
10 to 12 years: (32 - 41 kg)
10 to 12 mL.
Do not give more than 4 doses in 24 hours.
Adults:
10 to 20 mL
This dosage may be repeated every 4 to 6 hours if necessary up to 4 times in 24 hours.
Do not take more than 80 mL per day.
Panamax 240 Elixir is not recommended for children under 5 years of age.
It can be given in water if necessary.
Your doctor may ask you to take a different dose. If so, you should follow the doctor's instructions.
If you are unsure what dose to take ask your pharmacist.
If you forget to take it
If you miss a dose, take it as soon as you remember. If it is almost time for your next dose, skip the dose you missed, and take your next dose when you are meant to.
Do not take a double dose of Panamax to make up for the dose missed.
If you take too much (Overdose)
Immediately telephone your doctor or Poisons Information Centre (telephone 13 11 26) for advice or go to Accident and Emergency at your nearest hospital, if you think that you or anyone else has taken too much Panamax.
Do this even if there are no signs of discomfort or poisoning. You may need urgent medical attention.
Keep telephone numbers of these places handy.
If you take too much Panamax you may feel nauseous, dizzy or sweaty.
While you are taking Panamax
Things you must do
Take Panamax exactly as directed.
Tell all your doctors, dentists and pharmacists that you are taking Panamax.
Things you must not do
Do not take more than the recommended dose unless your doctor tells you to.
Children:
Do not give Panamax for more than 48 hours unless a doctor has told you to
Adults:
Do not take for more than a few days at a time unless your doctor tells you to.
Things to be careful of
Only drink small quantities of alcohol (beer, wine or spirits) while taking paracetamol. Drinking large quantities of alcohol while taking paracetamol may increase the risk of liver side effects.
Side Effects
Tell your pharmacist or doctor as soon as possible if you do not feel well while taking Panamax.
Like other medicines Panamax can cause some side effects. If they occur, they are most likely minor and temporary. However, sometimes they are serious, and need medical treatment.
Ask your pharmacist or doctor any questions you may have.
Tell your pharmacist or doctor if you notice any of the following and they worry you:
- Nausea and vomiting
- Stomach pain
- Indigestion
- Sweating
Tell your doctor as soon as possible if you notice any of the following:
- Skin rashes
- Painful red areas with blisters and peeling layers of skin which may be accompanied by fever and/or chills
- Hepatitis (symptoms include loss of appetite, itching, yellowing of the skin and eyes, light coloured bowel motions, dark coloured urine)
If any of the following happen, tell your pharmacist or doctor immediately or go to Accident and Emergency at your nearest hospital:
- shortness of breath
- wheezing or difficulty breathing
- swelling of the face, lips, tongue or other parts of the body
- Severe blisters and bleeding in the lips, eyes, mouth, nose and genitals
- rash, itching or hives on the skin
The above list includes very serious side effects. You may need urgent medical attention or hospitalisation. These side effects are very rare.
Some people may get other side effects while taking Panamax.
Tell your pharmacist or doctor if you notice anything else that is making you feel unwell while taking Panamax.
After taking Panamax
Storage
Keep your tablets in the blister pack until it is time to take them. If you take the tablets out of the box or the blister pack they will not keep well.
Keep bottles tightly closed, and ensure that the child resistant cap is engaged after use.
Keep Panamax in a cool, dry place where the temperature stays below 30°C. Heat and dampness can destroy some medicines. Do not leave Panamax in the car on hot days.
Do not store Panamax or any other medicine in the bathroom or near a sink.
Keep Panamax where young children cannot reach it. A locked cupboard at least one and a half metres above the ground is a good place to store medicines.
Disposal
If your doctor tells you to stop taking Panamax, ask your pharmacist what to do with any medication which is left over.
This is not all the information available on Panamax. If you have any more questions or are not sure about anything, ask your doctor or pharmacist.
Product Description
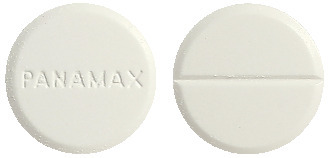
Panamax comes as a white tablet, marked PANAMAX.
A box may contain 50 or 100 tablets.
Panamax is also available as an Elixir (100 mL) or 240 Elixir (200 mL).
Each Panamax Tablet contains:
Active Ingredient:
- Paracetamol 500 mg
Other Ingredients:
- Maize starch
- Purified talc
- Pregelatinised starch
- Povidone
- Stearic acid
- Potassium sorbate
Panamax tablets do not contain gluten, lactose, sucrose, tartrazine or any azo dyes.
Panamax Elixir (100 mL) contains:
Active Ingredient:
- Paracetamol 120 mg in 5 mL
Other Ingredients:
- Macrogol 1500
- Glycerol
- Propylene Glycol
- Potassium Sorbate
- Benzoic Acid
- Brilliant scarlet 4R CI 16255
- Tutti Frutti Flavour 51880 A7
- Raspberry Flavour 54428 A7
- Lycasin
- Sodium Saccharin
- Water - Purified
Panamax 240 Elixir (200 mL) contains:
Active Ingredient:
- Paracetamol 240 mg in 5 mL
Other Ingredients:
- Macrogol 1500
- Propylene Glycol
- Potassium Sorbate
- Benzoic Acid
- Allura Red AC CI 16035
- Glycerol
- Raspberry Flavour 21820
- Sorbitol solution 70% crystallising
- Sodium Saccharin
- Water - Purified
- Imitation candied sugar 510155U
Sponsor
sanofi-aventis australia pty ltd
12-24 Talavera Road
Macquarie Park NSW 2113
AUST R 15490 (Tablets)
AUST R 13750 (Elixir)
AUST R 49623 (240 Elixir)
This leaflet was revised in October 2017
panamax-ccsiv1-cmiv11 11oct17
Published by MIMS December 2017

 MW: 151.17.
MW: 151.17.