1 Name of Medicine
Perindopril erbumine/ indapamide hemihydrate.
2 Qualitative and Quantitative Composition
Each tablet contains perindopril erbumine 4 mg and indapamide hemihydrate 1.25 mg, as the active ingredients.
Excipients with known effect.
Lactose.
For the full list of excipients, see Section 6.1 List of Excipients.3 Pharmaceutical Form
White, capsule-shaped, biconvex tablets, engraved "4/1.25" on one side and "APO" on the other side.
4.1 Therapeutic Indications
Treatment of hypertension. Treatment should not be initiated with this combination.
4.2 Dose and Method of Administration
Perindopril Indapamide-WGR 4/1.25 tablets are intended for oral administration.
Dosage.
One Perindopril Indapamide-WGR 4/1.25 tablet per day as a single dose, preferably to be taken in the morning.
Patients with renal impairment.
In cases of severe renal insufficiency (creatinine clearance below 30 mL/min), the treatment is contraindicated.
In patients with moderate renal insufficiency (creatinine clearance from 30 to 60 mL/min), the maximum dose should be at a low dose or with one component.
In patients with a creatinine clearance greater than 60 mL/min, no dose adaptation is required.
Normal medical practice includes periodic control for creatinine and potassium.
Patients with hepatic impairment.
In severe hepatic impairment, treatment is contraindicated.
In patients with moderate hepatic impairment, no dose modification is required.
Elderly patients.
Renal insufficiency is commonly observed in elderly people. Care should therefore be taken when prescribing perindopril-containing medicines to elderly hypertensive patients.
The initial daily dose in the elderly should always be at a low dose or with one component only, and patients should be monitored closely during the initial stages of treatment.
Particular care should be taken in elderly patients with congestive heart failure who have renal and/or hepatic insufficiency (see Section 4.4 Special Warnings and Precautions for Use).
Other "at risk" populations.
In patients with severe cardiac insufficiency (grade IV) or insulin dependent diabetes mellitus (spontaneous tendency to increased levels of potassium), treatment should be started with a reduced initial dose, and patients should be monitored closely during the initial stages of treatment.4.3 Contraindications
Related to a perindopril 4 mg/ indapamide 1.25 mg combination.
In patients with a history of previous hypersensitivity to either of the active ingredients, perindopril or indapamide, or excipient ingredients present in the tablets.
During pregnancy and for lactating women.
In patients with severe renal insufficiency (creatinine clearance below 30 mL/minute).
Due to the lack of sufficient therapeutic experience, perindopril/indapamide should not be used in: patients with severe untreated decompensated heart failure; patients on dialysis.
Related to perindopril component.
In patients with a history of previous hypersensitivity to the active ingredient perindopril, or to ACE inhibitors.
During pregnancy and for lactating women.
In patients with bilateral or unilateral renal artery stenosis (see Section 4.4 Special Warnings and Precautions for Use).
In patients with a history of hereditary and/or idiopathic angioedema or angioedema associated with previous ACE inhibitor treatment (see Section 4.4 Special Warnings and Precautions for Use).
In patients receiving extracorporeal treatments leading to contact of blood with negatively charged surfaces such as dialysis or haemofiltration with certain high-flux membranes (e.g. polyacrylonitrile membranes such as "AN69") and low density lipoprotein apheresis with dextran sulfate due to increased risk of severe anaphylactoid reactions following treatment with ACE inhibitors. This combination should therefore be avoided, either by use of alternative antihypertensive medicines or alternative membranes (e.g. cuprophane or polysulfone (PSF). (See Section 4.5 Interactions with Other Medicines and Other Forms of Interactions; Section 4.4 Special Warnings and Precautions for Use).
In combination with aliskiren-containing products in patients with diabetes or renal impairment (GFR < 60 mL/min/1.73 m2) (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions; Section 4.4 Special Warnings and Precautions for Use).
Combined use with sacubitril/valsartan fixed dose combinations - perindopril/ indapamide 4/1.25 must not be initiated earlier than 36 hours after the last dose of sacubitril/valsartan (see Section 4.4 Special Warnings and Precautions for Use; Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
Related to indapamide component.
In patients with a history of hypersensitivity to indapamide or any other sulfonamide derivatives.
In patients with severe renal impairment.
In patients with anuria.
In patients with progressive and severe oliguria.
In patients in a hepatic coma.
In patients with hepatic encephalopathy.
In patients with severe hepatic impairment.
In patients with hypokalaemia.
In combination with non-antiarrhythmic agents causing torsades de pointes.4.4 Special Warnings and Precautions for Use
Related to a perindopril 4 mg/ indapamide 1.25 mg combination.
Specific precautions relating to the use of the perindopril 4 mg/ indapamide 1.25 mg are the same as those which apply to the separate components of the combination. Consequently, caution should be observed when it is administered in patients with renal impairment and the risk of hypotension and electrolyte imbalance should be borne in mind (see Section 4.3 Contraindications; Section 4.2 Dose and Method of Administration; precautions specific to perindopril and indapamide under Special Warnings and Precautions for Use). Due to the possibility of an increased risk of idiosyncratic reactions following combined use of two antihypertensives, careful monitoring of patients is recommended.
Fluid and electrolyte imbalance.
Patients should be monitored for signs and symptoms of fluid or electrolyte imbalance; namely hyponatraemia, hypochloraemia, hyperuricaemia, hyperkalaemia (see Section 4.4 Special Warnings and Precautions for Use) and hypokalaemia (see Section 4.4 Special Warnings and Precautions for Use). Plasma urea and uric acid levels should also be monitored during treatment. Gout has been reported rarely. Tendency for gout attacks may be increased in patients with hyperuricaemia.
The clinical features of electrolyte imbalance include dryness of the mouth, thirst, weakness, lethargy, drowsiness, restlessness, muscle pains or cramps, muscle fatigue, hypotension, oliguria, gastrointestinal disturbances such as nausea and vomiting, tachycardia and ECG changes.
Potassium levels.
The combination of perindopril and indapamide does not prevent the onset of hypokalaemia particularly in patients with diabetes or in patients with renal failure. As with any antihypertensive agent in combination with a diuretic, regular monitoring of plasma potassium levels should be carried out.
Lactose intolerance.
Perindopril Indapamide-WGR 4/1.25 tablets contain lactose. Patients with an intolerance to lactose, rare hereditary problems of galactose intolerance, total lactase deficiency or glucose-galactose malabsorption should not take this medicine.
Hypotension and water and electrolyte depletion.
As there is a risk of sudden hypotension in the presence of pre-existing sodium depletion, particularly in those with renal artery stenosis, routine monitoring for clinical signs of water and electrolyte depletion which may occur concurrently with diarrhoea or vomiting is recommended. Regular monitoring of plasma electrolytes should be carried out in such patients.
Marked hypotension may require an intravenous infusion of isotonic saline.
Treatment with perindopril and indapamide 4/1.25 tablets should be stopped in patients with transient hypotension and possibly restarted, following re-establishment of a satisfactory blood volume and blood pressure, either at a low dose or with one component only.
Lithium.
The combination of lithium with perindopril or indapamide is not recommended. Diuretics should not be given with lithium because they reduce its renal clearance and add a high risk of lithium toxicity (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
Related to perindopril component.
Hyperkalaemia.
Diabetics and particularly the elderly may be at increased risk of hyperkalaemia. However, hyperkalaemia (> 5.5 mmol/L) is more likely in patients with some degree of renal impairment or those treated with potassium-sparing diuretics or with potassium supplements and/or consuming potassium containing salt substitutes. In some patients, hyponatraemia may coexist with hyperkalaemia.
Since ACE inhibitors reduce angiotensin II formation resulting in decreased production of aldosterone, increases in serum potassium have been observed in some patients treated with ACE inhibitors including perindopril. The effect is usually not significant in patients with normal renal function. Serum electrolytes (including sodium, potassium and urea) should be measured from time to time when ACE inhibitors are given, especially in combination with diuretics.
Hyperkalaemia can cause serious, sometimes fatal, arrhythmias. Risk factors for the development of hyperkalaemia include those with renal insufficiency, worsening of renal function, age (> 70 years), diabetes, intercurrent events, in particular dehydration, acute cardiac decompensation, metabolic acidosis and combined use with potassium-sparing diuretics (e.g. spironolactone, eplerenone, triamterene, or amiloride), potassium supplements or potassium-containing salt substitutes; or those patients taking other medicines associated with increases in serum potassium (e.g. heparin), other ACE-inhibitors, angiotensin receptor blocker, acetylsalicylic acid ≥ 3 g/day, COX-2 inhibitors and other non-selective NSAIDs, immunosuppressant agents such as ciclosporin or tacrolimus, co-trimoxazole (also known as trimethoprim/sulfamethoxazole). The use of potassium supplements, potassium-sparing diuretics, or potassium-containing salt substitutes particularly in patients with impaired renal function may lead to a significant increase in serum potassium. Potassium-sparing diuretics and angiotensin-receptor blockers should be used with caution in patients receiving ACE inhibitors, and serum potassium and renal function should be monitored.
Combined use of the above-mentioned medicines should be used with caution in combination with ACE inhibitors. Frequent monitoring of serum potassium (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions). In some patients hyponatraemia may co-exist with hyperkalaemia. It is recommended that serum electrolytes (including sodium, potassium and urea) should be measured from time to time when ACE inhibitors are given, especially when diuretics are also prescribed.
Patients with diabetes.
Glycaemic control should be closely monitored during the first month of treatment with an ACE inhibitor in patients with diabetes treated with oral medicines or insulin (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
Patients with insulin dependent diabetes mellitus (spontaneous tendency for hyperkalaemia) should be monitored closely during the initial stages of treatment. Treatment should be initiated with a reduced dose.
Potassium sparing medicines, potassium supplements or potassium-containing salt substitutes.
The combination of perindopril and potassium sparing medicines, potassium supplements or potassium-containing salt substitutes is not recommended (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
Angioedema.
ACE inhibitors should not be used in patients with a history of angioedema related to any other medicine as patients with a history of angioedema unrelated to ACE inhibitor treatment may be at increased risk of angioedema while receiving an ACE inhibitor (see Section 4.3 Contraindications).
Life-threatening angioedema has been reported with most ACE inhibitors. The overall incidence is approximately 0.1 to 0.2%. The aetiology is thought to be non-immunogenic and may be related to accentuated bradykinin activity. Usually the angioedema is non-pitting oedema of the skin, mucous membrane and subcutaneous tissue.
Angioedema of the face, extremities, lips, tongue, mucous membranes, glottis and/or larynx has been reported in patients with ACE inhibitors and has been reported uncommonly with perindopril (see Section 4.8 Adverse Effects (Undesirable Effects)). This may occur at any time during treatment. In such cases treatment should be promptly discontinued and the patient carefully observed until the swelling disappears.
Where such cases have been described with other ACE inhibitors and swelling has been confined to the face and lips, the condition has generally resolved without treatment although antihistamines have been useful in relieving symptoms. Angioedema associated with laryngeal oedema may be fatal or near fatal. In most cases symptoms occurred during the first week of treatment and the incidence appears to be similar in both sexes, or those with heart failure or hypertension.
Where there is involvement of the tongue, glottis or larynx likely to cause airway obstruction, appropriate treatment (e.g. adrenaline (epinephrine) and oxygen) should be given promptly. Treatment of progressive angioedema should be aggressive and failing a rapid response to medical treatment, mechanical methods to secure an airway should be undertaken before massive oedema complicates oral or nasal intubation.
Patients who respond to medical treatment should be observed carefully for a possible rebound phenomenon.
The onset of angioedema associated with use of ACE inhibitors may be delayed for weeks or months.
Patients may have multiple episodes of angioedema with long symptom-free intervals.
Angioedema may occur with or without urticaria.
Intestinal angioedema has been reported rarely in patients treated with ACE inhibitors. These patients presented with abdominal pain (with or without nausea or vomiting); in some cases there was no prior facial angioedema and C-1 esterase levels were normal. The angioedema was diagnosed by procedures including abdominal CT scan, ultrasound or during surgery and symptoms resolved after stopping the ACE inhibitor. Intestinal angioedema should be included in the differential diagnosis of patients on ACE inhibitors presenting with abdominal pain.
There are reports when changing a patient to another ACE inhibitor was followed by recurrence of angioedema and others where it was not. Because of the potential severity of this rare event, another ACE inhibitor should not be used in patients with a history of angioedema, to a drug of this class (see Section 4.3 Contraindications).
The combined use of perindopril/indapamide 4/1.25 tablets with sacubitril/valsartan fixed dose combinations is contraindicated due to the increased risk of angioedema (see Section 4.3 Contraindications).
Sacubitril/valsartan fixed dose combinations must not be initiated until 36 hours after taking the last dose of perindopril/indapamide 4/1.25 tablets. If treatment with sacubitril/valsartan fixed dose combinations is stopped, perindopril/indapamide 4/1.25 tablets must not be initiated until 36 hours after the last dose of sacubitril/valsartan fixed dose combination (see Section 4.3 Contraindications; Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
The combined use of perindopril/indapamide 4/1.25 tablets with NEP inhibitors, mammalian target of rapamycin (mTOR) inhibitors (e.g. temsirolimus, sirolimus, everolimus) and gliptins (e.g. linagliptin, saxagliptin, sitagliptin, vildagliptin, alogliptin) may lead to an increased risk of angioedema (e.g. swelling of the airways or tongue, with or without respiratory impairment) (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions). Caution should be used when commencing treatment with these above-mentioned medicines in a patient already taking an ACE inhibitor.
Anaphylactoid reactions during low-density lipoproteins (LDL) apheresis and haemodialysis.
Rarely, patients treated with ACE inhibitors during LDL apheresis with dextran sulfate have experienced life-threatening anaphylactoid reactions. These reactions were avoided by temporarily withholding ACE inhibitor treatment prior to each apheresis.
Anaphylactoid reactions have been reported in patients dialysed with high flux membranes, who are treated with an ACE inhibitor. Extracorporeal treatments leading to contact of blood with negatively charged surfaces (e.g. polyacrylonitrile membranes such as "AN69") are contraindicated. If treatment is required, consideration should be given to using a different type of dialysis membrane (e.g. cuprophane or polysulfone PSF) or a different class of antihypertensive medicine (see Section 4.3 Contraindications; Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
Anaphylactic reactions during desensitisation.
Patients treated with ACE inhibitors during desensitisation treatment (e.g. hymenoptera venom) have experienced anaphylactoid reactions. In the same patients, these reactions have been avoided when the ACE inhibitors were temporarily withheld, but they reappeared upon inadvertent re-challenge.
Hypotension.
Hypotension has been reported in patients commencing treatment with ACE inhibitors. Symptomatic hypotension is rarely seen in uncomplicated hypertension but is a potential consequence of perindopril use in patients with salt/volume depletion, for example, in patients vigorously treated with diuretics, on dialysis, with renal impairment, following severe diarrhoea or vomiting, on dietary restrictions, or in those with severe renin-dependent hypertension (see Section 4.8 Adverse Effects (Undesirable Effects)).
In patients with symptomatic heart failure, with or without associated renal insufficiency, symptomatic hypotension has been observed. This is more likely to occur in those patients
with severe heart failure, as reflected by the use of high doses of loop diuretics, hyponatraemia or functional renal impairment. This may be associated with syncope, neurological deficits, oliguria and/or progressive increase in blood nitrogen and rarely with acute renal failure and/or death. Because of the potential fall in blood pressure in these patients, treatment should be started at low doses under very close supervision. Such patients should be followed closely for the first two weeks of treatment and whenever the dose is increased.
Patients with ischaemic heart or cerebrovascular disease in whom an excessive fall in blood pressure could result in myocardial infarction or cerebrovascular accident should be closely followed for the first two weeks of treatment and whenever the dose is increased. In all high risk patients it is advisable to initiate treatment either at a low dose or with one component only.
In some patients with congestive heart failure who have normal or low blood pressure, additional lowering of systemic blood pressure may occur with perindopril. This is anticipated and is usually not a reason to discontinue treatment. If symptomatic hypotension occurs, a reduction of dose or discontinuation may be necessary.
If hypotension occurs the patient should be placed in a supine position and if necessary infused with normal saline. A transient hypotensive response is not a contraindication to further doses which can usually be given without difficulty when blood pressure has increased following volume expansion.
Renovascular hypertension.
If renovascular hypertension is also present, treatment should be started under close medical supervision with low doses and careful dose titration. There is an increased risk of severe hypotension and renal insufficiency. Since treatment with diuretics may be a contributing factor to the above, they should be discontinued and renal function should be monitored during the first weeks of perindopril treatment. Loss of renal function may occur with only minor changes in serum creatinine even in patients with unilateral renal artery stenosis.
Kidney transplantation.
There is no experience regarding the administration of perindopril in patients with a recent kidney transplantation.
Hepatic failure.
Rarely, ACE inhibitors have been associated with a syndrome that starts with cholestatic jaundice and progresses to fulminant hepatic necrosis and (sometimes) death. The mechanism of this syndrome is not understood. Patients receiving ACE inhibitors who develop jaundice or marked elevations of hepatic enzymes should discontinue the ACE inhibitor and receive appropriate medical follow-up (see Section 4.8 Adverse Effects (Undesirable Effects)).
Ethnicity.
ACE inhibitors cause a higher rate of angioedema in patients of indigenous African origin than in patients of other racial origin. As with other ACE inhibitors, perindopril may be less effective in lowering blood pressure in people of indigenous African origin than in people of other racial origin, possibly because of a higher prevalence of low-renin states in this population. It is unknown if the same observations have been made in patients of indigenous Australian origin.
Cardiac failure/severe cardiac insufficiency (grade IV).
Patients with severe cardiac insufficiency (grade IV) should be monitored closely during the initial stages of treatment. Treatment should be initiated with a reduced dose.
Treatment with beta-blockers in hypertensive patients with coronary insufficiency should not be stopped. When initiating, the ACE inhibitor should be added to the beta-blocker.
Cough.
A persistent dry (non-productive) irritating cough has been reported with most of the ACE inhibitors. The frequency of reports has been increasing since cough was first recognised as a class-effect of ACE inhibitor treatment with the incidence of cough varying depending upon the ACE inhibitor, dosage and duration of use.
The cough is often worse when lying down or at night, and has been reported more frequently in women (who account for two thirds of the reported cases). Patients who cough may have increased bronchial reactivity compared with those who do not. The observed higher frequency of this side-effect in non-smokers may be due to a higher level of tolerance of smokers to cough.
The cough is most likely due to stimulation of the pulmonary cough reflex by kinins (bradykinin) and/or prostaglandins which accumulate because of ACE inhibition. Once a patient has developed an intolerable cough, an attempt may be made to switch the patient to another ACE inhibitor; the reaction may recur but this is not invariably the case. A change to another class of medicines may be required in severe cases.
Proteinuria.
Perindopril monotherapy has occasionally been associated with mild or transient proteinuria (< 1 gram per 24 hours). However in the majority of patients with pre-existing proteinuria treated with perindopril, proteinuria disappeared or remained stable. ACE inhibitors have potential to delay the progression of nephropathy in patients with diabetes or hypertension.
Neutropaenia/ agranulocytosis/ thrombocytopaenia/ anaemia.
Neutropaenia/agranulocytosis, thrombocytopaenia and anaemia have been reported in patients treated with an ACE inhibitor. In patients with normal renal function and no other complicating factors, neutropaenia occurs rarely. Perindopril should be used with extreme caution in patients with collagen vascular disease, immunosuppressant therapy, treatment with allopurinol or procainamide, or a combination of these complicating factors, especially if there is pre-existing renal impairment. Some of these patients developed serious infections, which in a few instances did not respond to intensive antibiotic treatment. If perindopril is used in such patients, periodic monitoring of white blood cell counts is advised and patients should be instructed to report any sign of infection (e.g. sore throat, fever).
Dermatological reactions.
Dermatological reactions characterised by maculo-papular pruritic rashes and sometimes photosensitivity have been reported with another ACE inhibitor. Rare and sometimes severe skin reactions (lichenoid eruptions, psoriasis, pemphigus like rash, rosacea, Stevens-Johnson syndrome etc.) have been reported following administration of perindopril and may therefore occur. A causal relationship is difficult to assess.
Patients who develop a cutaneous reaction with one ACE inhibitor might not when switched to another medicine of the same class, but there are reports of cross-reactivity.
Taste disturbances (dysgeusia).
Taste disturbances were reported to be high (up to 12.5%) with high doses of one ACE inhibitor. The actual incidence of taste disturbance is probably low (< 0.5%) but data in this respect is scarce and difficult to interpret.
Taste disturbances with ACE inhibitors have been described as suppression of taste or a metallic sensation in the mouth. Dysgeusia usually occurs in the first weeks of treatment and may disappear in most cases within one to three months.
Medicines causing renin release.
The effects of perindopril may be enhanced by concomitant administration of antihypertensive medicines which cause renin release.
Dual blockade of the renin-angiotensin-aldosterone system (RAAS).
As a consequence of inhibiting the RAAS, hypotension, syncope, stroke, hyperkalaemia, and changes in renal function (including acute renal failure) have been reported in susceptible individuals, especially if combining medicines that affect this system. Dual blockade of the RAAS through the combined use of ACE inhibitors, angiotensin II receptor blockers or aliskiren is therefore not recommended (see Section 4.3 Contraindications; Section 4.5 Interactions with Other Medicines and Other Forms of Interactions). If dual blockade therapy is considered absolutely necessary, this should be limited to individually defined cases, under specialist supervision with frequent close monitoring of renal function, electrolytes and blood pressure.
The combination of perindopril with aliskiren is contraindicated in patients with diabetes or renal impairment (GFR < 60 mL/min/1.73 m2) (see Section 4.3 Contraindications; Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
ACE inhibitors and angiotensin receptor blockers should not be used in combination in patients with diabetic nephropathy.
Surgery and anaesthesia.
Perindopril may block angiotensin II formation secondary to compensatory renin release in patients undergoing major surgery or during anaesthesia, with agents that produce hypotension and cause further reduction in blood pressure. Treatment should be discontinued one day prior to surgery.
Perioperative hypotension can be corrected with, volume expansion.
Aortic or mitral valve stenosis/ hypertrophic cardiomyopathy.
There has been some concern on theoretical grounds that patients with mitral valve stenosis and obstruction in the outflow of the left ventricle such as with aortic stenosis or with hypertrophic cardiomyopathy might be at particular risk of decreased coronary perfusion when treated with vasodilators, including ACE inhibitors. Vasodilators may tend to drop diastolic pressure, and hence coronary perfusion pressure, without producing the concomitant reduction in myocardial oxygen demand that normally accompanies vasodilation. The true clinical importance of this concern is uncertain.
Stable coronary artery disease.
If an episode of unstable angina pectoris, regardless of severity, occurs during the first month of perindopril treatment, a careful appraisal of the benefits/risks of continuing treatment should be performed.
Primary aldosteronism.
Patients with primary hyperaldosteronism will generally not respond to antihypertensive medicines acting through inhibition of the renin-angiotensin system. Therefore, treatment with Perindopril Indapamide-WGR 4/1.25 is not recommended.
Related to indapamide component.
Water and electrolyte balance. Hypovolaemia, resulting from the loss of water and sodium caused by the diuretic at the start of treatment, causes a reduction in glomerular filtration. It may result in an increase in urea and creatinine levels. This transitory functional renal insufficiency is of no adverse consequence in patients with normal renal function but may however worsen a pre-existing renal impairment.
Plasma sodium.
This must be measured before starting treatment and then subsequently at regular intervals as treatment with any diuretic may cause hyponatraemia, sometimes with very serious consequences.
The decrease in plasma sodium may be asymptomatic initially. Regular monitoring is therefore essential, and should be even more frequent in the elderly and patients with cirrhosis (see Section 4.8 Adverse Effects (Undesirable Effects); Section 4.9 Overdose). Hyponatraemia with hypovolaemia may be responsible for dehydration and orthostatic hypotension. Concomitant loss of chloride ions may lead to secondary compensatory metabolic alkalosis.
Plasma potassium.
Potassium depletion with hypokalaemia is the major risk of thiazide and related diuretics.
Hypokalaemia may cause muscle disorders. Cases of rhabdomyolysis have been reported, mainly in the context of severe hypokalaemia. The risk of onset of hypokalaemia (< 3.4 mmol/L) must be prevented in certain high risk populations, i.e. the elderly, malnourished and/or polymedicated, cirrhotic patients with oedema and ascites, and patients with coronary artery disease and/or heart failure. In these patients, hypokalaemia increases the cardiac toxicity of digitalis preparations and increases the risk of arrhythmias.
Hypokalaemia will be more common when combined with a steroid or adrenocorticotropic (ACTH) treatment and when electrolyte intake is inadequate.
Individuals with a long QT interval, whether the origin is congenital or iatrogenic, are also at increased risk as hypokalaemia and bradycardia, are predisposing factors to the onset of severe arrhythmias, in particular, potentially fatal Torsades de pointes.
Plasma potassium should be measured in the first week of treatment. More frequent monitoring of plasma potassium is required in all the situations indicated above.
Hypokalaemia, if detected, should be corrected. Hypokalaemia found in association with low serum magnesium concentration can be refractory to treatment unless serum magnesium is corrected.
Plasma magnesium.
Thiazides and related diuretics have been shown to increase the urinary excretion of magnesium, which may result in hypomagnesaemia (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions; Section 4.8 Adverse Effects (Undesirable Effects)).
Plasma calcium.
Diuretic treatment should be withdrawn before the investigation of parathyroid function.
Thiazide and related diuretics may decrease urinary calcium excretion and cause a slight and transitory rise in calcium. Frank hypercalcaemia may be due to previously unrecognised hyperparathyroidism.
Hypokalaemia. Hypokalaemia is a particular hazard in digitalised patients since dangerous or fatal arrhythmias may be precipitated by it.
Orthostatic hypotension. Orthostatic hypotension may occur and may be potentiated by alcohol, barbiturates, narcotics or concurrent treatment with other antihypertensives.
When indapamide is combined with other non-diuretic antihypertensive agents, the effects on blood pressure are additive.
Lupus erythematosus. Sulfonamide derivatives have been reported to exacerbate or activate systemic lupus erythematosus. Serious allergic skin reactions (such as Stevens-Johnson syndrome) have also occasionally been reported associated with sulfonamides. This should be considered when using indapamide.
Photosensitivity. Cases of photosensitivity reactions have been reported with thiazides and thiazide-related diuretics. If photosensitivity reaction occurs during treatment, it is recommended to stop the treatment. If a re-administration of the diuretic is deemed necessary, it is recommended that areas exposed to the sun or to artificial UVA are protected.
Blood glucose. Monitoring of blood glucose is important in patients with diabetes, in particular in the presence of hypokalaemia.
Athletes. This product contains indapamide which may give a positive result in doping tests.
Choroidal effusion, acute myopia and secondary angle-closure glaucoma. Sulfonamide, or sulfonamide derivatives such as indapamide can cause an idiosyncratic reaction resulting in choroidal effusion with visual field defect, transient myopia and acute angle-closure glaucoma.
Symptoms include acute onset of decreased visual acuity or ocular pain and typically occur within hours to weeks of drug initiation. Untreated acute angle-closure glaucoma can lead to permanent vision loss. The primary treatment is to discontinue drug intake as rapidly as possible. Prompt medical or surgical treatments may need to be considered if the intraocular pressure remains uncontrolled. Risk factors for developing acute angle-closure glaucoma may include a history of sulfonamide and penicillin allergy.
Use in hepatic impairment.
Related to perindopril component.
Biotransformation of perindopril to perindoprilat mainly occurs in the liver. Studies in patients with hepatic impairment have shown that kinetic parameters of perindopril were not modified by hepatic failure. With the exception of bioavailability, which was increased, kinetic parameters of perindoprilat (including Tmax) were also unchanged. The increase in bioavailability could be due to inhibition of the formation of perindopril metabolites other than perindoprilat (see Section 5.2 Pharmacokinetic Properties, Pharmacokinetics of perindopril). The administration of perindopril leads to the formation of a glucuronoconjugate derivative of perindoprilat by a hepatic first pass effect. The kinetic parameters of perindoprilat glucuronide are not modified by hepatic failure. The small changes in the kinetics of perindoprilat do not justify the need to change the usual dose in most patients with hepatic failure.
Related to indapamide component.
When liver function is impaired, thiazide and thiazide-related diuretics may cause, particularly in case of electrolyte imbalance, hepatic encephalopathy which can progress to hepatic coma. Caution should be used in treating patients with severe hepatic disease to avoid metabolic alkalosis in cases of potassium depletion, which may precipitate episodes of hepatic encephalopathy. Treatment with the diuretic must be stopped immediately if this occurs.
Use in renal impairment.
Related to perindopril 4 mg/indapamide 1.25 mg combination.
Use of perindopril 4 mg/indapamide 1.25 mg is contraindicated in patients with severe renal impairment (creatinine clearance < 30 mL/min).
Perindopril 4 mg/indapamide 1.25 mg is not recommended in patients with bilateral renal artery stenosis or a single functioning kidney.
Treatment with the perindopril 4 mg/indapamide 1.25 mg product should be stopped and possibly restarted either at a low dose or with either indapamide or perindopril only in hypertensive patients whose blood tests show functional renal insufficiency. In these patients usual medical follow-up should include frequent monitoring of potassium and creatinine, after two weeks of treatment and then every two months during therapeutic stability period. Renal failure has been reported mainly in patients with severe heart failure or underlying renal failure including renal artery stenosis.
Related to perindopril component.
As a consequence of inhibiting the renin-angiotensin-aldosterone system (RAAS), changes in renal function may be anticipated in susceptible individuals. In patients with severe congestive heart failure whose renal function may depend on RAAS activity, treatment with ACE inhibitors may be associated with oliguria and/or progressive increase in blood nitrogen, and rarely with acute renal failure and/or death.
In patients with symptomatic heart failure, hypotension following the initiation of treatment with ACE inhibitors may lead to further impairment in renal function. Acute renal failure, usually reversible, has been reported in this situation.
ACE inhibitors should be avoided in patients with known or suspected renal artery stenosis (see Section 4.3 Contraindications).
In clinical studies in patients with hypertension with unilateral or bilateral renal artery stenosis, increases in blood urea, nitrogen and serum creatinine were observed in 20% of patients. Acute renal impairment may also occur. These increases are usually reversible upon discontinuation of treatment. When an ACE inhibitor is given to a patient with stenosis of the artery supplying a solitary kidney or bilateral renal artery stenosis, acute renal insufficiency may occur. Renal function may also be reduced in patients with stenosis of the artery supplying a transplanted kidney. It is believed that renal artery stenosis reduces the pressure in the afferent glomerular arteriole, and transglomerular hydrostatic pressure is then maintained by angiotensin II-induced constriction of the efferent arteriole. When an ACE inhibitor is given, the efferent arteriole relaxes, glomerular filtration pressure falls, and renal failure may result. ACE inhibitors can lead to the thrombotic occlusion of a stenosed renal artery.
Some patients with hypertension and no apparent pre-existing renovascular disease have developed increases in blood urea, nitrogen and serum creatinine which are usually minor and transient, particularly when perindopril has been combined with a diuretic. However, increases in blood urea nitrogen and serum creatinine are more likely to occur in patients with pre-existing renal impairment or in those on diuretics. Dose reduction of the ACE inhibitor and/or discontinuation of the diuretic may be required.
Renal function should always be assessed (see Section 4.2 Dose and Method of Administration). In the case of renal impairment, the initial perindopril dose should be adjusted according to the patient's creatinine clearance (see Section 4.2 Dose and Method of Administration). Routine monitoring of potassium and creatinine are part of normal medical practice for these patients (see Section 4.8 Adverse Effects (Undesirable Effects)). If a deterioration in renal function has occurred after treatment with one ACE inhibitor, then it is likely to be precipitated by another and in these patients use of another class of antihypertensive agent would be preferable. Patients with unilateral renal artery disease present a special problem as deterioration of function may not be apparent from measurement of blood urea and serum creatinine.
Some ACE inhibitors have been associated with the occurrence of proteinuria (up to 0.7%) and/or decline in renal function in patients with one or more of the following characteristics: old age, pre-existing renal disease, combined use with potassium sparing diuretics or high doses of other diuretics, limited cardiac reserve or treatment with a non-steroidal anti-inflammatory medicine (NSAID).
Anaemia has been observed in patients who have had a kidney transplant or have been undergoing dialysis. The reduction in haemoglobin levels is more apparent as initial values were high. This effect does not seem to be dose-dependent but may be linked to the mechanism of action of angiotensin converting enzyme inhibitors. This reduction in haemoglobin is slight, occurs within one to six months, and then remains stable. It is reversible when treatment is stopped. Treatment can be continued with regular haematological testing.
Perindopril is dialysable with a clearance of 70 mL/minute.
Related to indapamide component.
Although indapamide can safely be administered to hypertensive patients with impaired renal function, the treatment should be discontinued if increases in blood nitrogen and oliguria occur. Studies in functionally anephric patients on indapamide monotherapy for one month undergoing chronic haemodialysis have not shown evidence of drug accumulation, despite the fact that indapamide is not dialysable.
Thiazide diuretics and thiazide-related diuretics are only fully effective when renal function is normal or only slightly impaired (creatinine levels lower than approximately 25 mg/L, i.e. 220 micromol/L for an adult).
Use in the elderly.
Renal impairment is commonly observed in elderly people. Care should therefore be taken when prescribing perindopril-containing medicines to elderly hypertensive patients. The initial daily dose in the elderly should always be at a low dose or with one component only, and patients should be monitored closely during the initial stages of treatment. (See Section 4.2 Dose and Method of Administration).
In the elderly the value of plasma creatinine levels should be adjusted to take account of the age, weight and sex of the patient.
In a study of 91 elderly patients with a mean age of 71.9 years, a 6% increase in serum potassium occurred in the first month of treatment and subsequently remained stable. There was no change in the group in blood urea, creatinine or creatinine clearance.
Particular care should be taken in elderly patients with congestive heart failure who have renal and/or hepatic impairment.
Paediatric use.
Use of a perindopril 4 mg/ indapamide 1.25 mg combination product in children is not recommended as no data establishing safety or effectiveness in children are available.
Effects on laboratory tests.
Potassium depletion with particularly serious reduction in levels of potassium in some at risk populations (see Section 4.4 Special Warnings and Precautions for Use).
Reduced sodium levels with hypovolaemia causing dehydration and orthostatic hypotension.
Increase in uric acid levels and blood glucose levels during treatment.
Slight increase in urea and in plasma creatinine levels, reversible when treatment is stopped. This increase is more frequent in cases of renal artery stenosis, arterial hypertension treated with diuretics, renal insufficiency, renovascular hypertension and severe heart failure.
Increased levels of potassium, usually transitory.
Elevation of liver enzymes and serum bilirubin have been reported rarely.
Rarely, raised plasma calcium levels have been noted.4.5 Interactions with Other Medicines and Other Forms of Interactions
The combined use of perindopril and indapamide in a perindopril 4 mg/ indapamide 1.25 mg fixed dose product is not associated with additional interactions with concomitant medicines other than those known for each of these components.
Shared by perindopril and indapamide.
Combined use which is not recommended. Lithium.
Reversible increases in serum lithium concentrations and toxicity have been reported during concomitant administration of lithium with ACE inhibitors. Combined use of thiazide diuretics may increase the risk of lithium toxicity and enhance the already increased risk of lithium toxicity with ACE inhibitors. Use of perindopril combined with indapamide with lithium is not recommended, but if the combination is necessary, careful monitoring of serum lithium levels should be performed (see Section 4.4 Special Warnings and Precautions for Use).
Combined use which requires special care. Baclofen.
Increased antihypertensive effect. It is recommended that adequate hydration and renal function be monitored at the start of treatment. Monitoring of blood pressure and renal function, and adequate hydration is recommended with dose adaptation of the antihypertensive to occur if necessary.
Non-steroidal anti-inflammatory medicines (NSAIDs), including acetylsalicylic acid ≥ 3 g/day.
Medicines with prostaglandin synthetase inhibitor properties (e.g. indometacin) an NSAID (i.e. acetylsalicylic acid at anti-inflammatory dose regimens, non-selective NSAIDs or COX-2 inhibitors), may diminish the antihypertensive efficacy of concomitantly-administered ACE inhibitors. However, clinical studies have not demonstrated any interaction between perindopril/ indapamide or indometacin or other NSAIDs. Treatment with an NSAID may lead to an increased risk of worsening of renal function, including possible acute renal failure, and an increase in serum potassium, especially in patients with poor pre-existing renal function. The combination should be administered with caution, especially in the elderly.
Patients should be adequately hydrated. Due to the risk of acute renal failure in patients with dehydration as a result of decreased glomerular filtration, it is recommended that hydration and serum potassium and renal function be monitored at the start of treatment, and periodically thereafter.
Combined use which requires some care. Tricyclic antidepressants/ antipsychotics/ anaesthetics.
Combined use of certain anaesthetic medicines, tricyclic antidepressants and antipsychotics with ACE inhibitors may result in further reduction of blood pressure (see Section 4.4 Special Warnings and Precautions for Use).
Other antihypertensive agents.
Combined use of other antihypertensive agents may increase hypotensive effects.
Related to perindopril component.
Clinical trial data has shown that dual blockade of the renin-angiotensin-aldosterone-system (RAAS) through the combined use of ACE-inhibitors, angiotensin II receptor blockers or aliskiren is associated with higher frequency of adverse events such as hypotension, hyperkalaemia and decreased renal function (including acute heart failure) compared to the use of a single RAAS-acting agent (see Section 4.3 Contraindications; Section 4.4 Special Warnings and Precautions for Use; Section 5.1 Pharmacodynamic Properties, Clinical trials).
Combined use which is contraindicated. See Section 4.3 Contraindications; Section 4.4 Special Warnings and Precautions for Use.
Aliskiren.
Patients with diabetes or renal impairment (GFR < 60 mL/min/1.73 m2), may be at risk of hypotension, syncope, stroke, hyperkalaemia, worsening of renal function (including acute renal failure) and cardiovascular morbidity and mortality increase (see Section 4.3 Contraindications; Section 4.4 Special Warnings and Precautions for Use).
Extracorporeal treatments.
Extracorporeal treatments leading to contact of blood with negatively charged surfaces such as dialysis or haemofiltration with certain high-flux membranes (e.g. polyacrylonitrile membranes such as "AN69") and low density lipoprotein apheresis with dextran sulfate are contraindicated due to increased risk of severe anaphylactoid reactions (see Section 4.3 Contraindications; Section 4.4 Special Warnings and Precautions for Use). If such treatment is required, consideration should be given to using a different type of dialysis membrane (e.g. cuprophane or polysulfone PSF) or a different class of antihypertensive agent.
Sacubitril/valsartan.
The combined use of perindopril/indapamide 4/1.25 tablets with sacubitril/valsartan fixed dose combinations is contraindicated as the concomitant inhibition of neprilysin and ACE may increase the risk of angioedema. Sacubitril/valsartan fixed dose combinations must not be started until 36 hours after taking the last dose of perindopril/indapamide 4/1.25 tablets. Perindopril/indapamide 4/1.25 tablets must not be started until 36 hours after the last dose of sacubitril/valsartan fixed dose combination (see Section 4.3 Contraindications; Section 4.4 Special Warnings and Precautions for Use).
Combined use which is not recommended. See Section 4.4 Special Warnings and Precautions for Use.
Potassium sparing diuretics (amiloride, triamterene).
The combined use of perindopril/indapamide and potassium sparing diuretics may result in potentially lethal hyperkalaemia especially in patients with renal impairment (additive hyperkalaemic effects). The combination of perindopril with the above-mentioned medicines is not recommended (see Section 4.4 Special Warnings and Precautions for Use). If the combination is required, it should be used with caution and with frequent monitoring of serum potassium. For use of spironolactone and eplerenone in heart failure, see paragraph under 'Related to perindopril component, Combined use which requires special care'.
Aliskiren.
Patients other than those with diabetes or renal impairment may be at risk of hyperkalaemia, worsening of renal function and cardiovascular morbidity, and an increase in mortality (see Section 4.3 Contraindications; Section 4.4 Special Warnings and Precautions for Use).
Combined use with ACE inhibitor and angiotensin-receptor blocker.
It is reported in the literature that in patients with established atherosclerosis, heart failure, or diabetes with end organ damage, combined use with an ACE inhibitor and an angiotensin-receptor blocker is associated with a higher frequency of hypotension, syncope, hyperkalaemia, and worsening renal function (including acute renal failure) as compared to use of a single RAAS agent. Dual blockade (e.g. by combining an ACE inhibitor with an angiotensin receptor blocker) should be limited to individually defined cases with close monitoring of renal function, serum potassium, and blood pressure.
Allopurinol, cytostatic or immunosuppressant agents, corticosteroids (main route) or procainamide.
Combination with ACE inhibitors may lead to an increased risk for leucopenia.
Co-trimoxazole (trimethoprim/sulfamethoxazole).
Patients on combined treatment with an ACE inhibitor and co-trimoxazole (trimethoprim/sulfamethoxazole) may be at increased risk of hyperkalaemia (see Section 4.4 Special Warnings and Precautions for Use).
Combined use which requires special care. Medicines used to treat diabetes (insulin, oral hypoglycaemic medicines).
The use of ACE inhibitors may increase the hypoglycaemic effect, with risk of hypoglycaemia, in diabetics receiving treatment with insulin or oral hypoglycaemic sulphonylureas. The onset of hypoglycaemic episodes is very rare (improvement in glucose tolerance with a resulting reduction in insulin requirements) and appeared to be more likely to occur during the first weeks of combined treatment and in patients with renal impairment.
Non-potassium-sparing diuretics.
Patients treated with diuretics, especially those who are volume and/or salt depleted, may experience an excessive reduction in blood pressure after initiation of treatment with an ACE inhibitor.
The possibility of hypotensive effects can be reduced by discontinuation of the diuretic or by increasing salt intake prior to commencing treatment with low and progressive doses of perindopril 4 mg/ indapamide 1.25 mg combination product. If it is not possible to discontinue the diuretic, the starting dose of the ACE-inhibitor should be reduced. The patient should be closely observed for several hours following the initial dose of the ACE inhibitor and until the blood pressure has stabilised. In arterial hypertension, when prior diuretic treatment has caused salt/volume depletion, the diuretic must be discontinued before commencing treatment with the ACE inhibitor. The ACE inhibitor must be commenced at a low dose and progressively increased prior to a non-potassium-sparing diuretic being commenced.
In diuretic-treated congestive heart failure, the ACE inhibitor should be initiated at a very low dose, possibly after reducing the dose of the associated non-potassium-sparing diuretic.
In all cases, renal function (creatinine levels) must be monitored during the first few weeks of ACE inhibitor treatment.
Potassium-sparing diuretics (eplerenone, spironolactone).
As the combination of perindopril and potassium sparing medicines (e.g. eplerenone and spironolactone), potassium supplements or potassium-containing salt substitutes is generally not recommended:
Ensure patients do not have hyperkalaemia or renal impairment before commencing treatment with this combination.
There is a risk of potentially lethal hyperkalaemia with this combination in patients treated for NYHA Class II-IV heart failure with a reduced ejection fraction, who have been previously treated with ACE inhibitors and loop diuretics. This risk is particularly high when recommendations for use of this combination have not been followed.
Weekly monitoring of serum potassium and creatinine levels is recommended in the first month of the treatment and, monthly thereafter.
Combined use of ACE inhibitors, anti-inflammatory medicines and thiazide diuretics.
The combined use of an ACE inhibiting medicine (ACE-inhibitor or angiotensin receptor blocker), an anti-inflammatory medicine (NSAID or COX-2 inhibitor) and a thiazide diuretic increases the risk of renal impairment. This includes use in fixed-combination products. The combination of medicines from these three classes should be used with caution particularly in elderly patients or those with pre-existing renal impairment. Combined use of these medications should be accompanied by increased monitoring of serum creatinine, particularly at initiation.
Ciclosporin.
Hyperkalaemia may occur during the combined use of ACE inhibitors with ciclosporin. Frequent monitoring of serum potassium is recommended.
Heparin.
Hyperkalaemia may occur during the combined use of ACE inhibitors with heparin. Frequent monitoring of serum potassium is recommended.
Mammalian target of rapamycin (mTOR) inhibitor (e.g. temsirolimus, sirolimus, everolimus).
Patients on combined treatment with an ACE inhibitor and an mTOR inhibitor may be at increased risk of angioedema (see Section 4.4 Special Warnings and Precautions for Use).
Gliptins (e.g. linagliptin, saxagliptin, sitagliptin, vildagliptin, alogliptin).
When an ACE inhibitor and a gliptin are used in combination, there is an increased risk of angioedema due to the decreased activity of the dipeptidyl peptidase IV (DPP-IV).
Combined use which requires some care. Gold.
Nitritoid reactions (symptoms include facial flushing, nausea, vomiting and hypotension) have been reported rarely in patients on therapy with injectable gold (sodium aurothiomalate) and concomitant ACE inhibitor therapy including perindopril.
Antihypertensive agents and vasodilators.
Combined use of these medicines may increase the hypotensive effects of perindopril. Combined use with nitroglycerin and other nitrates, or other vasodilators, may further reduce blood pressure.
Medicines affecting sympathetic activity.
As the sympathetic nervous system plays an important part in physiological blood pressure regulation, caution should be exercised when combined administration of a medicine with sympathetic activity. Sympathomimetics may reduce the antihypertensive effects of ACE inhibitors.
Acetylsalicylic acid, thrombolytics, beta-blockers, nitrates.
Perindopril may be combined with thrombolytics, aspirin (when used as a thrombolytic), beta-blockers and/or nitrates.
Tetracycline and other medicines that interact with magnesium.
The simultaneous administration of tetracycline with an ACE inhibitor may significantly reduce the absorption of tetracycline, possibly due to the magnesium content in the ACE inhibitor tablets. This interaction should be considered if co-prescribing an ACE inhibitor and tetracycline or other medicines that interact with magnesium.
Related to indapamide component.
No interactions have been reported between indapamide anticoagulants and, uricosurics, however it is recommended that these products not be used in combination with a diuretic agent since the combination may produce hypokalaemia and hyperuricaemia.
Combined use which requires special care. Torsades de pointes inducing medicines.
The combined use of indapamide and Torsades de pointes-inducing medicines, is not recommended due to the increased risk of ventricular arrhythmias and hypokalaemia. Medicines which can induce Torsades de pointes include:
class Ia antiarrhythmic agents (e.g. disopyramide) and class Ic antiarrhythmic agents (e.g. flecainide);
class III antiarrhythmic agents (e.g. amiodarone, sotalol);
some antipsychotics: phenothiazines (e.g. chlorpromazine, trifluoperazine), benzamides (e.g. amisulpride, sulpiride), butyrophenones (e.g. droperidol, haloperidol) and other antipsychotics;
psychoanaleptic (e.g. donepezil);
tricyclic antidepressants (e.g. citalopram, escitalopram);
antimicrobial agents: fluoroquinolones (e.g. moxifloxacin, ciprofloxacin), macrolides (e.g. erythromycin IV, clarithromycin), azole antifungals (e.g. fluconazole);
antiparasitics (e.g. chloroquine, pentamidine);
antihistamines;
antiemetics (e.g. ondansetron, domperidone);
antineoplastic and immunomodulating agents (e.g. vandetanib, oxaliplatin, anagrelide);
anaesthetics (e.g. propofol, sevoflurane);
others: diphemanil, methadone, papaverine, cilostazol.
This list is indicative and not exhaustive. While not recommended, if combination of indapamide and a Torsades de pointes-inducing medicine is deemed necessary; hypokalaemia should be monitored (using plasma electrolytes and ECG) and corrected if required, before using indapamide and a Torsades de pointes-inducing medicine in combination.
Antiarrhythmics should not be administered in cases of torsades de pointes (management by pacemaker).
Potassium lowering medications: amphotericin B (amphotericin) (IV route), glucocorticoids and mineralocorticoids (systemic route), tetracosactide (tetracosactrin), stimulant laxative.
Due to the increased risk of hypokalaemia (additive effect): monitoring, and correction if required, of plasma potassium (especially during treatment with digoxin) is recommended; the use of non-stimulant laxatives is recommended.
Digitalis preparations.
Hypokalaemia and/or hypomagnesaemia predispose to the toxic effects of digitalis. Monitoring of plasma potassium, magnesium and ECG is recommended and, if necessary, adjusting the treatment. Monitoring of plasma potassium, plasma magnesium and ECG is recommended during treatment with digitalis, and if necessary, adjust the treatment as hypokalaemia and/or hypomagnesaemia predispose to the toxic effects of digitalis.
Allopurinol.
Combined use with indapamide may increase the incidence of hypersensitivity reactions to allopurinol.
Combined use which requires some care. Potassium sparing diuretics (amiloride, spironolactone, triamterene).
Due to the increased risk of either hyperkalaemia or hypokalaemia, particularly in patients with renal failure or diabetes, care should be taken when co-administering potassium-sparing diuretics. Potassium levels and ECG should be monitored and, if necessary, treatment reviewed.
Metformin.
Do not combine with metformin when plasma creatinine exceeds 135 micromol/L (15 mg/L) in men and 110 micromol/L (12 mg/L) in women due to the increased risk of metformin induced lactic acidosis as a result of the possibility of functional renal failure associated with diuretics, particularly loop diuretics.
Iodinated contrast media.
Adequate hydration before administration of the iodinated compound is recommended due to an increased risk of acute renal failure resulting from dehydration, particularly when large doses of iodinated contrast media are used.
Calcium (salts).
Caution is recommended with these combinations due to the risk of hypercalcaemia resulting from decreased urinary elimination of calcium.
Immunosuppressants (ciclosporin, tacrolimus).
Caution is recommended with this combination due to the risk of increased plasma creatinine without any change in circulating ciclosporin levels, even in the absence of water/sodium depletion.
Corticosteroids (systemic route), tetracosactide (tetracosactrin).
Caution is recommended with this combination due to the risk of reduced antihypertensive effect (water/sodium retention due to corticosteroids).4.6 Fertility, Pregnancy and Lactation
Effects on fertility.
The effect of the combination on fertility has not been investigated.
Related to perindopril component.
Studies in rats showed no impairment of male or female fertility at oral perindopril doses up to 10 mg/kg/day.
Related to indapamide component.
A reproduction study in rats showed no impairment of male or female fertility at oral indapamide doses up to 25 mg/kg/day, however, the number of implantation sites was reduced at the highest dose. In animals treated with oral doses of indapamide, a reduction in the number of implantation sites was seen at 25 mg/kg/day.
(Category D)
As this combination contains an ACE inhibitor, perindopril/indapamide is contraindicated during pregnancy (see Section 4.3 Contraindications).
Related to a perindopril 4 mg/ indapamide 1.25 mg combination.
Reproductive toxicity studies in rats and rabbits showed evidence of increased maternal toxicity and increased embryotoxicity (including delayed foetal development and embryonic deaths) when perindopril and indapamide are given in combination than when each medicine is given separately.
Related to perindopril component.
The use of ACE inhibitors is contra-indicated during pregnancy (see Section 4.3 Contraindications).
As with all ACE inhibitors, perindopril should not be taken during pregnancy. Pregnancy should be excluded before starting treatment with Perindopril Indapamide-WGR 4/1.25 and avoided during the treatment.
Unless continued treatment with an ACE inhibitor therapy is considered essential, patients planning pregnancy should be changed to alternative anti-hypertensive treatments which have an established safety profile for use in pregnancy. If a patient intends to become pregnant, treatment with ACE inhibitors must be discontinued and replaced by another form of treatment. If a patient becomes pregnant while on ACE inhibitors, she must immediately inform her doctor to discuss a change in medication and further management.
Perindopril or its metabolites have been shown to cross the placenta and distribute to the foetus in pregnant animals. There are no adequate and well controlled studies of ACE inhibitors in pregnant women, but foetotoxicity is well documented in animal models. Data, however, show that ACE inhibitors cross the human placenta. Post marketing experience with all ACE inhibitors suggests that exposure in utero may be associated with hypotension and decreased renal perfusion in the foetus.
The ACE inhibitor class has also been associated with foetal death in utero.
Epidemiological evidence regarding the risk of teratogenicity following exposure to ACE inhibitors during the first trimester of pregnancy has not been conclusive; however a small increase in risk cannot be excluded.
A historical cohort study in over 29,000 infants born to non-diabetic mothers has shown 2.7 times higher risk for congenital malformations in infants exposed to ACE inhibitors during first trimester compared to no exposure. The risk ratios for cardiovascular and central nervous system malformations were 3.7 times (95% confidence interval 1.89 to 7.3) and 4.4 times (95% confidence interval 1.37 to 14.02) respectively, compared to no exposure.
When ACE inhibitors have been used during the second and third trimesters of pregnancy, there have been reports of foetal and neonatal toxicity: hypotension, hyperkalaemia, renal failure, skull hypoplasia, oligohydramnios and death.
Oligohydramnios has been reported, presumably resulting from decreased foetal renal function; oligohydramnios has been associated with foetal limb contractures, craniofacial deformities, hypoplastic lung development and intra-uterine growth retardation. Prematurity and patent ductus arteriosus have been reported, however it is not clear whether these events were due to ACE inhibitor exposure or to the mother's underlying disease.
Infants exposed in utero to ACE inhibitors should be closely observed for hypotension, oliguria and hyperkalaemia. Should exposure to ACE inhibitors have occurred from the second trimester of pregnancy, ultrasound check of renal function and skull is recommended. If such complications arise, appropriate medical treatment should be initiated to support blood pressure and renal perfusion.
Related to indapamide component.
Indapamide should be avoided in pregnant women and should not be used to treat oedema in pregnancy.
There are limited data with the use of indapamide in pregnant women. Prolonged exposure to thiazides during the third trimester of pregnancy can reduce maternal plasma volume as well as uteroplacental blood flow, which may cause foetal-placental ischaemia and growth retardation.
Animal studies do not indicate direct or indirect harmful effects with respect to reproductive toxicity.
Indapamide or its metabolites have been shown to cross the placenta and distribute in the foetus in pregnant animals. Thiazides, related diuretics and loop diuretics enter the foetal circulation and may cause electrolyte disturbances. Neonatal thrombocytopaenia has been reported with thiazides and related diuretics. Loop diuretics like furosemide (frusemide) and bumetanide are probably also associated with this risk. During the latter part of pregnancy products of this type should only be given on sound indications, and then in the lowest effective dose.
Diuretics can cause foetoplacental ischaemia, with a risk of impaired foetal growth.
Thiazides, related diuretics and loop diuretics enter the foetal circulation and may cause electrolyte disturbances. Neonatal thrombocytopaenia has been reported with thiazides and related diuretics. Loop diuretics like furosemide (frusemide) and bumetanide are probably also associated with this risk. During the latter part of pregnancy medicines of this type should be used with caution, and at the lowest effective dose.
A perindopril 4 mg/ indapamide 1.25 mg combination product is contraindicated during lactation (see Section 4.3 Contraindications). A decision, taking into account the importance of this treatment for the mother, should therefore be made whether to discontinue nursing or to discontinue the perindopril 4 mg/ indapamide 1.25 mg combination product.
Related to perindopril component.
Animal studies have shown that perindopril and its metabolites are excreted in milk during lactation, but there are no human data. It is therefore recommended that perindopril should not be given to lactating women as the possible effect on the newborn is unknown. Alternative treatments with better established safety profiles during breast-feeding are preferable, especially while nursing a newborn or preterm infant.
Related to indapamide component.
Indapamide should not be used during breastfeeding (see Section 4.3 Contraindications).
Indapamide is excreted in human breast milk and the possible effect on the newborn is unknown and cannot be excluded. Indapamide is closely related to thiazide diuretics which have been associated with decrease in or even suppression of lactation. Serious adverse reactions might occur in nursing infants such as hypersensitivity to sulfonamide-derived medicines, hypokalaemia and nuclear icterus.4.7 Effects on Ability to Drive and Use Machines
Neither of the two active substances nor the combination affect alertness but individual reactions related to low blood pressure may occur in some patients, particularly at the start of treatment or in combination with another antihypertensive medication. As a result the ability to drive or operate machinery may be impaired.
4.8 Adverse Effects (Undesirable Effects)
Reported with use of a perindopril 4 mg/ indapamide 1.25 mg combination.
Adverse effects have generally been mild and transient and have not required discontinuation of treatment. In controlled clinical trials, discontinuation of treatment due to clinical adverse effects was required in only 2.1% of patients treated with both the combination or placebo.
The administration of perindopril inhibits the renin-angiotensin-aldosterone axis and tends to reduce the potassium loss caused by indapamide. During clinical trials a reduction in potassium levels to less than 3.4 mmol/L was observed in 4% of patients taking a perindopril 4 mg/ indapamide 1.25 mg combination product for 12 weeks. After 12 weeks of treatment, the mean reduction in potassium levels was 0.20 mmol/L.
In a long term study involving 165 patients treated with a dose of perindopril and indapamide equivalent to a perindopril 4 mg/ indapamide 1.25 mg combination product for up to one year, the nature and frequency of adverse effects were similar to those listed below.
The most frequent treatment-emergent adverse effects (incidence > 1%) reported in 3-month controlled clinical trials, including a total of 1,898 patients treated with a dose of perindopril 4 mg/ indapamide 1.25 mg or perindopril 2 mg/ indapamide 0.625 mg and 717 patients treated with placebo, were as follows (see Table 1):
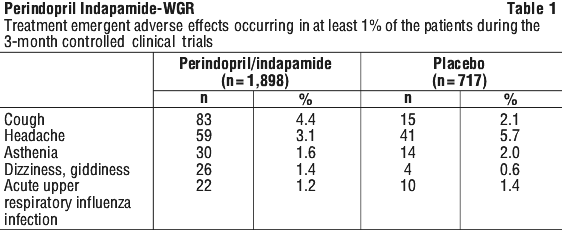
Tabulated list of adverse effects observed during clinical trials and/or postmarketing use.
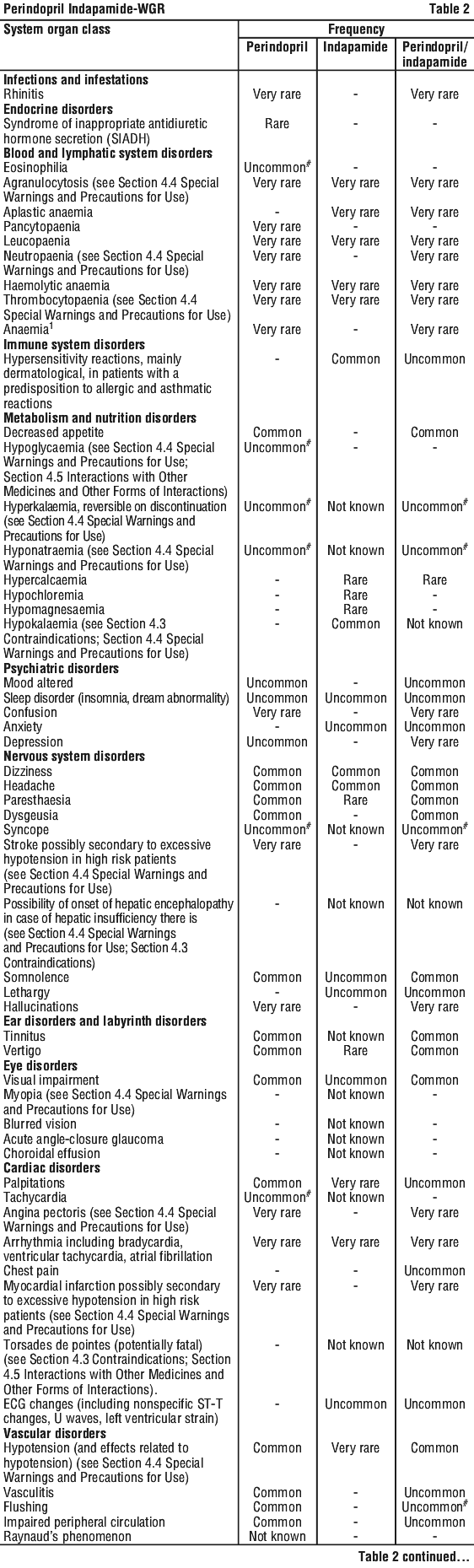
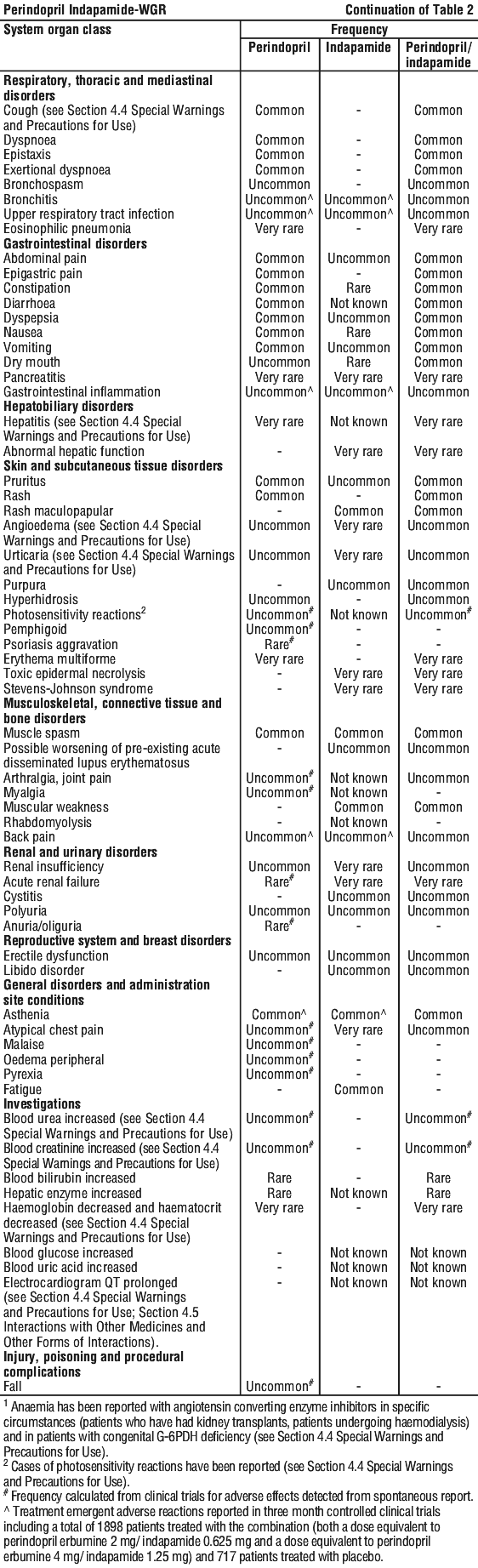
The following adverse effects have been observed during clinical trials and/or post-marketing use and ranked under the following frequency: very common (> 10%); common (> 1%, < 10%); uncommon (> 0.1%, < 1%); rare (> 0.01%, < 0.1%), very rare (> 0.001%, < 0.01%), not known (cannot be estimated from the available data). See Table 2.
Reporting suspected adverse effects.
Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at http://www.tga.gov.au/reporting-problems.4.9 Overdose
Symptoms.
The most likely adverse event in cases of overdose is hypotension, with the possibility of nausea, vomiting, cramps, dizziness, sleepiness, mental confusion, polyuria or oliguria which may progress to anuria (due to hypovolaemia). Salt and water disturbances (low sodium levels, low potassium levels) may occur.
Symptoms associated with overdosage of ACE inhibitors may include hypotension, circulatory shock, electrolyte disturbances, renal failure, hyperventilation, tachycardia, palpitations, bradycardia, dizziness, anxiety, and cough.
Treatment.
If hypotension occurs, the patient should be placed in the shock position. The recommended treatment of overdosage is intravenous infusion of normal saline solution. Vital signs, serum electrolytes and creatinine concentrations should be monitored continuously.
Perindoprilat, the active form of perindopril, may be removed from the general circulation via haemodialysis (see Section 4.4 Special Warnings and Precautions for Use; Section 5.2 Pharmacokinetic Properties, Pharmacokinetics of perindopril).
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action.
Perindopril Indapamide-WGR 4/1.25 is a combination of perindopril erbumine, an angiotensin converting enzyme (ACE) inhibitor, and indapamide, a chlorosulfamoyl diuretic, in which the doses of the ACE inhibitor and diuretic components are up to two times lower than the usual doses used for monotherapy.
Its pharmacological properties are derived from each of its components, in addition to those due to the synergistic action of the two products when combined on vascular endothelium and the target-organs of hypertension, with:
an increase in vasorelaxation and a reduction in vasoconstriction, which are endothelium-dependent;
a regression in renal effects (glomerulosclerosis, proteinuria), myocardial effects (left ventricular hypertrophy) and a reduction in capillary density.
A perindopril 4 mg/ indapamide 1.25 mg combination exerts a dose-dependent antihypertensive effect on diastolic and systolic arterial pressure whilst supine or standing in hypertensive patients regardless of age. This antihypertensive effect lasts for 24 hours. The reduction in blood pressure is obtained in less than one month without tachyphylaxis. Stopping treatment has no associated effects. During clinical trials, the concomitant administration of perindopril and indapamide produced antihypertensive effects of a synergistic nature when compared with each of the products administered alone.
The combination of perindopril with indapamide is justified in the treatment of hypertension due to its action on several pathophysiological mechanisms, and due to the lessening of counter-regulatory mechanisms by one or other of the two components:
indapamide reduces the vascular response to angiotensin II by depleting the cell of sodium and of calcium, whilst perindopril opposes the stimulation of the renin-angiotensin system (RAS) and the sympathetic nervous system induced by indapamide;
the stimulation of the RAS caused by indapamide is blocked by perindopril;
the potassium depletion linked to indapamide is compensated by the potassium-sparing effect of perindopril.
A perindopril 4 mg/ indapamide 1.25 mg combination does not adversely affect lipid metabolism (total cholesterol, HDL and LDL cholesterol, triglycerides) or carbohydrate metabolism, even in hypertensive patients with diabetes.
Pharmacology of perindopril.
Perindopril (pro-drug), following hydrolysis to perindoprilat, inhibits ACE both in vitro and in vivo. It is thought that ACE inhibitors reduce blood pressure by inhibiting the enzyme which catalyses the conversion of angiotensin I to angiotensin II. Decreased plasma angiotensin II leads to increased plasma renin activity and a decrease in aldosterone. In addition to its effects on circulating ACE, perindopril binds to and inhibits tissue converting enzyme, predominantly in the kidney and vascular wall. The contribution of this mechanism to the overall antihypertensive effect of perindopril is unknown. Animal studies have demonstrated reversal of vascular hypertrophy and an improvement in the ratio of elastin to collagen in the vessel wall. Studies in humans have demonstrated an improvement in the visco-elastic properties of large vessels and in compliance. Studies in animals and humans suggest that specific and competitive suppression of the renin-angiotensin-aldosterone system (RAAS) is the main mechanism by which blood pressure is reduced. However, antihypertensive activity has also been observed in patients with low renin activity. Perindopril may also inhibit the degradation of the potent vasodepressor peptide, bradykinin, and this action may contribute to its antihypertensive action. Perindopril appears to reduce peripheral resistance and may influence arterial compliance.
Studies carried out in animal models of hypertension have shown that perindopril is a specific competitive angiotensin I converting enzyme inhibitor. The administration of perindopril to patients with essential hypertension results in a reduction in supine and standing blood pressure without any significant effect on heart rate. Abrupt withdrawal of perindopril has not been associated with a rebound rise in blood pressure. Single dose studies have demonstrated that peak initiation of ACE activity and peak reduction in blood pressure occurs four to six hours after administration of perindopril. The duration of these effects is dose related and, at the recommended dose range, both effects have been shown to be maintained over a 24 hour period.
In haemodynamic studies carried out in animal models of hypertension, blood pressure reduction after perindopril administration was accompanied by a reduction in peripheral arterial resistance and improved arterial wall compliance. In studies carried out in patients with essential hypertension the reduction in blood pressure was accompanied by a reduction in peripheral resistance with no change, or a small increase in renal blood flow and no change in glomerular filtration rate. An increase in the compliance of large arteries was also observed.
When perindopril is administered together with a thiazide-type diuretic, the antihypertensive activity of perindopril may be potentiated in some patients, and this effect is evident after four weeks of treatment. Perindopril like other ACE inhibitors may compensate thiazide induced hypokalaemia.
Pharmacology of indapamide.
Indapamide is an oral antihypertensive medicine. The mechanism whereby indapamide exerts its antihypertensive action has not been completely elucidated; both vascular and renal actions have been implicated.
The renal effects of indapamide are minimal and the antihypertensive effect of indapamide has been attributed to a reduction in vascular reactivity to pressor amines. The finding that indapamide retains its antihypertensive activity in those who are functionally anephric lends support to this hypothesis.
The renal site of action of indapamide is the proximal segment of the distal tubule. Indapamide appears to have natriuretic properties (sodium and chloride being excreted in equivalent amounts) with less effect on potassium or uric acid excretion. Only at doses greater than 2.5 mg/day, i.e. at doses of indapamide two times greater than the amount present in one Perindopril Indapamide-WGR 4/1.25 tablet, is an appreciable increase in urinary volume observed in humans. No significant changes in plasma sodium levels have been observed in clinical studies.
Indapamide does not adversely affect serum triglycerides, LDL cholesterol, the LDL-HDL cholesterol ratio, or glucose tolerance.
Clinical trials.
Pharmacokinetics of a perindopril 4 mg/ indapamide 1.25 mg combination.
The co-administration of perindopril and indapamide does not change their pharmacokinetic properties by comparison to separate administration.
In a bioequivalence study comparing perindopril 4 mg and indapamide 1.25 mg (as single component capsules) with a fixed dose formulation (perindopril 4 mg/ indapamide 1.25 mg tablet), the pharmacokinetics of each active ingredient were shown to be predominantly unchanged. Bioequivalence was established based on AUC and Cmax. Only the Tmax of indapamide was shorter after administration of a perindopril 4 mg/ indapamide 1.25 mg combination (see Table 3).
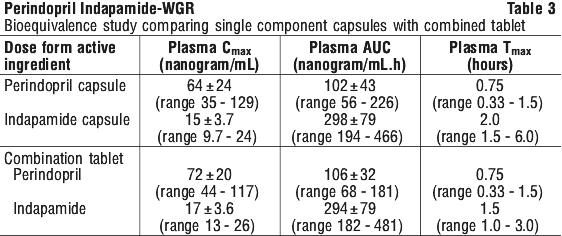
5.2 Pharmacokinetic Properties
Pharmacokinetics of perindopril.
Absorption.
Following oral administration, perindopril is rapidly absorbed and is 61-85% bioavailable. Elimination is rapid, occurring predominantly via the urine. Plasma half-life is approximately 1 hour. Biotransformation of perindopril to the active metabolite perindoprilat is approximately 20%.
Distribution.
Peak plasma concentrations of perindoprilat occur three to four hours after oral administration of perindopril and peak pharmacological activity occurs after 4 to 6 hours. Protein binding of perindoprilat is below 30%. When perindopril is administered chronically, steady-state perindoprilat concentration is reached within 4 days, and perindoprilat does not accumulate.
Metabolism.
Apart from perindoprilat, the administration of perindopril leads to the formation of five other metabolites, all of which are inactive and exist in very low quantities. One of these is the glucurono-conjugate of perindoprilat which is formed by a hepatic first pass effect. This effect does not appear to have any influence on the kinetics of perindoprilat. Food intake may reduce hepatic biotransformation to perindoprilat.
Excretion.
Perindoprilat binds to plasma and tissue ACE, and free perindoprilat is eliminated through the urine. The elimination half-life of the free fraction is between three and five hours. The terminal half-life which corresponds to the dissociation of perindoprilat from ACE, is approximately 25 to 30 hours.
The elimination of perindoprilat is reduced in elderly patients and in patients with cardiac and renal failure (see Section 4.2 Dose and Method of Administration).
Pharmacokinetics of indapamide.
Absorption.
Possibly related to its high lipid solubility, absorption of indapamide from the gastrointestinal tract is rapid (within 0.5 to 1 hour after an oral dose) and complete.
Distribution and metabolism.
Indapamide is widely distributed throughout the body, with extensive binding to some specific sites. In blood, it is highly bound to red blood cells (80%) and, more specifically, to carbonic acid anhydrase (98%) without having any inhibiting activity on this enzyme. In plasma, it is relatively highly bound to plasma proteins (79%). It is also taken up to a significant degree in the vascular compartment, indapamide has a relatively low apparent volume of distribution (approximately 60 L) and 40% of the dose is located in the blood one hour after administration. The decrease in plasma concentrations of unchanged indapamide is biphasic with half-lives between 14 and 25 hours. Both single and multiple dose data indicate that the kinetics of indapamide are linear. Steady state plasma levels are reached within three to four days after starting treatment and indapamide does not accumulate in hypertensive patients with various degrees of renal insufficiency. Indapamide is extensively metabolised in the liver, mainly by CYP2C9 and CYP3A4 isozymes and by cytosolic hydrolysis enzymes. Care should be taken when administering indapamide in combination with medicines that alter the activity of these enzymes (also see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
Excretion.
Following radioactivity studies using carbon-14, the main route of elimination is the urine, but only 5% to 7% of the dose is excreted into the urine as unchanged indapamide; 20% to 23% of total radioactivity is eliminated into the faeces. Renal clearance of indapamide (as unchanged indapamide) is approximately 5 mL/minute, representing less than 10% of systemic clearance. The high lipid solubility of the indoline moiety confers to indapamide its highly localised binding to structures in the cardiovascular system.
5.3 Preclinical Safety Data
Perindopril displays the typical effects of ACE inhibitors. In the rat, the target organ is the kidney; perindopril causes anatomical modifications in arteries which result in intrarenal haemodynamic changes and an increase in blood urea and creatinine levels. The highest doses of indapamide administered by the oral route in different animal species manifested as an exacerbation of the diuretic properties of indapamide. The main symptoms in acute toxicity studies with indapamide administered by the intravenous or intraperitoneal routes are related to the pharmacological action of indapamide, i.e. bradypnoea and peripheral vasodilatation.
In animal models, the combination of perindopril and indapamide has greater toxicity than that of each individual component. Renal manifestations of the effects of perindopril in the rat are increased when it is given in combination with indapamide (about 3 fold relative to the effects of perindopril alone). Renal impairment, resulting from loss of functional nephrons and irreversible renal fibrosis, is observed when perindopril is given in combination with indapamide in the rat. The combination of perindopril and indapamide produced gastrointestinal toxicity (haemorrhage, erosion and necrosis) in dogs, but similar effects were not observed in dogs with much higher doses of the individual components. The mechanism underlying perindopril/indapamide-induced gastrointestinal toxicity in dogs is unknown and the clinical relevance of this finding is questionable. In addition, the toxic effects of perindopril/indapamide in pregnant rats and rabbits are increased when compared to the effects of the individual components.
Genotoxicity.
No genotoxicity studies of perindopril in combination with indapamide have been conducted.
Related to perindopril component.
Results from a broad set of assays for gene mutation and chromosomal damage with perindopril arginine suggest no genotoxic potential at clinical doses. Perindopril showed no evidence of genotoxicity potential in assays for gene mutation (Ames reverse mutation test, mouse lymphoma thymidine kinase assay), chromosomal damage (mouse micronucleus test, Chinese hamster bone marrow cells in vivo, human lymphocytes in vitro) and other genotoxic effects (gene conversion assay in Saccharomyces cerevisiae, unscheduled DNA synthesis in rat hepatic cells).
Related to indapamide component.
Indapamide was negative in mutagenicity tests in bacteria and in a bone marrow micronucleus test in mice.
In animals treated with oral doses of indapamide, a reduction in the number of implantation sites was seen at 25 mg/kg/day and decreases were seen in weight gain of the F1 generation from rats treated at doses ≥ 2.5 mg/kg/day. Galactopoiesis was reduced in the F1 generation from rats treated orally at 0.5 mg/kg/day and this led to increased mortality of the F2 generation during the first 48 hours of life. No embryotoxicity or teratogenic potential was seen in rats (up to 150 mg/kg/day) or rabbits (up to 180 mg/kg/day).
Carcinogenicity.
No carcinogenicity studies of perindopril in combination with indapamide have been conducted.
In studies of perindopril erbumine and indapamide hemihydrate no evidence of carcinogenic activity was observed in mice and rats when indapamide was administered via the diet at levels up to 100 mg/kg/day, or when perindopril erbumine was administered via drinking water at levels up to 7.5 mg/kg/day for two years.
Related to perindopril component.
At least one ACE inhibitor has caused an increase in the incidence of oxyphilic renal tubular cells and oncocytomas in rats. The potential of the ACE inhibitor class to cause this effect in humans is unknown. Moreover, the progression of oxyphilic cells to oncocytomas is rare in humans and when it does occur, it is considered as benign.
Related to indapamide component.
Indapamide was negative in mutagenicity tests in bacteria and in a bone marrow micronucleus test in mice.6 Pharmaceutical Particulars
6.1 List of Excipients
Lactose, magnesium stearate.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 25°C. Store in original package.
6.5 Nature and Contents of Container
Perindopril Indapamide-WGR 4/1.25 tablets.
Blister pack (Aluminium/Aluminium silver foil) of 30 tablets. (AUST R 127123).
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
Perindopril erbumine.
Perindopril erbumine is a dipeptide monoacid monoester with a perhydroindole group and no sulfhydryl radical. It is a white powder, readily soluble in purified water, ethanol and chloroform. Perindopril has five asymmetric centres and is synthesised stereoselectively so that it is a single enantiomer (all S stereochemistry).
Chemical structure.
 Chemical name: 2-methylpropan-2-amine (2S,3aS,7aS)-1-[(2S)-2-[[(1S)-1- (ethoxycarbonyl)butyl]amino]propanoyl]octahydro-1H-indole-2-carboxylate.
Chemical name: 2-methylpropan-2-amine (2S,3aS,7aS)-1-[(2S)-2-[[(1S)-1- (ethoxycarbonyl)butyl]amino]propanoyl]octahydro-1H-indole-2-carboxylate.
Molecular formula: C19H32N2O5, C4H11N.
Molecular weight: 441.6.
CAS number.
107133-36-8.
Indapamide hemihydrate.
Indapamide hemihydrate is a nonthiazide indole derivative of chlorosulfonamide. It is a white or almost white powder, practically insoluble in water, soluble in alcohol, slightly soluble in ether.
Chemical structure.
 Chemical name: 4-chloro-N-[(2RS)-2-methyl-2,3-dihydro-1H-indol-1-yl]-3-sulfamoylbenzamide.
Chemical name: 4-chloro-N-[(2RS)-2-methyl-2,3-dihydro-1H-indol-1-yl]-3-sulfamoylbenzamide.
Molecular formula: C16H16ClN3O3S, ½ H2O.
Molecular weight: 374.8.
CAS number.
26807-65-8.7 Medicine Schedule (Poisons Standard)
S4 - Prescription Only Medicine.
Summary Table of Changes







 Chemical name: 2-methylpropan-2-amine (2S,3aS,7aS)-1-[(2S)-2-[[(1S)-1- (ethoxycarbonyl)butyl]amino]propanoyl]octahydro-1H-indole-2-carboxylate.
Chemical name: 2-methylpropan-2-amine (2S,3aS,7aS)-1-[(2S)-2-[[(1S)-1- (ethoxycarbonyl)butyl]amino]propanoyl]octahydro-1H-indole-2-carboxylate. Chemical name: 4-chloro-N-[(2RS)-2-methyl-2,3-dihydro-1H-indol-1-yl]-3-sulfamoylbenzamide.
Chemical name: 4-chloro-N-[(2RS)-2-methyl-2,3-dihydro-1H-indol-1-yl]-3-sulfamoylbenzamide.