What is in this leaflet
Please read this leaflet carefully before taking PHARMACOR LINEZOLID. This leaflet answers some common questions about PHARMACOR LINEZOLID. It does not contain all the available information. It does not take the place of talking to your or your child's doctor or pharmacist.
All medicines have risks and benefits. Your doctor has weighed the risks of you or your child taking PHARMACOR LINEZOLID against the expected benefits it will have.
Ask the doctor if you have any concerns about you or your child taking this medicine.
Keep this leaflet even after your treatment with PHARMACOR LINEZOLID is finished. You may need to read it again.
What PHARMACOR LINEZOLID is used for
PHARMACOR LINEZOLID contains the active ingredient, linezolid.
PHARMACOR LINEZOLID is an antibiotic (an agent used to destroy certain types of bacteria). It is used in the treatment of bacterial infections such as pneumonia, skin infections or blood infections.
Depending on the type of bacteria, you may be given additional medicines.
Your doctor, however, may prescribe PHARMACOR LINEZOLID for another purpose.
Ask your doctor if you have any questions about why PHARMACOR LINEZOLID has been prescribed for you or your child.
This medicine is not addictive.
Before treatment with PHARMACOR LINEZOLID
When PHARMACOR LINEZOLID must not be Used
PHARMACOR LINEZOLID must not be given if you or your child:
- are allergic to linezolid or any of the other ingredients listed at the end of this leaflet.
Some of the symptoms of an allergic reaction may include: shortness of breath, wheezing or difficulty breathing, swelling of the face, lips, tongue or other parts of the body, rash, itching or hives on the skin. - have uncontrolled high blood pressure
- have pheochromocytoma (a type of tumour of the adrenal gland)
- have thyrotoxicosis (an overactive thyroid gland)
- have flushing or other symptoms caused by a carcinoid tumour
- are taking or have taken in the last two weeks any medicine that is a monoamine oxidase inhibitor (e.g., moclobemide, phenelzine or tranylcypromine to treat depression or selegiline to treat Parkinson's disease)
- any cold or flu medicine containing pseudoephedrine
- adrenaline, a medicine used to treat severe allergic reactions
- any other medicine that increases blood pressure (e.g., noradrenaline, dopamine, dobutamine)
- are taking any medicine that is an SSRI or serotonin re-uptake inhibitor, which are types of medicine to treat depression, anxiety, panic attacks, obsessivecompulsive disorders or obesity (e.g., citalopram, escitalopram, fluoxetine, fluvoxamine, paroxetine, sertraline, duloxetine, sibutramine, venlafaxine)
- tricyclic antidepressants, which are medicines to treat depression (e.g., amitriptyline, clomipramine, dothiepin, doxepin, imipramine, nortriptyline, trimipramine)
- some medicines to treat migraine (e.g.naratriptan, sumatriptan, zolmitriptan)
- pethidine, a medicine to treat pain.
- buspirone, a medicine to treat anxiety
Do not use PHARMACOR LINEZOLID if the packaging is torn or shows signs of tampering.
Do not use PHARMACOR LINEZOLID tablets after the expiry date printed on the pack has passed.
Before treatment with PHARMACOR LINEZOLID
Tell the doctor if you or your child:
- have diarrhoea
- have allergies to any other medicines or substances, such as foods, preservatives or dyes
- have phenylketonuria
- are anaemic or have had any abnormal blood test results (e.g., low haemoglobin or platelets).
- are diabetic
Tell the doctor if you:
- are pregnant or planning to become pregnant.
- are breastfeeding or intend to breastfeed
If you are pregnant or breastfeeding, the doctor may decide not to prescribe PHARMACOR LINEZOLID.
Taking other medicines
Tell your doctor if you are taking any other medicines, including medicines that you buy without a prescription from a pharmacy, supermarket or health food shop.
In particular, tell the doctor if you or your child have been treated with or are taking:
- any medicine that inhibits monoamine oxidase, (e.g., moclobemide, phenelzine or tranylcypromine to treat depression or selegiline to treat Parkinson's disease)
- any cold or flu medicine containing pseudoephedrine
- adrenaline , a medicine used to treat severe allergic reactions
- any other medicine that increases blood pressure (e.g., noradrenaline , dopamine, dobutamine)
- are taking any medicine that is an SSRI or serotonin re-uptake inhibitor, which are types of medicine to treat depression, anxiety, panic attacks, obsessivecompulsive disorders or obesity (e.g., citalopram, escitalopram, fluoxetine, fluvoxamine, paroxetine, sertraline, duloxetine, sibutramine, venlafaxine)
- tricyclic antidepressants, which are medicines to treat depression (e.g., amitriptyline, clomipramine, dothiepin, doxepin, imipramine, nortriptyline, trimipramine)
- some medicines to treat migraine (e.g., naratriptan, sumatriptan, zolmitriptan)
- any medicine that is an opioid, a medicine to treat pain.
- buspirone, a medicine to treat anxiety
- rifampicin, a medicine to treat tuberculosis and some other infections
- any medicine that could reduce the levels of haemoglobin (the pigment in red blood cells which carries oxygen) or platelets (blood cells which help blood to clot)
Ask the doctor or pharmacist if you are not sure about this list of medicines.
Also tell the doctor if your or your child's diet contains a lot of mature cheese, yeast extracts, meat extracts, soya bean extracts (e.g., soy sauce), draught beers or wine. PHARMACOR LINEZOLID may react with a substance which is naturally present in these foods.
How PHARMACOR LINEZOLID is given
It is recommended that treatment with PHARMACOR LINEZOLID begin in a hospital.
Follow all directions given by your or your child's doctor carefully. These directions may differ from the information contained in this leaflet.
If you do not understand the instructions on the box, ask the doctor or pharmacist for help.
When to take it
PHARMACOR LINEZOLID can be taken before, during or after meals.
If you or your child are on dialysis, take PHARMACOR LINEZOLID after dialysis.
How much is given
Tablets
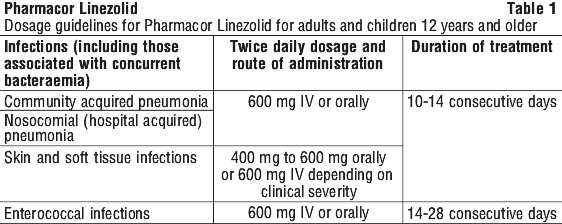
The recommended dose for adults and adolescents 12 years and older is one 600mg tablet twice daily (every 12 hours).
Continue taking PHARMACOR LINEZOLID until you or your child finish the tablets unless your doctor recommends otherwise.
Do not stop taking PHARMACOR LINEZOLID unless your doctor tells you to, even if you feel better.
Do not stop giving PHARMACOR LINEZOLID to your child unless your child's doctor tells you to, even if your child feels better. If you or your child do not complete the full course prescribed by the doctor, the bacteria causing the infection may continue to grow and multiply. The infection may not clear completely or it may return.
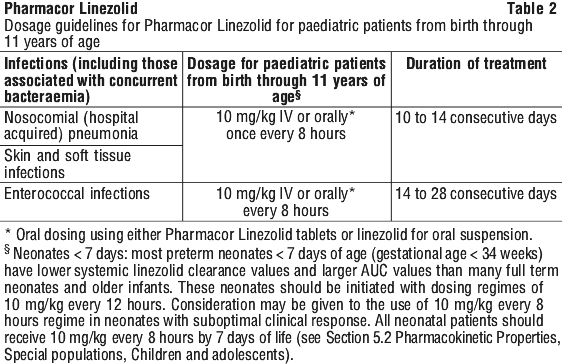
A course of treatment usually lasts 10 to 14 days, but may be up to 28 days.
Ask the doctor if you want more information about the dose of PHARMACOR LINEZOLID and how it is given.
In case of overdose
Immediately telephone your doctor or pharmacist or Poisons Information Centre on 13 11 26 or go to Accident and Emergency at your nearest hospital, if you think you or anyone else may have been given too much PHARMACOR LINEZOLID.
Do this even if there are no signs of discomfort or poisoning. You or your child may need urgent medical attention.
Keep the telephone numbers for these places handy.
The possible effects of overdose are vomiting, tremors (shaking), unsteadiness or lack of coordination.
While being treated With PHARMACOR LINEZOLID
Things you must do
Follow all instructions given by the doctor. In some cases, additional blood tests may be required.
If the symptoms of the infection do not improve within a few days, or if they become worse, tell your or your child's doctor.
As part of the treatment, you or your child may be given other medicines including other antibiotics. It is important to keep taking these medicines as well as PHARMACOR LINEZOLID unless you are told otherwise by your doctor or pharmacist.
It is important to tell your or your child's doctor if you or your child develop diarrhoea during or after treatment with PHARMACOR LINEZOLID. Do this even if it occurs several weeks after PHARMACOR LINEZOLID has been stopped.
Do not take the medicine to treat diarrhea without first checking with the doctor. Diarrhoea may be caused by a serious condition affecting the bowel.
You or your child may need urgent medical care.
If you or your child get a sore white mouth or tongue during or soon treatment with PHARMACOR LINEZOLID tell the doctor.
Tell the doctor if you or your child get vaginal itching or discharge. This may mean you or your child have a fungal infection called thrush. Sometimes the use of PHARMACOR LINEZOLID allows fungi to grow which causes the symptoms described above. PHARMACOR LINEZOLID does not work against fungi.
Tell your doctor immediately if you become pregnant while you are being treated with PHARMACOR LINEZOLID.
Tell all doctors, dentists and pharmacists who are treating you or your child that you are taking PHARMACOR LINEZOLID.
Things you must not do
Do not start taking any other medicines, prescription or not, without first telling your doctor or pharmacist.
Do not give your child any other medicines, prescription or purchased from a health food shop, pharmacy or supermarket without first telling your child's doctor or pharmacist.
Do not take any medicine or treat diarrhea without first checking with the doctor. Diarrhoea may be caused by a serious condition affecting the bowel.
You or your child may need urgent medical care.
Avoid eating too much mature cheese, yeast extracts, meat extracts or soya bean extracts (e.g., soy sauce). Avoid drinking alcohol, especially draught beers and wine. This is because PHARMACOR LINEZOLID may react with a substance which is naturally present in these foods.
If you or your child develop a throbbing headache after eating, tell your doctor or health care professional.
Do not give PHARMACOR LINEZOLID to anyone else, even if they seem to have the same condition as you.
Do not take PHARMACOR LINEZOLID to treat other complaint unless your doctor tells you to do so.
Side effects
Tell your or your child's doctor or pharmacist as soon as possible if you or your child do not feel well while you are being treated with PHARMACOR LINEZOLID
This medicine helps most people, but it may have unwanted side effects in a few people. All medicines can have side effects. If they occur, most are likely to be minor and temporary.
However, some may be serious and need medical attention.
Do not be alarmed by the following lists of side effects. You or your child may not experience any of them.
Ask the doctor or pharmacist to answer any questions you may have.
Tell your or your child's doctor or pharmacist if you notice any of the following and they worry you:
- headache
- sore, white mouth or tongue (oral thrush)
- vaginal itching or discharge (vaginal thrush)
- pain, cramping or "bloating" of the abdomen
- nausea or vomiting
- metallic taste
- change in the colour of the tongue
- change in the colour of teeth. This may be reversible.
Tell your doctor immediately and before you or your child are given the next dose of PHARMACOR LINEZOLID if you notice any of the following:
- skin reactions (hives, rash or itching)
- visual disturbances or numbness or weakness of the arms and legs (rare side effects that have been primarily reported in patients treated for longer than 28 days)
- tiredness, headaches, being short of breath when exercising, dizziness, looking pale, dark circles around the eyes, fever and chills, sore throat or bruising (these symptoms may indicate a decrease in the level of your blood cells)
- sweating, feeling drunk and dizzy, muscle twitching, fever and shivering, confusion.
If any of the following happen, tell your or you child’s doctor immediately or go to Accident or Emergency at your nearest hospital.
- seizure
- shortness of breath
- wheezing or difficulty breathing
- swelling of the face, lips, tongue or other parts of the body
- rash, severe itching or hives or blisters on the skin and bleeding in the lips, eyes, mouth, nose and genitals
- widespread painful red areas, followed by blisters and ends with peeling of layers of skin. This may be accompanied by fevers, chills, aching muscles and generally feeling unwell.
These may be signs of a serious allergic reaction or side effect. You or your child may need urgent medical attention or hospitalisation. These side effects are rare.
After stopping your treatment
Tell your or your child's doctor or pharmacist if you notice any of the following side effects, particularly if they occur several weeks after stopping treatment with PHARMACOR LINEZOLID:
- severe stomach cramps
- watery and severe diarrhea (which may be bloody), fever, in combination with one or both of the above.
PHARMACOR LINEZOLID can cause some bacteria, which are normally present in the bowel and normally harmless, to multiply and therefore cause the above symptoms. You may need urgent medical attention.
Tell your or your child’s doctor if you notice any other side effects.
Other side effects not listed above may also occur in some people.
After using PHARMACOR LINEZOLID
Storage
Keep your tablets in the original packaging, including outer carton, until it is time to take them. If you take the medicine out of the pack it may not keep well.
Keep PHARMACOR LINEZOLID in a cool dry place where the temperature stays below 25°C.
Do not store PHARMACOR LINEZOLID or any other medicines in a bathroom or near a sink.
Do not leave it in the car or on window sills. Heat and dampness can destroy some medicines.
Keep PHARMACOR LINEZOLID where children cannot reach it. A locked cupboard at least one and a- half meters above the ground is a good place to store medicines.
Product Description
What PHARMACOR LINEZOLID looks like
PHARMACOR LINEZOLID tablets are off white capsule shape film coated tablet debossed with "LIN 600" on one side, plain on other side. The tablets are available in blister packs; 10’s and 30’s and HDPE bottle pack of 20’s and 100’s.
Linezolid solution for injection and granules for oral suspension are unavailable in the PHARMACOR LINEZOLID brand. For intravenous and oral suspension dosing requirements other brands are available. Information obtained using linezolid solution for injection and granules for oral suspension formulations have been retained throughout the CMI, where appropriate, for continuity and prescriber information.
Ingredients
The active ingredient in PHARMACOR LINEZOLID is linezolid.
Tablets
PHARMACOR LINEZOLID tablets contain 600mg of linezolid.
The tablets also contain:
- Pre-gelatinised starch (lycatab c),
- Mannitol (pearlitol 160c),
- Colloidal silicon dioxide,
- Copovidone (plasdone s 630),
- Sodium stearyl fumarate
The film coating contains:
- Opadry White YS-1-18202-A
- Water
Supplier
PHARMACOR LINEZOLID is supplied in Australia by:
Pharmacor Pty Ltd.
Chatswood, NSW, 2067
Australia
www.pharmacor.com.au
Australian Registration Numbers
PHARMACOR LINEZOLID can be identified by an Australian Register Number which is found on the carton.
Tablets
296543 (blister pack)
296542 (bottle pack)
Date of preparation
This leaflet was revised in 09/2021.
Published by MIMS November 2021

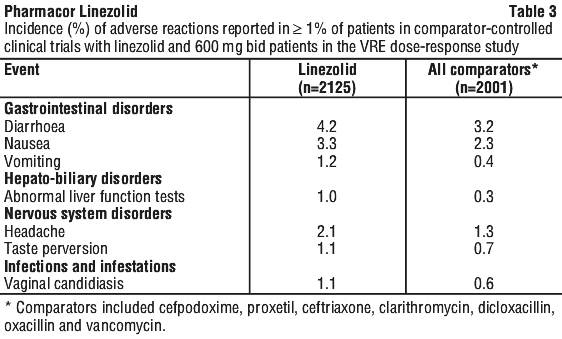 Changes observed in laboratory parameters (without regard to drug relationship) generally reflected resolution of the infection, were not clinically significant, did not lead to discontinuation of therapy and were reversible. The incidence of patients with at least one substantially abnormal haematologic or serum chemistry value is presented in Table 4.
Changes observed in laboratory parameters (without regard to drug relationship) generally reflected resolution of the infection, were not clinically significant, did not lead to discontinuation of therapy and were reversible. The incidence of patients with at least one substantially abnormal haematologic or serum chemistry value is presented in Table 4.
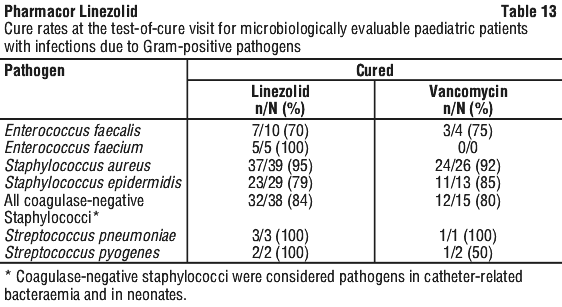
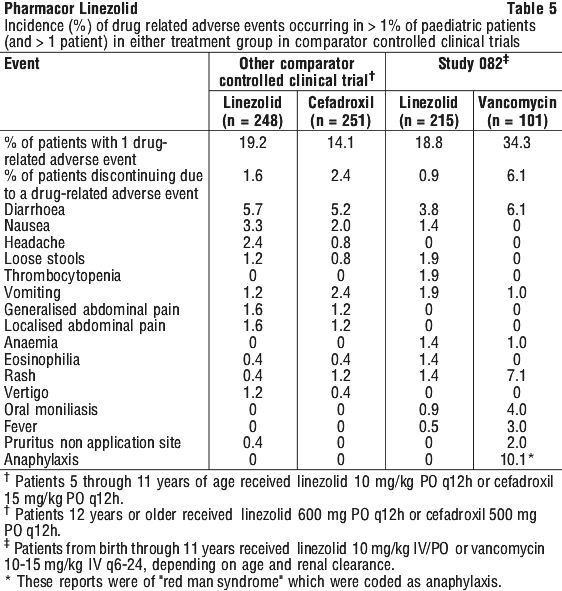 In a study of severely ill, hospitalised paediatric patients ranging in age from birth through 11 years, the percentage of patients who developed a substantially low platelet count was 12.9% with linezolid and 13.4% with vancomycin. In an outpatient study of paediatric patients aged from 5 through 17 years, the percentage of patients who developed a substantially low platelet count was 0% with linezolid and 0.4% with cefadroxil. Other changes observed in laboratory parameters, were not clinically significant, did not lead to discontinuation of therapy and were reversible. The incidence of paediatric patients with at least one substantially abnormal haematologic or serum chemistry value is presented in Table 6.
In a study of severely ill, hospitalised paediatric patients ranging in age from birth through 11 years, the percentage of patients who developed a substantially low platelet count was 12.9% with linezolid and 13.4% with vancomycin. In an outpatient study of paediatric patients aged from 5 through 17 years, the percentage of patients who developed a substantially low platelet count was 0% with linezolid and 0.4% with cefadroxil. Other changes observed in laboratory parameters, were not clinically significant, did not lead to discontinuation of therapy and were reversible. The incidence of paediatric patients with at least one substantially abnormal haematologic or serum chemistry value is presented in Table 6.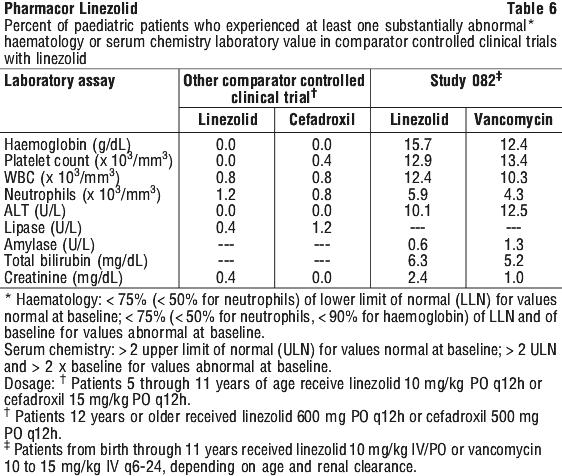
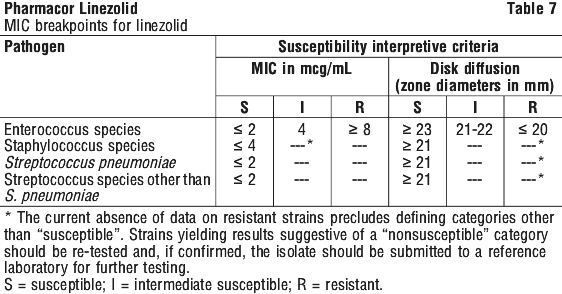 The studies used to define the above breakpoints employed standard NCCLS (National Committee for Clinical Laboratory Standards) microdilution and agar diffusion methods.
The studies used to define the above breakpoints employed standard NCCLS (National Committee for Clinical Laboratory Standards) microdilution and agar diffusion methods.
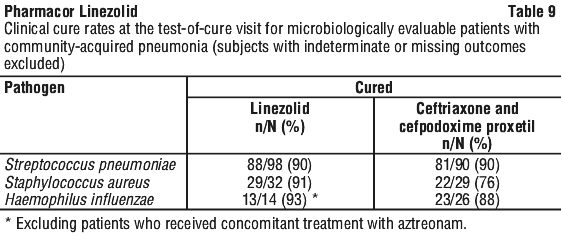
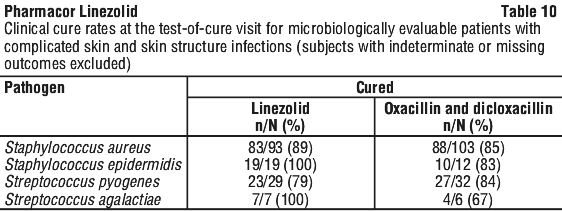
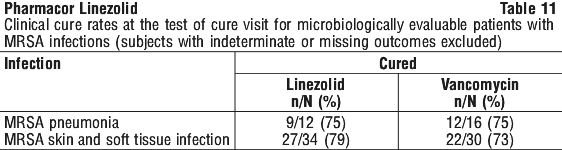
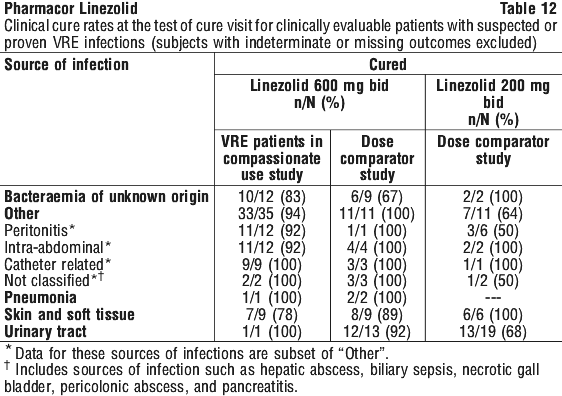
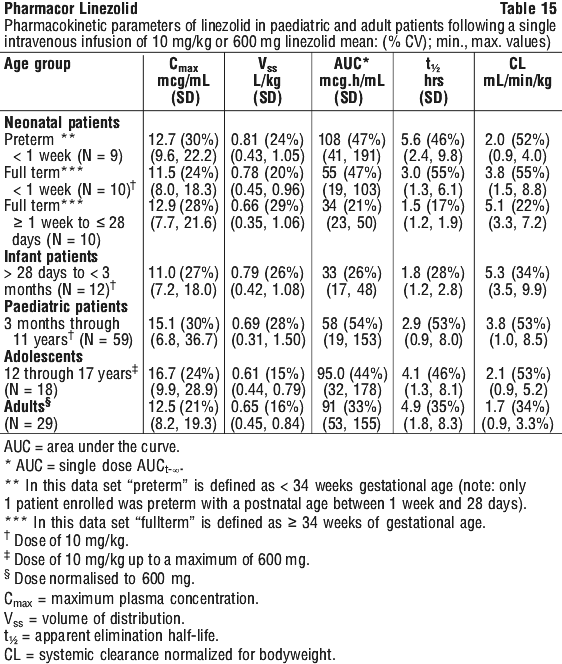
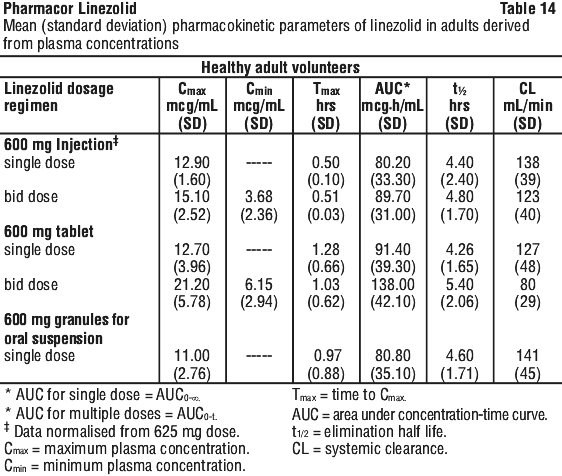 As can be seen from Table 14, average Cmin values achieved in plasma using the 600 mg bid dosage regimen approximate to the highest MIC90 (4 microgram/mL) for the least susceptible microorganisms.
As can be seen from Table 14, average Cmin values achieved in plasma using the 600 mg bid dosage regimen approximate to the highest MIC90 (4 microgram/mL) for the least susceptible microorganisms.