What is in this leaflet
This leaflet answers some common questions about Physiotens.
It does not contain all the available information. It does not take the place of talking to your doctor or pharmacist.
All medicines have risks and benefits. Your doctor has weighed the risks of you taking Physiotens against the benefits they expect it will have for you.
If you have any concerns about taking this medicine, ask your doctor or pharmacist.
Keep this leaflet with the medication. You may want to read it again.
What Physiotens is used for
Physiotens lowers high blood pressure, which is sometimes called hypertension.
Everyone has blood pressure. This pressure helps to move your blood around your body. Your blood pressure may change at different times of the day. You have hypertension when your blood pressure stays higher than normal, even when you are calm or relaxed.
There are usually no symptoms of hypertension. The only way of knowing if you have hypertension is to have your blood pressure checked regularly. If high blood pressure is not treated it can lead to serious health problems, including stroke, heart disease and kidney failure.
Ask your doctor if you have any questions about why it has been prescribed for you. Your doctor may have prescribed Physiotens for another use.
This medicine is only available with a doctor's prescription.
This medicine is not addictive.
Before you take Physiotens
When you must not take it
Do not take Physiotens if you have an allergy to:
- any medicine containing moxonidine
- any of the ingredients listed at the end of this leaflet
Some symptoms of an allergic reaction include skin rash, itching, shortness of breath or swelling of the face, lips or tongue, which may cause difficulty breathing.
Do not take this medicine if you are pregnant or think you may be pregnant.
Do not breastfeed if you are taking this medicine. Physiotens passes into breast milk. Your doctor will discuss the possible risks and benefits of taking Physiotens when breastfeeding.
Do not give Physiotens to a child under the age of 16 years. Safety and effectiveness in children younger than 16 years have not been established.
Do not take Physiotens if you:
- are aged 75 years or more
- have heart problems such as heart failure or abnormal rhythm
- have severe kidney disease
Do not take it after the expiry date (EXP) printed on the pack or if the packaging is torn or shows signs of tampering. If it has expired or is damaged return it to your pharmacist for disposal.
If you are not sure whether you should start taking this medicine, talk to your doctor.
Before you start to take it
Tell your doctor if you have any allergies to any other medicines, foods, preservatives or dyes.
Tell your doctor if you have, or have had, any of the following medical conditions:
- kidney problems
- heart problems
- angio-oedema, which is unusual swelling of the face, lips or tongue
- leg pains or cramps caused by poor blood circulation
- Raynaud's disease, where your fingers go pale and blue and are painful in the cold
- Parkinson's disease, a disease of the nerves which causes uncontrolled shaking and stiffness
- epilepsy
- glaucoma, a disease of increased pressure in the eye
- depression
Tell your doctor or pharmacist if you are pregnant, intend to become pregnant or are breastfeeding. Your doctor or pharmacist can discuss the risks and benefits involved.
If you have not told your doctor about any of the above, tell them before you take Physiotens.
Taking other medicines
Tell your doctor if you are already taking medicines to lower your blood pressure. These medicines can have a combined effect when used with Physiotens to reduce blood pressure and your doctor may need to adjust the dose you have to take.
Tell your doctor if you are taking any other medicines, including any that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may affect the way other medicines work. These include:
- some medicines used to treat depression called "tricyclic" medicines (such as imipramine and amitriptyline)
- sleeping tablets or other medicines which make you feel drowsy
These medicines may be affected by Physiotens, or may affect how well it works. You may need to use different amounts of your medicine, or take different medicines.
Your doctor or pharmacist has more information on medicines to be careful with or to avoid while taking Physiotens.
How to take Physiotens
Follow all directions given to you by your doctor or pharmacist carefully. They may differ from the information contained in this leaflet.
If you do not understand the instructions on the box, ask your doctor or pharmacist for help.
How much to take
Your doctor or pharmacist will tell you how many tablets to take each day.
The usual starting dose of Physiotens is one 0.2 mg (200 microgram) tablet once per day.
Depending on how your blood pressure responds your dosage may be increased by your doctor to 0.4 mg (400 micrograms) after 2 weeks. After a further 2 weeks your doctor may increase your dosage to 0.6 mg per day (600 micrograms). You should not take 0.6 mg as one dose, it should be taken as a divided dose.
Ask your doctor how to divide your daily dose.
How to take it
Swallow the tablets whole with a full glass of water or another liquid.
Do not chew the tablets.
When to take it
Take Physiotens at about the same time each day. Taking it at the same time each day will have the best effect. It will also help you remember when to take it.
It does not matter if you take this medicine with or without food.
How long to take it
Continue taking your medicine for as long as your doctor tells you.
The medicine helps control your condition, but it does not cure it. It is important to keep taking your medicine even if you feel well.
If you forget to take it
If it is almost time for your next dose, skip the dose you missed and take your next dose when you are meant to.
If there is still a long time to go before your next dose, take it as soon as you remember, and then go back to taking it as you would normally.
Do not take two doses within 6 hours of each other. Do not take a double dose to make up for the dose that you missed. This may increase the chance of getting an unwanted side effect.
If you have trouble remembering when to take your medicine, ask your pharmacist for advice.
If you take too much (overdose)
Immediately telephone your doctor, or the Poisons Information Centre (13 11 26), or go to Accident and Emergency at your nearest hospital, if you think you or anyone else may have taken too much Physiotens. Do this even if there are no signs of discomfort or poisoning. You may need urgent medical attention.
If possible, show the doctor the pack of tablets.
While you are using Physiotens
Things you must do
If you are about to be started on any new medicine, remind your doctor and pharmacist that you are taking Physiotens.
Tell any other doctors, dentists and pharmacists who treat you that you are taking this medicine.
If you are going to have surgery, tell the surgeon that you are taking this medicine.
If you become pregnant while you are taking this medicine, tell your doctor or pharmacist immediately.
If you are about to have any blood tests, tell your doctor that you are taking this medicine.
Keep all of your doctor's appointments so that your progress can be checked.
Make sure you drink enough water during exercise and hot weather when you are taking Physiotens, especially if you sweat a lot. If you do not drink enough water while taking Physiotens, you may faint or feel light-headed or sick. This is because your body does not have enough fluid and your blood pressure is too low.
Tell your doctor if you have excessive vomiting and/or diarrhoea while taking Physiotens. This can also mean that you are losing too much water and your blood pressure may become too low.
Things you must not do
Do not use this medicine to treat any other complaints unless your doctor or pharmacist tells you to.
Do not give this medicine to anyone else, even if they have the same condition as you.
Do not stop taking Physiotens abruptly, or change the dosage, without checking with your doctor.
Things to be careful of
Be careful driving or operating machinery until you know how Physiotens affects you. Physiotens may cause drowsiness, dizziness or light-headedness in some people. If you have any of these symptoms, do not drive, operate machinery or do anything else that could be dangerous.
You should avoid drinking alcohol when taking Physiotens. The effect of taking alcohol with Physiotens has not been studied. Ask your doctor for advice.
If you feel light-headed, dizzy or faint when getting out of bed or standing up, get up slowly. Standing up slowly, especially when you get up from bed or chairs, will help your body get used to the change in position and blood pressure. If this problem continues or gets worse, talk to your doctor.
Side effects
Tell your doctor or pharmacist as soon as possible if you do not feel well while you are taking Physiotens.
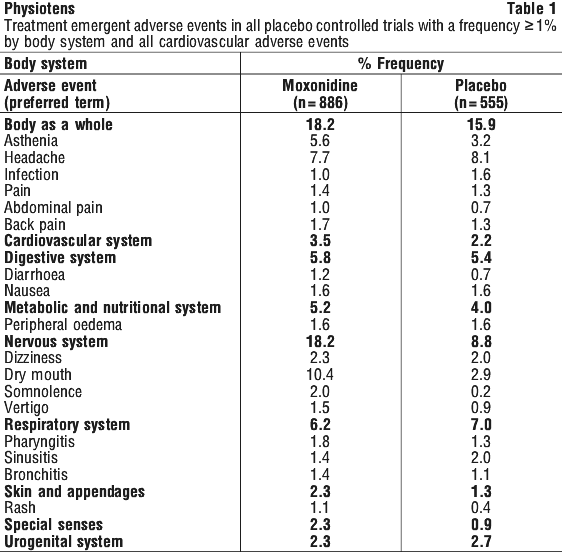
It helps most people with high blood pressure, but it may have unwanted side effects in a few people. All medicines have some unwanted side effects. Sometimes they are serious, but most of the time they are not. You may need medical attention if you get some of the side-effects.
Do not be alarmed by this list of possible side effects. You may not experience any of them.
Ask your doctor or pharmacist to answer any questions you may have.
Tell your doctor if you notice any of the following and they worry you:
- dry mouth
- headache
- lack of energy
- dizziness
- drowsiness
- feeling sick, nausea
- problems sleeping
- skin flushing
- anxiety
The above list includes the more common side effects of your medicine. They are usually mild and short-lived.
Tell your doctor as soon as possible if you notice any of the following:
- severe allergic skin reactions (rash, itching, inflamed or reddened skin)
- swelling of the limbs
The above list includes serious side effects that may require medical attention. Serious side effects are rare.
If any of the following happen, tell your doctor immediately or go to Accident and Emergency at your nearest hospital:
- angio-oedema (unusual swelling of the face, eyes, lips, inside the nose, mouth or throat)
- shortness of breath, breathing or swallowing difficulties.
The above list includes very serious side effects. You may need urgent medical attention. These side effects are rare.
Tell your doctor if you notice anything else that is making you feel unwell. Other side effects not listed above may occur in some consumers.
After using Physiotens
Storage
Keep your tablets in the original pack until it is time to take them. If you take the tablets out of the box they may not keep well.
Keep the medicine in a cool dry place where the temperature stays below 25°C.
Do not store it in the bathroom, near the sink, or on a window sill. Do not leave it in the car. Heat and damp can destroy some medicines.
Keep it where children cannot reach it. A locked cupboard at least one-and-a-half metres above the ground is a good place to store medicines.
Disposal
If your doctor tells you to stop taking this medicine, or the medicine has passed its expiry date, ask your pharmacist what to do with any that are left over.
Return any unused medicine to your pharmacist.
Product description
What it looks like
Physiotens 0.2 mg (200 micrograms) tablets - Pale pink, round, biconvex, film-coated tablet, imprinted 0.2 on one side. Packs of 30 tablets.
Physiotens 0.4 mg (400 micrograms) tablets - Dull red, round, biconvex, film-coated tablet, imprinted 0.4 on one side. Packs of 30 tablets.
Ingredients
Physiotens contains either 0.2 mg (200 micrograms) or 0.4 mg (400 micrograms) of moxonidine as the active ingredient.
It also contains:
- lactose monohydrate
- povidone
- crospovidone
- magnesium stearate
- hypromellose
- ethylcellulose
- macrogol 6000
- purified talc
- iron oxide red
- titanium dioxide
Physiotens contains sugars (as lactose).
Physiotens does not contain tartrazine or any other azo dyes.
Supplier
Physiotens is made in France.
Physiotens is supplied in Australia by:
Mylan Health Pty Ltd
Level 1, 30 The Bond
30-34 Hickson Road
Millers Point, NSW 2000
www.mylan.com.au
Phone: 1800 314 527
® Registered Trademark
This leaflet was prepared in January 2021.
Australian Registration Number(s)
0.2 mg tablets: AUST R 114119.
0.4 mg tablets: AUST R 114121
Physiotens_cmi\Jan21/00
Published by MIMS March 2021



 The combination of hydrochlorothiazide and moxonidine was evaluated in a further placebo controlled, double blind and prospectively randomized trial. The study comprised a total of 161 randomized patients: 37 received moxonidine 400 microgram, 40 received hydrochlorothiazide 25 mg, 42 received the combination 400 microgram/25 mg and 41 received placebo, all once daily. Sitting systolic and diastolic blood pressures at trough after eight weeks treatment were statistically significantly reduced for all active treatment arms versus placebo. Furthermore, the difference of the combination for sitting diastolic blood pressure was statistically significant in favour of the combination for both monotherapies. The incidence of treatment emergent adverse events did not differ significantly between groups: 32.5% with moxonidine, 25% with HCT, 30% with the combination and 23% with placebo. See Table 3.
The combination of hydrochlorothiazide and moxonidine was evaluated in a further placebo controlled, double blind and prospectively randomized trial. The study comprised a total of 161 randomized patients: 37 received moxonidine 400 microgram, 40 received hydrochlorothiazide 25 mg, 42 received the combination 400 microgram/25 mg and 41 received placebo, all once daily. Sitting systolic and diastolic blood pressures at trough after eight weeks treatment were statistically significantly reduced for all active treatment arms versus placebo. Furthermore, the difference of the combination for sitting diastolic blood pressure was statistically significant in favour of the combination for both monotherapies. The incidence of treatment emergent adverse events did not differ significantly between groups: 32.5% with moxonidine, 25% with HCT, 30% with the combination and 23% with placebo. See Table 3.

