What is in this leaflet
This leaflet answers some common questions about Postinor-1.
It does not contain all the available information.
It does not take the place of talking to your doctor or pharmacist.
All medicines have risks and benefits. Your doctor or pharmacist will have discussed and considered the risks of you taking Postinor-1 against the benefits they expect it will have for you.
If you have any concerns about taking this medicine, ask your doctor or pharmacist. Your doctor and pharmacist have more information.
Keep this leaflet with the medicine. You may need to read it again.
What Postinor-1 is used for
Postinor-1 is an emergency contraceptive only. Postinor-1 is not intended as a regular method of contraception. It is used to prevent pregnancy when taken within 72 hours (3 days) of unprotected intercourse (sex).
It is estimated that Postinor-1 will prevent 85% of expected pregnancies when you take it within 72 hours of having unprotected sex. It will not prevent a pregnancy every time and is more effective if you take it as soon as possible after unprotected sex.
Postinor-1 is most effective if taken within 72 hours of unprotected sex. But remember that the sooner you take it, the more likely it will prevent pregnancy.
Postinor-1 will not prevent you from catching sexually transmitted diseases. Ask your doctor or pharmacist if you have any questions about why you are taking Postinor-1.
Postinor-1 is thought to work by:
- stopping your ovaries from releasing an egg;
- preventing sperm from fertilising any egg you may have already released.
Postinor-1 can only prevent you becoming pregnant if you take it within 72 hours of unprotected sex. It does not work if you are already pregnant. If another unprotected intercourse takes place after the use of Postinor-1 (also if this is during the same menstrual cycle), the tablet will not work and there is again the risk of pregnancy.
Before you take Postinor-1
Before you start treatment with Postinor-1, your doctor or pharmacist should:
- Check you are not already pregnant if he/she suspects you may already have been pregnant before this occurrence of unprotected intercourse
- Ask you if you are taking any other medicines (see Taking other medicines)
- Ask if you suffer from high blood pressure
- Advise you of the need for careful use of a barrier method of contraception until you have your next period
- Advise you to see your doctor in three weeks' time, especially if you have not had a period by then
- Discuss the need for regular contraception
You should consult your doctor before you take it if:
- You have diabetes mellitus (with kidney, eye or nerve damage, or vascular disease) – Diabetes mellitus is a condition in which the body does not produce enough insulin or else the body tissues are not able to use the insulin present. This leads to hyperglycaemia (too much sugar in the blood)
- You have ischaemic heart disease (heart disease caused by reduced blood flow in the blood vessels of the heart muscle)
- You have suffered a stroke
- You have had breast cancer
- If you are pregnant or think that you may already be pregnant. This medicine will not work if you are already pregnant. If you are already pregnant, Postinor-1 cannot terminate pregnancy. Postinor-1 is not an “abortion pill”.
You may be already pregnant if:
- your period is more than 5 days late, or you have experienced unusual bleeding when your next period is due;
- you have had unprotected sex more than 72 hours ago, and since your last period.
If any of these conditions apply to you it may not be suitable for you to take Postinor-1.
The use of Postinor-1 is not advised if
- you have a disease of your gastrointestinal tract (such as Crohn’s disease) that interferes with the digestion and absorption of your food;
- you have severe liver disease
- you have a history of ectopic pregnancy (where the baby develops somewhere outside the womb)
- you have a history of salpingitis (inflammation of the Fallopian tubes)
A previous ectopic pregnancy or previous infection of the Fallopian tubes increase the risk of a new ectopic pregnancy. - you are vomiting or have severe diarrhoea
Emergency contraception should be taken as soon as possible after unprotected intercourse.
Children and adolescents
Postinor-1 is not recommended for children. The is only limited information available on Postinor-1 when taken by women aged 14-16 years and no information on its use in younger women or children
When you must not take it
Do not take Postinor-1 if:
- You are pregnant
- You have previously had unprotected intercourse more than 72 hours earlier in the same menstrual cycle, as you may already be pregnant
- You are experiencing vaginal bleeding for which the reason is not known
- You have breast cancer
- You are allergic to any of the ingredients of the tablets (See Product Description below)
- The tablets do not look quite right
Do not take Postinor-1 after the expiry date printed on the pack or if the packaging is torn or shows signs of tampering.
Taking other medicines
Tell your doctor or pharmacist if you are taking any other medicines, including those purchased from a supermarket or health food store. Some medicines may prevent Postinor-1 from working properly or Postinor-1 may affect the way other medicines work. Examples of medicines which may interfere with Postinor-1 are:
- Medicines used to treat epilepsy (e.g. barbiturates, primidone, phenytoin, and carbamazepine).
- Medicines used to treat tuberculosis (e.g. rifampicin, rifabutin).
- Medicines used to treat HIV infection (ritonavir, efavirenz).
- Medicines used to treat fungal infections (griseofulvin).
- Herbal remedies containing St John’s Wort (Hypericum perforatum)
Postinor-1 may affect the way other medicines work
- a medicine called cyclosporine (suppresses the immune system).
How often can you use Postinor-1
You should only use Postinor-1 in emergencies and not as a regular method of contraception. If Postinor-1 is used more than once in a menstrual cycle, it is less reliable and it is more likely to upset your menstrual cycle (period).
Postinor-1 does not work as well as regular methods of contraception. Your doctor, practice nurse or family planning clinic can tell you about long-term methods of contraception which are more effective in preventing you from getting pregnant.
Pregnancy, breastfeeding and fertility
If you are pregnant or breastfeeding, think you may be pregnant or are planning to have a baby, ask your doctor or pharmacist for advice before taking any medicine.
Pregnancy
You should not take this medicine if you are already pregnant. If you do become pregnant even after taking this medicine, it is important that you see your doctor. Your doctor may want to check that the pregnancy is not ectopic (where the baby develops somewhere outside the womb). This is especially important if you develop severe abdominal pain after taking Postinor-1 or if you have previously had an ectopic pregnancy, Fallopian tube surgery or pelvic inflammatory disease.
Breastfeeding
The active ingredient of this medicine is excreted into your breast milk. Therefore, it is suggested that you take your tablet immediately after a breastfeeding and avoid nursing at least 3 days following taking this medicine.
Fertility
Postinor-1 increases the possibility of menstruation disturbances which can sometimes lead to an earlier or later ovulation date resulting in a modified fertility date. Although there are no fertility data in the long term, after treatment with Postinor-1 a rapid return to fertility is expected and therefore, regular contraception should be continued or initiated as soon as possible after Postinor-1 use.
How to take Postinor-1
Always use this medicine exactly as described in the leaflet or as your pharmacist or doctor has told you. Check with your doctor or pharmacist if you are not sure.
Take the tablet as soon as possible, preferably within 12 hours, and no later than 72 hours (3 days) after you have had unprotected sex. Do not delay taking the tablet. The tablet works best the sooner you take it after having unprotected sex. It can only prevent you becoming pregnant, if you take it within 72 hours of unprotected sex.
- Postinor-1 can be taken at any time in your menstrual cycle assuming you are not already pregnant or think you may be pregnant. Swallow the tablet whole with water. Do not chew it.
- If you are using one of the medicines that may prevent Postinor-1 from working properly (see section above “Other medicines and Postinor-1”) or if you have used one of these medicines in the past 4 weeks, Postinor-1 may not work as well for you.
- If you are already using a regular method of contraception such as the contraceptive pill, you can continue to take this at your regular times.
If another unprotected intercourse takes place after the use of Postinor-1 (also if this is during the same menstrual cycle), the tablet will not be effective and there is again the risk of pregnancy.
Speak to a healthcare professional if you are concerned about any problems related to taking emergency contraception.
Postinor-1 is not recommended for children. There is only limited information available on Postinor-1 when taken by women aged 14-16 years and no information on its use in younger women or children.
While taking Postinor-1
If you vomit within 2 hours of taking the tablet you should return to your pharmacy, doctor or clinic as the tablet may not be absorbed and you will need to take an additional tablet.
Things you must do
Tell any doctor or pharmacist who is treating you that you have taken Postinor–1.
Things you must not do
Do not give Postinor-1 to anyone else.
Things to be careful of
If Postinor-1 makes you drowsy or dizzy, do not drive a car or operate machinery.
Postinor-1 contains lactose Each Postinor-1 tablet contains 142.5 mg lactose monohydrate.
If you have been told by your doctor that you have an intolerance to some sugars, contact your doctor before taking this product.
Side effects
Tell your doctor or pharmacist as soon as possible if you do not feel well after you take Postinor-1.
All medicines have side effects. Often they are not serious but sometimes they can be. You may need medical treatment if you get some side effects.
Do not be alarmed by this list of possible side effects.
You may not experience any of them.
Ask your doctor or pharmacist to answer any questions you may have.
Common side effects are tiredness, nausea and vomiting. Some patients have also experienced stomach pain, diarrhoea, dizziness, headache, tender breasts, increased vaginal bleeding and skin reactions.
Other side effects not listed above may also occur in some patients. Tell your doctor or pharmacist if you notice anything that is making you feel unwell.
After using Postinor-1
You should see your doctor within 3 weeks of taking Postinor-1 to make sure that Postinor-1 has worked. You may experience spotting or vaginal bleeding earlier than expected.
If you do not get your period, it is more than 5 days late or is unusually light or unusually heavy, you must see your doctor, as you may be pregnant. If Postinor-1 does not work, you could be pregnant. Your doctor will order a pregnancy test. If stomach pain is severe you should see your doctor immediately as on rare occasions a tubal pregnancy could occur.
Postinor-1 is only intended as an emergency measure. If you have not already done so you should discuss with your doctor other methods of long term contraception.
You should not breast feed within three days after taking Postinor-1.
If you take too much (Overdose)
Telephone your doctor, or the Poisons Information Centre (13 11 26 in Australia) if you take too many Postinor-1 tablets, or if a child has taken Postinor-1.
Although there have been no reports of serious harmful effects from taking too many tablets at once, you may feel sick, actually be sick (vomit), or have vaginal bleeding. You should ask your pharmacist, doctor, practice nurse or family planning clinic for advice, especially if you have been sick, as the tablet may not have worked properly.
Product description:
Postinor-1 contains one blister sheet containing one flat white tablet. The tablet contains 1.5 milligrams of levonorgestrel and the following inactive ingredients: potato starch, maize starch, colloidal anhydrous silica, magnesium stearate, talc, lactose.
Storage:
Keep your tablet in the pack until it is time to take it. If you take the tablet out of the pack it will not keep well.
Keep your tablet in a cool, dry place where the temperature stays below 25°C.
Do not store Postinor-1 or any other medicine in the bathroom or near a sink.
Do not leave it in the car on hot days or on window sills.
Heat and dampness can destroy some medicines.
Keep it where children cannot reach it.
A locked cupboard at least 1½ metres above the ground is a good place to store medicines.
The expiry date is printed on the pack and the foil blister. Do not use Postinor-1 after this expiry date.
Where to go for further information
If you require further information you should contact your pharmacist, doctor or clinic.
Additional information is also available at the website: www.emergencycontraception.com.au
Supplier:
The sponsor for Postinor-1 in Australia is:
Gedeon Richter Australia Pty Ltd
Unit 33 & 34
23 Narabang Way
Belrose NSW 2085
AUSTRALIA
The distributor for Postinor-1 in Australia is:
Pharmacor Pty Ltd
Suite 803, Level 8,
Tower A, The Zenith,
821 Pacific Highway,
Chatswood, NSW, 2067
AUSTRALIA
Ph: 1300 138 805
www.pharmacor.com.au
The AUST R Number for Postinor-1 is 149269.
This leaflet was updated in March 2020.
Published by MIMS October 2020


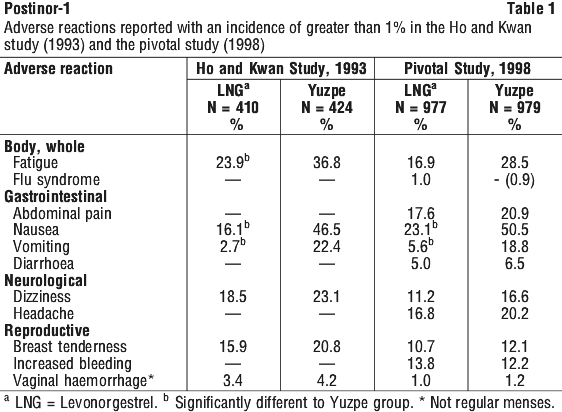 Side effects did not result in any discontinuations in either study. No ectopic pregnancies or congenital abnormalities were reported in either study. However, such a possibility must always be considered, as there have been rare reports of ectopic pregnancies reported during postmarketing surveillance. Cutaneous reactions have also been reported from postmarketing surveillance on rare occasions.
Side effects did not result in any discontinuations in either study. No ectopic pregnancies or congenital abnormalities were reported in either study. However, such a possibility must always be considered, as there have been rare reports of ectopic pregnancies reported during postmarketing surveillance. Cutaneous reactions have also been reported from postmarketing surveillance on rare occasions.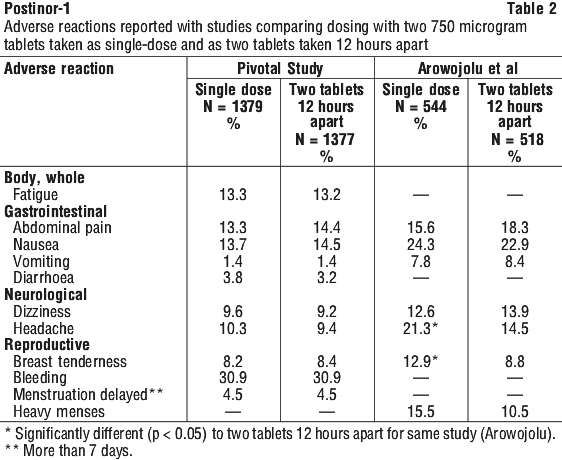 One ectopic pregnancy was observed in the pivotal study and none in the supporting (Arowojolu) study.
One ectopic pregnancy was observed in the pivotal study and none in the supporting (Arowojolu) study.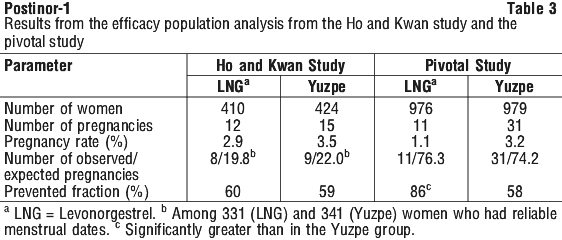 The relative risk of pregnancy in the Pivotal study for the Yuzpe versus levonorgestrel regimens was 2.8 with a lower one-sided 95% confidence bound of 1.53.
The relative risk of pregnancy in the Pivotal study for the Yuzpe versus levonorgestrel regimens was 2.8 with a lower one-sided 95% confidence bound of 1.53.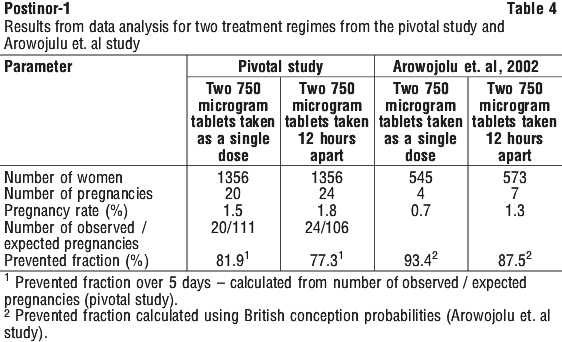 There was no significant difference in efficacy between the two levonorgestrel treatment groups in the pivotal study. Shorter intervals between intercourse and treatment were associated with lower pregnancy rates in both groups.
There was no significant difference in efficacy between the two levonorgestrel treatment groups in the pivotal study. Shorter intervals between intercourse and treatment were associated with lower pregnancy rates in both groups. C21H28O2. Molecular weight: 312.45.
C21H28O2. Molecular weight: 312.45.