What is in this leaflet
This leaflet answers some common questions about Pregabalin GH.
It does not contain all the available information. It does not take the place of talking to your doctor or pharmacist.
All medicines have risks and benefits. Your doctor has weighed the risks of you taking Pregabalin GH against the benefits it is expected to have for you.
If you have any concern about taking this medicine, ask your doctor or pharmacist.
Keep this leaflet with the medicine. You may need to read it again.
What Pregabalin GH is used for
Pregabalin GH is used to treat neuropathic pain, which is pain caused by an abnormality of, or damage to, the nerves.
Pregabalin GH is also used to control epilepsy. Epilepsy is a condition where you have repeated seizures (fits). There are many different types of seizures, ranging from mild to severe.
Pregabalin GH belongs to a group of medicines called anticonvulsants. These medicines are thought to work by controlling brain chemicals which send signals to nerves so that seizures do not happen. Pregabalin GH also has pain relieving effects.
Pregabalin GH may be used alone, or in combination with other medicines, to treat your condition.
Your doctor may prescribe Pregabalin GH in addition to your current therapy when your current treatment is no longer working as well as before.
Ask your doctor if you have any questions about why this medicine has been prescribed for you. Your doctor may have prescribed it for another reason.
This medicine is available only with a doctor's prescription.
Use in Children
There is not enough information to recommend the use of this medicine in children under the age of 18 years.
Before you take Pregabalin GH
When you must not take it
Do not take Pregabalin GH if you have an allergy to:
- pregabalin, the active ingredient in Pregabalin GH;
- any of the ingredients listed at the end of this leaflet.
Some of the symptoms of an allergic reaction may include:
- shortness of breath, wheezing or difficulty breathing;
- swelling of the face, lips, tongue or other parts of the body;
- rash, itching or hives on the skin.
Do not take this medicine after the expiry date printed on the pack or if the packaging is torn or shows signs of tampering. If the capsules have expired or the pack is damaged, return to your pharmacist for disposal.
If you are not sure whether you should start taking this medicine, talk to your doctor.
Before you start to take it
Tell your doctor or pharmacist if you have allergies to:
- any other medicines, especially barbiturates or any other anticonvulsant medicines;
- any other substances, such as foods, preservatives or dyes.
Tell your doctor if you have or have had any of the following medical conditions:
- congestive heart failure;
- hereditary problems with galactose metabolism;
- kidney problems;
- diabetes;
- depression.
Tell your doctor if you have a history of drug abuse. Pregabalin GH poses risks of misuse, abuse and dependence. Your body may become used to you taking Pregabalin GH and this may result in physical dependence. It means that you may experience withdrawal symptoms if you stop taking Pregabalin GH suddenly. So it is important to strictly follow the directions given by your doctor.
Tell your doctor if you are pregnant or plan to become pregnant. Pregabalin GH is not recommended for use during pregnancy. However, if you have epilepsy, it is very important to control your fits while you are pregnant. If it is necessary for you to take Pregabalin GH, your doctor can help you decide whether or not to take it during pregnancy.
Tell your doctor if you are breastfeeding or plan to breastfeed. The active ingredient in Pregabalin GH passes into breast milk and its safety in infants is unknown. It is recommended that you do not breastfeed while taking Pregabalin GH.
If you have not told your doctor about any of the above, tell them before you start taking Pregabalin GH.
Taking other medicines
Tell your doctor or pharmacist if you are taking any other medicines, including:
- all prescription medicines;
- all medicines, vitamins, herbal supplements or natural therapies you buy without a prescription from a pharmacy, supermarket, naturopath or health food shop.
Some medicines may be affected by Pregabalin GH or may affect how well it works. You may need different amounts of your medicines, or you may need to take different medicines. Your doctor will advise you.
Pregabalin GH and certain other medicines may influence each other.
Using Pregabalin GH with other medicines that can make you feel drowsy, such as sleeping tablets and other pain relievers (eg. benzodiazepines and opioids), antihistamines, antidepressants, anti-psychotics, cannabis, and alcohol may result in severe drowsiness, decreased awareness, breathing problems, coma and death. Your doctor will minimise the dose and duration of use; and monitor you for signs and symptoms of breathing difficulties and sedation.
Your doctor and pharmacist have more information on medicines to be careful with or avoid while taking Pregabalin GH.
How to take Pregabalin GH
Follow all directions given to you by your doctor carefully. They may differ from the information contained in this leaflet.
If you do not understand the instructions on the box, ask your doctor or pharmacist for help.
How much to take
Your doctor will tell you how many capsules you need to take each day. This may depend on your age, your condition and whether or not you are taking any other medicines. Your doctor may recommend that you start with a low dose of Pregabalin GH and slowly increase the dose to the lowest amount needed to control your epilepsy/convulsions or neuropathic pain.
The usual dose range is 150 mg per day to 600 mg per day given in two divided doses.
How to take it
Swallow the capsules whole with a full glass of water.
When to take it
Take your medicine at about the same time each day. Taking it at the same time each day will have the best effect. It will also help you remember when to take it. It does not matter if you take this medicine before or after food.
How long to take it
Continue taking your medicine for as long as your doctor tells you.
This medicine helps to control your condition, but does not cure it. It is important to keep taking your medicine, even if you feel well.
Do not stop taking Pregabalin GH, or lower the dosage, without checking with your doctor. Do not let yourself run out of medicine over the weekend or on holidays. Stopping Pregabalin GH suddenly may worsen your condition or increase your chance of experiencing withdrawal symptoms, such as sleeplessness, headache, nausea (feeling sick), anxiety, excessive sweating or diarrhoea (runny stools). If appropriate, your doctor will slowly reduce your dose before you can stop taking it completely.
If you forget to take it
If it is almost time for your next dose (within 4 hours), skip the dose you missed and take your next dose when you are meant to.
Otherwise, take it as soon as you remember, and then go back to taking your medicine as you would normally.
Do not take a double dose to make up for the dose that you missed. This may increase the chance of you getting an unwanted side effect.
If you are not sure what to do, ask your doctor or pharmacist.
If you have trouble remembering to take your medicine, ask your pharmacist for some hints.
If you take too much (overdose)
Immediately telephone your doctor or the Australian Poisons Information Centre (telephone 13 11 26) or the New Zealand National Poisons Information Centre (telephone 0800 POISON or 0800 764 766) for advice, or go to Accident and Emergency at the nearest hospital, if you think that you or anyone else may have taken too much Pregabalin GH. Do this even if there are no signs of discomfort or poisoning. You may need urgent medical attention.
Symptoms of an overdose with Pregabalin GH may include mood changes, feeling tired, confusion, depression, agitation, restlessness or seizures.
While you are taking Pregabalin GH
Things you must do
If you are about to be started on any new medicine, remind your doctor, dentist or pharmacist that you are taking Pregabalin GH.
Tell any other doctors, dentists and pharmacists who treat you that you are taking this medicine.
If you are going to have surgery, tell the surgeon or anaesthetist that you are taking this medicine. It may affect other medicines used during surgery.
Tell your doctor immediately if you experience any changes in your vision. Pregabalin GH may cause blurring or other changes in eyesight. Your doctor may ask you to stop taking Pregabalin GH to improve these symptoms.
Tell your doctor immediately if you have any thoughts of suicide or self-harm, any unusual changes in mood or behaviour, or show signs of depression. Some people taking medicines to treat convulsions, such as Pregabalin GH have had thoughts of harming themselves or taking their life.
Patients and caregivers should be alert and monitor for these effects.
Signs and symptoms of suicidal risk include:
- thoughts or talk of death or suicide;
- thoughts or talk of self-harm or harm to others;
- any recent attempts of self-harm;
- new or an increase in aggressive behaviour, irritability or agitation;
- new or worsening depression.
Mention of suicide or violence must be taken seriously.
If you or someone you know is demonstrating these warning signs of suicide while taking Pregabalin GH, contact your doctor or a mental health professional right away.
Tell your doctor if you feel Pregabalin GH is not helping your condition. Your doctor may need to change your medicine.
Tell your doctor if, for any reason, you have not taken Pregabalin GH exactly as prescribed. Otherwise, your doctor may change your treatment unnecessarily.
If you become pregnant while taking Pregabalin GH, tell your doctor immediately.
Keep all of your doctor's appointments so that your progress can be checked. Your doctor may do some tests from time to time to make sure the medicine is working and to prevent unwanted side effects.
Things you must not do
Do not take Pregabalin GH to treat any other complaints unless your doctor tells you to.
Do not give your medicine to anyone else, even if their symptoms seem similar to yours or they have the same condition as you.
Do not stop taking your medicine or lower the dosage without checking with your doctor.
Things to be careful of
Be careful driving or operating machinery until you know how Pregabalin GH affects you. As with other anticonvulsant medicines, Pregabalin GH may cause dizziness and drowsiness in some people. If you have any of these symptoms, do not drive, operate machinery or do anything else that could be dangerous.
Be careful when drinking alcohol while you are taking this medicine. If you drink alcohol, symptoms such as dizziness and drowsiness may be worse.
Side effects
Tell your doctor or pharmacist as soon as possible if you do not feel well while you are taking Pregabalin GH (Pregabalin capsules).
Pregabalin GH helps most people with neuropathic pain or epilepsy, but it may have unwanted side effects in a few people. All medicines can have side effects. Sometimes they are serious, most of the time they are not. You may need medical attention if you get some of the side effects.
It can be difficult to tell whether side effects are the result of taking Pregabalin GH, effects of your condition or side effects of other medicines you may be taking. For this reason, it is important to tell your doctor of any change in your condition.
If you are over 65 years of age you may have an increased chance of getting side effects.
Do not be alarmed by the list of side effects. You may not experience any of them.
If you get any side effects, do not stop taking Pregabalin GH without first talking to your doctor or pharmacist.
Ask your doctor or pharmacist to answer any questions you may have.
Tell your doctor if
Tell your doctor or pharmacist if you notice any of the following and they worry you:
- dizziness;
- feeling tired or drowsy;
- constipation;
- diarrhoea;
- nausea;
- headache;
- increase in weight;
- unsteadiness when walking, reduced co-ordination, shaking or tremors;
- dry mouth;
- blurred or double vision.
The above list includes the more common side effects of your medicine. They are usually mild and short-lived.
Tell your doctor as soon as possible if
Tell your doctor as soon as possible if you notice any of the following:
- unusual changes in mood or behaviour;
- signs of new or increased irritability or agitation;
- signs of depression;
- swelling of the hands, ankles or feet;
- enlargement of breasts;
- unexplained muscle pain, tenderness and weakness;
- passing little to no urine.
The above list includes serious side effects that may require medical attention. Serious side effects are rare.
Go to hospital if
Tell your doctor immediately or go to Accident and Emergency at your nearest hospital, if you notice any of the following:
- shortness of breath, swelling of the feet and legs, weight increase due to fluid build-up;
- irritated red eyes that are sensitive to light;
- more frequent or more severe seizures (fits);
- sudden signs of allergy such as rash, itching or hives, swelling of the face, lips, tongue or other parts of the body, shortness of breath, wheezing or difficulty breathing.
The above list includes very serious side effects. You may need urgent medical attention or hospitalisation. These side effects are very rare.
Tell your doctor or pharmacist if you notice anything else that is making you feel unwell. Other side effects not listed above may also occur in some people. Some of these side effects (for example, changes in blood pressure) can only be found when your doctor does tests from time to time to check your progress.
After taking Pregabalin GH
Storage
Keep your capsules in the pack until it is time to take them. If you take the capsules out of the pack they may not keep well.
Keep your capsules in a cool dry place where the temperature stays below 25°C.
Do not store Pregabalin GH or any other medicine in the bathroom or near a sink. Do not leave it on a windowsill or in the car on hot days. Heat and dampness can destroy some medicines.
Keep it where children cannot reach it. A locked cupboard at least one-and-a-half metres above the ground is a good place to store medicines.
Disposal
If your doctor tells you to stop taking this medicine or the expiry date has passed, ask your pharmacist what to do with any medicine that is left over.
Product description
What it looks like
Pregabalin GH 25 mg: Size '4' capsules with white cap and white body, imprinted with "PG" on cap and "25" on body in black ink, containing white to off white powder.
Pregabalin GH 50 mg: Size '4' capsules with white cap and white body, imprinted with "PG" on cap and "50 and band" on body in black ink, containing white to off white powder.
Pregabalin GH 75 mg: Size '4' capsules with dark brown cap and white body, imprinted with "PG" on cap and "75" on body in black ink, containing white to off white powder.
Pregabalin GH 100 mg: Size '3' capsules with dark brown cap and dark brown body, imprinted with "PG" on cap and "100" on body in black ink, containing white to off white powder.
Pregabalin GH 150 mg: Size '2' capsules with white cap and white body, imprinted with "PG" on cap and "150" on body in black ink, containing white to off white powder.
Pregabalin GH 200 mg: Size '1' capsules with light brown cap and light brown body, imprinted with "PG" on cap and "200" on body in black ink, containing white to off white powder.
Pregabalin GH 225 mg: Size '1' capsules with light brown cap and white body, imprinted with "PG" on cap and "225" on body in black ink, containing white to off white powder.
Pregabalin GH 300 mg: Size '0' capsules with dark brown cap and white body, imprinted with "PG" on cap and "300" on body in black ink, containing white to off white powder.
Pregabalin GH comes in PVC/Al blister pack of 14’s, 20’s, 56’s and 60’s.
Not all pack sizes may be marketed.
Ingredients
Active Ingredient
Each Pregabalin GH tablet contains 25 mg, 50 mg, 75 mg, 100 mg, 150 mg, 200 mg, 225 mg or 300 mg of pregabalin.
Other ingredients
- pregelatinised starch;
- purified talc;
- gelatin;
- titanium dioxide;
- sodium lauryl sulfate;
- purified water;
- Tek Print SW-9008 Black Ink (PI 2328) or Marking Solution Black BKI004 Black Ink ACG PAM BKI004 (PI 139674);
- iron oxide red (75 mg, 100 mg, 200 mg, 225 mg and 300 mg capsules only).
Australian Registration Numbers
Pregabalin GH 25 mg: AUST R 215685.
Pregabalin GH 50 mg: AUST R 215690.
Pregabalin GH 75 mg: AUST R 215683.
Pregabalin GH 100 mg: AUST R 215691.
Pregabalin GH 150 mg: AUST R 215680.
Pregabalin GH 200 mg: AUST R 215695.
Pregabalin GH 225 mg: AUST R 215697.
Pregabalin GH 300 mg: AUST R 215688.
Distributor
Generic Health Pty Ltd
Suite 2, Level 2
19-23 Prospect Street
Box Hill, VIC, 3128
Australia
E-mail: [email protected]
Telephone: +61 3 9809 7900
Website: www.generichealth.com.au
This leaflet was prepared in January 2021.
Published by MIMS March 2021





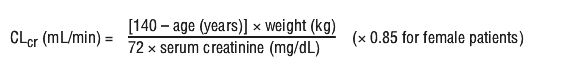 Pregabalin is removed effectively from plasma by haemodialysis (50% of drug in 4 hours). For patients receiving haemodialysis, the pregabalin daily dose should be adjusted based on renal function. In addition to the daily dose, a supplementary dose should be given immediately following every 4-hour haemodialysis treatment (see Table 1).
Pregabalin is removed effectively from plasma by haemodialysis (50% of drug in 4 hours). For patients receiving haemodialysis, the pregabalin daily dose should be adjusted based on renal function. In addition to the daily dose, a supplementary dose should be given immediately following every 4-hour haemodialysis treatment (see Table 1).
 The relative risk for suicidal thoughts or behaviour was higher in clinical trials for epilepsy than in clinical trials for psychiatric or other conditions, but the absolute risk differences were similar for the epilepsy and psychiatric indications.
The relative risk for suicidal thoughts or behaviour was higher in clinical trials for epilepsy than in clinical trials for psychiatric or other conditions, but the absolute risk differences were similar for the epilepsy and psychiatric indications. Additional adverse reactions reported in a pooled analysis of all pregabalin clinical trials are listed in Table 4 by System Organ Class (SOC). The frequency of these terms have been based on all causality adverse drug reactions in the clinical trial data set (very common (≥ 1/10), common (≥ 1/100, < 1/10), uncommon (≥ 1/1000, < 1/100) and rare (< 1/1000)).
Additional adverse reactions reported in a pooled analysis of all pregabalin clinical trials are listed in Table 4 by System Organ Class (SOC). The frequency of these terms have been based on all causality adverse drug reactions in the clinical trial data set (very common (≥ 1/10), common (≥ 1/100, < 1/10), uncommon (≥ 1/1000, < 1/100) and rare (< 1/1000)).


 A significant reduction in seizure frequency was observed by Week 1. Overall, there was a significant reduction in seizure frequency over the 12-week treatment period.
A significant reduction in seizure frequency was observed by Week 1. Overall, there was a significant reduction in seizure frequency over the 12-week treatment period. Chemical Name: (S)-3-(aminomethyl)-5-methylhexanoic acid.
Chemical Name: (S)-3-(aminomethyl)-5-methylhexanoic acid.