1 Name of Medicine
Prilocaine hydrochloride.
2 Qualitative and Quantitative Composition
The active ingredient in Prilotekal is prilocaine hydrochloride.
Prilotekal solution for intrathecal injection contains the following inactive ingredients: glucose sodium hydroxide 1N (for pH adjustment), water for injections.
3 Pharmaceutical Form
Solution for injection. Clear, colourless solution.
The pH is between 4.0 and 6.0.
The solution is hyperbaric with osmolality comprised between 490 and 540 mOsm/kg.
4.1 Therapeutic Indications
Prilotekal is indicated in adults for spinal anaesthesia in short-term surgical procedures.
4.2 Dose and Method of Administration
Prilotekal is only to be used for spinal anaesthesia.
The product contains no antimicrobial preservative. An ampoule should be used on one patient on one occasion only. Solutions showing discolouration and unused portions of solutions from ampoules should be discarded. The medicinal product has to be visually inspected prior to use. Only clear solutions practically free from particles should be used.
The safety and effectiveness of Prilotekal depend on proper dosage, correct technique and adequate precautions.
The equipment, drugs and personnel capable of dealing with an emergency, e.g. maintaining the patency of the airways and administering oxygen, must be immediately available, since in rare cases severe reactions, sometimes with a fatal outcome, have been reported after using local anaesthetics, even in the absence of individual hypersensitivity in the patient's case history.
Because of the possibility of hypotension and bradycardia, an IV cannula should be inserted before the local anaesthetic is injected.
If signs of acute systemic toxicity or total spinal block are observed, the injection of the local anaesthetic must be stopped immediately (see Section 4.4 Special Warnings and Precautions for Use).
Dosage.
Dosage must be established on an individual basis in accordance with the characteristics of the specific case. When determining the dose, take into consideration the patient's physical condition and the concomitant administration of other medicinal products. The lowest possible dose should be chosen.
The indications relating to recommended doses are valid in healthy adults of average height and weight (approximately 70 kg) for obtaining an effective block with one single administration. There are wide individual variations with regard to extent and duration of action. The experience of the anaesthetist and knowledge of the patient's general condition are essential for establishing the dose.
With regard to dosage the following guidelines are applied:
Adults. See Table 1.

Note.
i. Recommended doses.
Toxic doses vary widely between patients and toxic effects may occur after any local anaesthetic procedure. Careful observation of the patient must therefore be maintained. It is recommended that the dose of prilocaine at any one time should not exceed maximum recommended dose of 80 mg of prilocaine hydrochloride (= 4 mL Prilotekal).
The administered dose of Prilotekal must be tailored to the individual patient and procedure, and the maximum doses here quoted should be used as a guide only.
ii. Hypotension.
During spinal anaesthesia, a marked fall in blood pressure and/or intercostal paralysis may be seen, possibly due to the use of excessive doses or improper positioning of the patient. Hypotension and bradycardia may occur as a result of sympathetic blockade. Standard textbooks should be consulted with respect to techniques for administration of Prilotekal for spinal anaesthesia.
Paediatrics. The safety and efficacy of Prilotekal in paediatric population have not been established. No data are available.
The use of Prilotekal in children and adolescents is not recommended.
The use of Prilotekal in children younger than 6 months is contraindicated (see Section 4.3 Contraindications).
Geriatrics. A reduction in dosage may be necessary for elderly patients, especially those with compromised cardiovascular and/or hepatic function.
With impaired hepatic function. Although prilocaine is partly metabolised by the liver, dosage reduction is probably not warranted. However, caution should be exercised with repeated doses.
With impaired renal function. Impairment of renal function is unlikely to affect prilocaine clearance in the short-term (24 hours). However, toxicity due to accumulation may develop with prolonged or repeated administration.
Special population. It is advisable to reduce the dose in patients in a compromised general condition. In addition, in patients with established concomitant disorders (e.g. vascular occlusion, arteriosclerosis, diabetic polyneuropathy) a reduced dose is indicated.
Method of administration.
Due to the glucose content Prilotekal is only to be used for spinal anaesthesia. It is not for use in epidural anaesthesia.
Administer the injection slowly, after having aspirated a minimum quantity of CSF to confirm the correct position and check the patient's vital functions extremely carefully maintaining continuous verbal contact.
If signs of acute systemic toxicity or total spinal block are observed, the injection of the local anaesthetic must be stopped immediately (see Section 4.4 Special Warnings and Precautions for Use).4.3 Contraindications
Prilotekal is contraindicated for techniques of regional anaesthesia requiring continuous administration.
Prilotekal must not be used in patients with:
Hypersensitivity to prilocaine hydrochloride, other amide-type local anaesthetics or to any of the excipients listed in Section 6.1 List of Excipients. Detection of suspected sensitivity by skin testing is of limited value.
Serious problems with cardiac conduction including complete heart block.
In patients with hypoxemia e.g. as a result of severe anaemia, cardiac insufficiency etc, large doses of prilocaine may produce methaemoglobinaemia.
Decompensated cardiac insufficiency.
In patients with uncorrected hypotension including cardiogenic and hypovolemic shock.
Congenital or idiopathic acquired methaemoglobinemia.
Concomitant anticoagulant therapy general and specific contraindications for the technique of subarachnoid anaesthesia.
The use of Prilotekal in children younger than 6 months is contraindicated due to a higher risk of developing methaemoglobinemia. The intravascular injection of Prilotekal is contraindicated. Prilotekal must not be injected into infected areas. Also see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions.
4.4 Special Warnings and Precautions for Use
Due to the glucose content Prilotekal is only to be used for spinal anaesthesia. It is not for use in epidural anaesthesia.
Prilocaine may potentiate the formation of methaemoglobin by medicinal products known to induce methaemoglobin (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
It is essential for the doctor to recognize and be ready to treat undesirable effects, systemic toxicity and other complications. If signs of acute systemic toxicity or total spinal block are observed, the injection of the local anaesthetic must be stopped immediately (see Section 4.9 Overdose). Delay in proper management of dose-related toxicity, under-ventilation from any cause and/or altered sensitivity may lead to the development of acidosis, cardiac arrest and death.
The most effective prophylactic measures are scrupulous compliance with the recommended dose for Prilotekal, with it being essential for the doctor to check its action (visual and verbal contact with the patient), as well as careful aspiration prior to injecting the solution.
Some patients require special attention in order to reduce the risk of serious undesirable effects, even when locoregional anaesthesia constitutes the optimum choice for the surgical intervention:
Patients treated with class III antiarrhythmic agents (e.g. amiodarone). These patients should be subjected to careful observation and ECG monitoring, since cardiac effects may be added (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
In patients with acute porphyria, Prilotekal should only be administered when there is a compelling indication for its use, as Prilotekal may potentially precipitate porphyria. Appropriate precautions should be taken in all patients with porphyria.
Ensuring the presence of reliable venous access and the availability of vasopressor drugs and oxygen is recommended. As with all local anaesthetics, a drop in arterial pressure may occur and cardiac frequency may slow.
A serious, undesirable effect of spinal anaesthesia is high or total spinal block, with consequent cardiovascular and respiratory depression. Cardiovascular depression is induced by an extended block of the sympathetic nervous system, which may induce severe hypotension and bradycardia to the point of cardiac arrest. Respiratory depression is induced by the block of the respiratory musculature and the diaphragm.
Rarely, neurological damage may occur after spinal anaesthesia, manifesting as paresthesia, loss of sensitivity, motor weakness and paralysis. Occasionally these symptoms persist.
Local anaesthetics should be given with great caution (if at all) to patients with pre-existing abnormal neurological conditions.
Careful evaluation of the risk-benefit ratio is recommended prior to treatment.
Local anaesthetics react with certain metals and cause the release of their respective ions which, if injected, may cause severe irritation. Adequate precautions should be taken to avoid prolonged contact between Prilotekal solutions and metal surfaces such as metal bowls, cannulae and syringes with metal parts.
Methaemoglobinemia.
Methaemoglobinemia may follow the administration of prilocaine.
The doses used in single-shot subarachnoid anaesthesia do not induce blood levels capable of inducing methaemoglobinemia, which occurs if the quantity of prilocaine hydrochloride administered is equal to or higher than 600 mg.
There is a metabolite of prilocaine, o-toluidine, which can induce methaemoglobin formation. In general, methaemoglobin formation is clinically negligible, except in cases of extremely severe anaemia and high grade cardiac decompensation.
Patients with severe anaemia may develop hypoxia. It is important to exclude other serious causes of cyanosis, e.g. acute hypoxia and/or cardiac insufficiency.
Management of methaemoglobinemia.
Proven methaemoglobinemia resolves 15 minutes after the i.v. injection of 2-4 mg/kg body weight of toluidine blue.
Even low concentrations of methaemoglobin can alter measurements of pulse oximetry.
Use in hepatic impairment.
Although prilocaine is partly metabolized by the liver, dosage reduction is probably not warranted. However, caution should be exercised with repeated doses. See Section 4.2 Dose and Method of Administration.
Use in renal impairment.
Impairment of renal function is unlikely to affect prilocaine clearance in the short-term (24 hours). However, toxicity due to accumulation may develop with prolonged or repeated administration. See Section 4.2 Dose and Method of Administration.
Use in the elderly.
In elderly patients there is an increased risk of high or total spinal block: consequently, it is advisable to reduce the anaesthetic dose. An unexpected drop in arterial pressure may occur as a complication of spinal anaesthesia. A reduction in dosage may be necessary for elderly patients, especially those with compromised cardiovascular and/or hepatic function. See Section 4.2 Dose and Method of Administration.
Paediatric use.
The safety and efficacy of Prilotekal in paediatric population have not been established. No data are available.
The use of Prilotekal in children and adolescents is not recommended.
The use of Prilotekal in children younger than 6 months is contraindicated (see Section 4.3 Contraindications).
Effects on laboratory tests.
No data available.4.5 Interactions with Other Medicines and Other Forms of Interactions
1. Methaemoglobinemia.
Prilocaine may potentiate the formation of methaemoglobin by medicinal products known to induce methaemoglobin (e.g. sulfonamides, antimalarials, sodium nitroprusside and nitroglycerine). See Section 4.4 for more details.
2. Anti-arrhythmic drugs.
In the event of the concomitant use of prilocaine and other local anaesthetics or medicinal products with a chemical structure similar to prilocaine, e.g. certain antiarrhythmics such as lidocaine and mexiletine, it is possible for undesirable effects to be added. No studies have been performed on interactions between prilocaine and class III antiarrhythmics (e.g. amiodarone), but care must also be taken in this case (also see Section 4.4 Special Warnings and Precautions for Use).
3. Cardiovascular system and CNS.
The combination of various local anaesthetics induces additional effects which affect the cardiovascular system and the CNS. Prilotekal should be used with caution in patients receiving other local anaesthetics or agents structurally related to amide-type local anaesthetics, since the systemic toxic effects are additive.4.6 Fertility, Pregnancy and Lactation
Effects on fertility.
No data available.
(Category A)
Although prilocaine is indicated for anaesthesia in obstetrics there is no information on prilocaine's use in early pregnancy. Therefore, with the exception of its use in obstetrics, the drug should not be used in pregnant women, or those likely to become pregnant, unless the expected benefit outweighs any potential risk.
Prilocaine crosses the placenta and almost equal concentrations have been found in fetal and maternal blood following epidural blocks. During prolonged epidural blockade, prilocaine may cause maternal and fetal methaemoglobinaemia which could lead to fetal hypoxia.
However, if prilocaine is used for paracervical block, fetal heart rate should always be monitored since fetal bradycardia/ tachycardia frequently follows paracervical block and may be associated with foetal acidosis and hypoxia. The possible undesired consequences of a paracervical block should be weighed against its advantages when fetal distress is expected or when factors predisposing to fetal distress are present (such as toxaemia, prematurity, diabetes).
Neonatal methaemoglobinaemia has been reported after paracervical block or pudendal block in the obstetric patient.
It is not known whether prilocaine or its metabolites appear in breast milk.4.7 Effects on Ability to Drive and Use Machines
Depending on dosage local anaesthetics may have a very mild effect on mental function and may temporarily impair locomotion and coordination.
Patients should be warned of this possibility and advised not to drive a motor vehicle or operate machinery if affected.
4.8 Adverse Effects (Undesirable Effects)
The possible undesirable effects due to the use of Prilotekal are generally similar to the undesirable effects of other local anaesthetics for spinal anaesthesia from the amide group.
The undesirable effects include the physiological effects of the nerve block (e.g. reduction in arterial pressure, bradycardia, temporary urine retention).
The effects of the spinal insertion technique should be distinguished (e.g. spinal hematoma, post-spinal headache, meningitis).
The frequency of onset of undesirable effects is classified as follows: very common (≥ 1/10), common (≥ 1/100 to < 1/10), uncommon (≥ 1/1000 to < 1/100), rare (≥ 1/10,000 to < 1/1000), very rare (< 1/10,000). Within each frequency grouping, undesirable effects are presented in order of decreasing seriousness. See Table 2.
 Adverse effects may be due to high plasma levels as a result of excessive dosage, rapid absorption, delayed elimination or metabolism, or inadvertent intravascular injection. Pronounced acidosis or hypoxia may increase the risk and severity of toxic reactions.
Adverse effects may be due to high plasma levels as a result of excessive dosage, rapid absorption, delayed elimination or metabolism, or inadvertent intravascular injection. Pronounced acidosis or hypoxia may increase the risk and severity of toxic reactions.
These undesirable effects mainly manifest as symptoms affecting the central nervous and cardiovascular system.
Mild undesirable effects (feeling dizzy or dazed) can be attributed to moderate overdose and generally resolve rapidly after reducing the dose or halting administration of Prilotekal.
Serious undesirable effects are attributable to significant overdose and/or accidental injection of local anaesthetic into a blood vessel. They manifest as symptoms affecting the central nervous system (restlessness, speech problems, disorientation, dizziness, muscle contractions, cramps, vomiting, loss of consciousness, respiratory arrest and mydriasis) and the cardiocirculatory system (raised arterial pressure and pulse frequency, arrhythmia, drop in arterial pressure, asystole) following irritation and/or depression of the cerebral cortex and the cerebral marrow (see Section 4.9).
In addition, following inhibition or block of the cardiac conduction system, cardiac frequency may slow down and myocardial depression may occur.
Any problems relating to metabolism (liver) or excretion (kidney) of Prilotekal should also be considered as other possible causes of undesirable effects.
Post marketing data.
Post-marketing surveillance has reported cases of the rare event of myoclonus. Transient neurologic symptoms have been reported in the published literature after the administration of intrathecal prilocaine.
Reporting suspected adverse effects.
Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.4.9 Overdose
It is unlikely that Prilotekal, at the recommended dose, will induce plasma levels capable of inducing systemic toxicity. After intrathecal administration, systemic toxicity is expected to be low, due to the low dose administered.
High or total spinal block.
However, an excessive dose administered into the intrathecal space may give rise to total spinal block, with consequent cardiovascular and respiratory depression. Cardiovascular depression is induced by an extended block of the sympathetic nervous system, which may induce severe hypotension and bradycardia to the point of cardiac arrest. Respiratory depression is induced by the block of the respiratory musculature and the diaphragm.
If cardiovascular depression occurs (hypotension, bradycardia), appropriate treatment with intravenous fluids, vasopressor, inotropic agents, assisted or controlled ventilation (intubation) with oxygen may be necessary.
Acute systemic toxicity.
With accidental intravenous administration, the toxic effect occurs within 1-3 minutes. With an overdose maximum plasma concentrations are only reached after 20-30 minutes, depending on the injection site, and the onset of signs of toxicity is delayed.
Signs of overdose can be classified into two different sets of symptoms which differ in terms of quality and intensity:
a) Symptoms affecting the central nervous system.
Generally, the first symptoms are paresthesia in the mouth area, feeling of numbness of the tongue, feeling dazed, problems with hearing and tinnitus. Visual problems and muscle contractions are more severe and precede a generalized convulsion. These signs must not be erroneously mistaken for neurotic behaviour.
Subsequently loss of consciousness and tonic-clonic seizure may occur, generally lasting between a few seconds and a few minutes. The convulsions are immediately followed by hypoxia and increased levels of carbon dioxide in the blood (hypercapnia), attributable to increased muscular activity associated with respiratory problems. In serious cases respiratory arrest may occur. Acidosis potentiates the toxic effects of local anaesthetics.
The reduction or improvement of symptoms affecting the central nervous system can be attributed to the redistribution of local anaesthetics outside the CNS, with its consequent metabolism and excretion. Regression may be rapid, unless enormous quantities have been used.
b) Cardiovascular symptoms.
In serious cases cardiovascular toxicity may occur rapidly and with little warning and can lead to peripheral vasodilation, hypotension, myocardial depression, bradycardia, arrhythmia and also cardiac arrest may occur in the presence of a high systemic concentration of local anaesthetics. The first signs of toxic symptoms affecting the central nervous system generally precede toxic cardiovascular effects. This statement does not apply if the patient is under general anaesthesia or heavily sedated with medicinal products such as benzodiazepine or barbiturates.
Management of acute systemic toxicity. If signs of acute systemic toxicity appear injection of the local anaesthetic should be stopped immediately.
If convulsions occur then immediate attention is required for the maintenance of a patent airway and assisted or controlled ventilation with oxygen, via a positive airway pressure delivery system mask. Adequacy of the circulation should then be evaluated, bearing in mind that drugs used to treat convulsions depress the circulation when administered intravenously.
Should convulsions persist despite adequate respiratory support, and if the status of the circulation permits, appropriate anticonvulsant medication such as an ultra-short acting barbiturate (e.g. thiopentone) or a benzodiazepine (e.g. diazepam) may be administered IV. The clinician should be familiar with these anticonvulsant drugs prior to the use of local anaesthetics.
If ventricular fibrillation or cardiac arrest occurs, effective cardiovascular resuscitation treatment must be instituted and maintained for a prolonged period if necessary.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action.
Prilocaine stabilises the neuronal membrane and reversibly prevents the initiation and conduction of nerve impulses thereby producing local anaesthesia.
Prilocaine hydrochloride has a similar time of onset and potency to lignocaine. Prilocaine has lower CNS toxicity than lignocaine.
The onset and duration of anaesthesia depend on the route of administration, status of the patient and the dosage (volume and concentration) employed.
Clinical trials.
In a phase III, prospective, double-blind, randomized trial, the non-inferiority of two doses of a hyperbaric formulation of prilocaine 20 mg/mL (60 mg and 40 mg) was evaluated compared to an isobaric formulation of prilocaine 20 mg/mL (60 mg) in terms of sensory block onset at the T10 level (primary endpoint). A maximum difference of 4 minutes in terms of time to onset of anesthesia between the product tested and the reference was set as a non-inferiority margin based on extrapolation from the literature.
Ninety (90) adult patients undergoing outpatient surgery lasting less than 60 minutes were randomized 1:1:1 to receive before surgery, an intrathecal dose of prilocaine (40 mg or 60 mg of prilocaine hyperbaric or 60 mg of isobaric prilocaine).
The primary analysis was to compare the time to achieve a T10 level of sensory block (Tsb) between hyperbaric doses and isobaric dose. The reference treatment (60 mg 2% prilocaine isobaric) had a 20% failure (6/30) to achieve the primary endpoint.
Median Tsb was 5 minutes (5 to 20 minutes) in the group treated with 60 mg hyperbaric prilocaine, 10 minutes (5 to 20 minutes) in the group treated with 40 mg hyperbaric prilocaine and 15 minutes (5 to 25 minutes) in the group treated with 60 mg of isobaric prilocaine. Non-inferiority of both the hyperbaric doses to the isobaric formulation was shown.
The test treatments differed from the reference treatment, in terms of motor block onset (Tmb).
The median Tmb was 7.5 minutes (5 to 15 minutes) in the group treated with 60 mg hyperbaric prilocaine, 5 minutes (4 to 20 minutes) in the group treated with 40 mg hyperbaric prilocaine and 10 minutes (5 to 25 minutes) in the group treated with 60 mg of isobaric prilocaine.
5.2 Pharmacokinetic Properties
Absorption and distribution.
The plasma concentration should be negligible with truly intrathecal use.
Metabolism and excretion.
Amidases in the liver, kidneys and lungs metabolise prilocaine directly. One metabolite excreted in the urine is o-toluidine which is believed to cause the methaemoglobinaemia observed after large doses of prilocaine (see Section 4.4 Special Warnings and Precautions for Use; Section 4.8 Adverse Effects (Undesirable Effects)).
5.3 Preclinical Safety Data
Genotoxicity.
There was no evidence for genotoxicity of prilocaine in assays for gene mutation (bacterial reverse mutation) or chromosomal damage (human lymphocytes in vitro, mouse micronucleus test). A metabolite of prilocaine, o-toluidine, has shown evidence for induction of DNA adducts and clastogenicity.
Carcinogenicity.
Carcinogenicity assays have not been conducted with prilocaine. A metabolite of prilocaine, o-toluidine, is carcinogenic in rodent bioassays and, based on analysis of cancer incidence in occupationally-exposed human populations, has been categorised as carcinogenic to humans. Risk assessments comparing expected human exposure to o-toluidine from infrequent/short term use of prilocaine with carcinogenic doses in rodent studies suggest a wide margin of safety for clinical use. This margin may be reduced with frequent/long term treatment with prilocaine.6 Pharmaceutical Particulars
6.1 List of Excipients
Glucose, sodium hydroxide 1N (for pH adjustment), water for injections.
6.2 Incompatibilities
Dilution or infusion of the injection is not recommended. Incompatibilities have not been assessed.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 25°C.
Do not refrigerate or freeze.
The medicinal product has to be used immediately after first opening. Restricted to hospital use only.
6.5 Nature and Contents of Container
Type I clear colourless glass ampoule.
Box of 10 ampoules each containing 5 mL of solution for injection. 1 ampoule with 5 mL solution, contains 100 mg of prilocaine hydrochloride [1 mL of solution for injection contains 20 mg of prilocaine hydrochloride (equivalent to 2%)].
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of in accordance with local requirements.
6.7 Physicochemical Properties
Pharmacotherapeutic group: anaesthetics, local; amides ATC code: N01BB04.
The chemical name for prilocaine hydrochloride is 2-propylamino propiono-o-toluidide hydrochloride; also known as propitocaine hydrochloride.
Prilocaine hydrochloride has a pKa of 7.89. Prilocaine base, which has a molecular weight of 220.3, is only slightly soluble in water. Prilocaine hydrochloride is soluble 1:5 in water.
Chemical structure.
The chemical structure of prilocaine hydrochloride is:
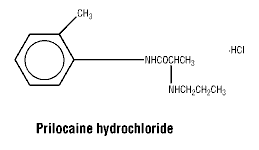
CAS number.
The CAS number for prilocaine hydrochloride is 1786-81-8.7 Medicine Schedule (Poisons Standard)
S4 (Prescription Only Medicine).
Summary Table of Changes



 Adverse effects may be due to high plasma levels as a result of excessive dosage, rapid absorption, delayed elimination or metabolism, or inadvertent intravascular injection. Pronounced acidosis or hypoxia may increase the risk and severity of toxic reactions.
Adverse effects may be due to high plasma levels as a result of excessive dosage, rapid absorption, delayed elimination or metabolism, or inadvertent intravascular injection. Pronounced acidosis or hypoxia may increase the risk and severity of toxic reactions.
