WHAT IS IN THIS LEAFLET
This leaflet answers some of the common questions about PRIORIX-TETRA vaccine. It does not contain all the available information. It does not take the place of talking to your doctor, nurse or pharmacist.
All medicines and vaccines have risks and benefits. Your doctor has weighed the possible risks of you or your child having PRIORIX-TETRA against the expected benefits.
If you have any concerns about PRIORIX-TETRA talk to your doctor, nurse or pharmacist.
Keep this leaflet with this vaccine. You may need to read it again.
WHAT PRIORIX-TETRA IS USED FOR
PRIORIX-TETRA is a vaccine used to prevent four diseases, measles, mumps, rubella (German measles) and varicella (chickenpox).
Measles, mumps, rubella and varicella are all infectious diseases caused by viruses. The vaccine works by causing the body to produce its own protection (antibodies) against these diseases.
Measles
Measles is a highly infectious viral illness. Measles is spread by small droplets from the nose, throat or mouth of an infected person (often before it is obvious they have measles). Measles often begins with a fever, runny nose, hacking cough and conjunctivitis (eye inflammation). The rash appears 3-5 days after the onset of symptoms and spreads rapidly all over the body. Measles is often a severe disease complicated by ear infection and pneumonia (lung infection). Encephalitis (swelling of the brain) can also occur and can sometimes result in permanent brain damage or death.
Unimmunised children in the following groups are at particular risk from severe measles infection:
- children with chronic conditions such as cystic fibrosis, congenital heart or kidney disease, failure to thrive, Down's Syndrome
- children from the age of 1 year upwards in child care centres, family day care and playgroups
- children living in institutions
- Aboriginal and Torres Strait Islander children.
Mumps
Mumps is an infectious viral disease spread by infected droplets of saliva. Symptoms often begin with chilly sensations, headache, loss of appetite, general unwellness and fever. The salivary glands on one or both sides of the face can then become very painful and swollen. Complications include meningoencephalitis (swelling around the brain) and in older patients the disease may involve other organs e.g. testes in males.
Rubella (German measles)
Rubella is generally a mild infectious viral illness spread by airborne droplets. It can cause mild fever, general unwellness, swollen glands and mild red rash. It often goes unnoticed in adolescents and adults. However rubella can cause miscarriage, stillbirth or birth defects in infants born to mothers infected with rubella during the early months of pregnancy.
Varicella (Chickenpox)
Chickenpox is a highly infectious disease, which usually causes an itchy, red rash with blisters. After about 1 week, most of the blisters have normally crusted over. The rash can appear on the face, scalp, body, or in and around the mouth, eyes and bottom. Other symptoms can include fever, headaches, chills, and muscle and/or joint aches and pains. Sometimes disease complications can occur such as bacterial infection of the skin (often due to scratching of the itchy rash/crusts), inflammation of the brain (varicella encephalitis), and lung infection (varicella pneumonia).
Full recovery from chickenpox generally occurs; however, later in life the virus can become active again. This condition is known as shingles or Herpes zoster.
Vaccination is the best way to protect against these severe diseases. The vaccine will not protect against diseases caused by other types of viruses or organisms.
Like other varicella vaccines, PRIORIX-TETRA cannot completely protect your child against catching chickenpox. However, people who have been vaccinated and catch chickenpox usually have a very mild disease, compared with people who have not been vaccinated.
Although PRIORIX-TETRA contains live viruses, they are too weak to cause severe disease in healthy people. Occasionally, some spots, blisters and fever may appear in the first two weeks after vaccination.
BEFORE RECEIVING PRIORIX-TETRA
DO NOT HAVE PRIORIX-TETRA IF:
- your child has had an allergic reaction to PRIORIX-TETRA, or any ingredient contained in this vaccine. The ingredients are listed at the end of this leaflet. Signs of an allergic reaction may include itchy skin, rash, shortness of breath and swelling of the face or tongue.
- your child is known to be allergic to neomycin. Signs of an allergic reaction are listed above. If your child has a history of skin allergy to neomycin they can still be vaccinated.
If your child has had PRIORIX-TETRA or another vaccine containing measles, mumps, rubella or varicella before and became unwell, tell your doctor or nurse before the next dose is given.
- your child has a severe infection with a high temperature. A minor infection such as a cold should not be a problem, but talk to your doctor or nurse about this before being vaccinated.
- your child has ever had a severe allergic reaction to eggs or anything that contained eggs.
- you are or think you may be pregnant or if you intend to become pregnant within one month. Your doctor will discuss with you the risks of receiving PRIORIX-TETRA prior to or during pregnancy.
- your child has lowered immunity. This can occur in persons:
- with inherited (or family history of) immune deficiency conditions
- with abnormal blood conditions or blood protein (immunoglobulin) disorders
- with cancer
- receiving or who have received certain drugs (ie cyclosporin, corticosteroids, and cancer medicines)
- receiving or who have received radiation therapy - the expiry date printed on the pack has passed
- the packaging is torn or shows signs of tampering
If you are not sure whether PRIORIX-TETRA should be given, talk to your doctor or nurse. Do not give this vaccine to anyone else; your doctor has prescribed it specifically for you/or your child.
BEFORE HAVING PRIORIX-TETRA TELL YOUR DOCTOR IF
- your child has any medical problems such as:
- a history or family history of convulsions (fits or seizures)
- a history or family history of allergic diseases
- a skin allergy to neomycin
- a bleeding disorder.
Sometimes PRIORIX-TETRA may need to be given differently in people with bleeding problems.
- a weakened immune system. Your child should be closely monitored as the response to the vaccine may not be sufficient to ensure protection against the illness. - your child has allergies to any other medicines or substances, such as dyes, foods or preservatives.
- you are breast feeding. Your doctor will discuss the risks and benefits of vaccination.
- your child has received another vaccine within the last month.
- your child has received a blood or plasma transfusion, or been given gamma globulin or other immunoglobulin within the last 3 months. PRIORIX-TETRA may be less effective if given within 3 months of these products. Your doctor will decide when to give the vaccine.
- your child is taking any prescription or OTC (over-the-counter) medicines. Some vaccines may be affected by other vaccines or medicines. Your doctor, nurse or pharmacist will be able to tell you what to do if PRIORIX-TETRA is to be given with another vaccine or medicine.
- Your child has had any testing for tuberculosis (TB). PRIORIX-TETRA may affect the result of the tuberculin test.
Fainting can occur following, or even before, any needle injection, therefore tell the doctor or nurse if you/your child fainted with a previous injection.
HOW PRIORIX-TETRA IS GIVEN
The doctor or nurse will give PRIORIX-TETRA as an injection.
If you have any concerns about how this vaccine is to be given, talk to your doctor, nurse or pharmacist.
HOW MUCH IS GIVEN
PRIORIX-TETRA is generally given as a single 0.5mL dose.
HOW IT IS GIVEN
The vaccine comes as a powder which is mixed with sterile water before use. PRIORIX-TETRA is injected under the skin (subcutaneously) or into the muscle (intramuscularly). PRIORIX-TETRA is generally injected into the upper leg in infants under 12 months of age. In children over 12 months of age the injection may be given in the upper arm.
PRIORIX-TETRA should never be given into a vein.
WHEN IT WILL BE GIVEN
The appropriate time and number of doses that will be given will be determined by your doctor on the basis of appropriate official recommendations.
IF A DOSE IS MISSED
If your child misses a scheduled dose, talk to your doctor or nurse and arrange another visit as soon as possible.
AFTER RECEIVING PRIORIX-TETRA
THINGS YOU MUST NOT DO:
- do not become pregnant for one month after receiving PRIORIX-TETRA vaccination. Talk to your doctor as soon as possible, if you do become pregnant within this time.
THINGS YOU MUST DO:
Tell your doctor your child has received PRIORIX-TETRA if:
- they are to have a tuberculin skin test for tuberculosis within 4-6 weeks after vaccination. The results of the test may be affected by the vaccine.
- they are to have another vaccine within 1 month after vaccination.
- they are to have a blood or plasma transfusion, or be given gamma globulin or other immunoglobulin within 2 weeks after vaccination.
THINGS TO BE CAREFUL OF:
It is advised to remain in the clinic for about 15 minutes after receiving the injection. There is a rare risk of allergic reactions. These may be local or widespread rashes that may be itchy or blistering, swelling of the eyes and face, difficulty in breathing or swallowing, a sudden drop in blood pressure and loss of consciousness. These reactions will usually occur before leaving the doctor’s surgery. If these symptoms occur, you should contact a doctor immediately.
SIDE EFFECTS
Tell your doctor, nurse or pharmacist as soon as possible if you (or your child) have troublesome symptoms after having had a dose of PRIORIX-TETRA.
PRIORIX-TETRA helps protect most people from measles, mumps, rubella and varicella infection, but it may have unwanted side effects in a few people. All medicines and vaccines can have side effects. Sometimes they are serious; most of the time they are not. Some side effects may need medical treatment. The chance of your child having a serious side effect is very much less than the chance of you or your child having a permanent injury from the natural infections.
Ask your doctor, nurse or pharmacist to answer any questions you may have.
Most unwanted effects with PRIORIX-TETRA are mild. These effects, as with other vaccines, generally occur around the injection site.
Side effects that occurred during clinical trials with PRIORIX-TETRA were as follows:
Very common (these may occur with more than 1 in 10 doses of the vaccine):
- pain and redness around the injection site
- fever greater than 37.5°C*
Common (these may occur with up to 1 in 10 doses of the vaccine):
- swelling around the injection site
- fever greater than 39°C*
- irritability
- rash (spots and/or blisters)
* Higher rates of fever were observed after administration of the first dose of PRIORIX-TETRA when compared to measles-mumps-rubella and varicella vaccines administered separately at the same visit.
Uncommon (these may occur with up to 1 in 100 doses of the vaccine):
- crying
- generally feeling unwell
- loss of appetite
- vomiting
- diarrhoea
- nervousness
- inability to sleep
- sleepiness
- tiredness (fatigue)
- lack of energy
- runny nose
- upper respiratory tract infection
- swollen glands in the cheek
- swollen glands in the neck, armpit and groin
Rare (these may occur with up to 1 in 1,000 doses of the vaccine):
- seizures with fever
- coughing
- infection of the middle ear
- bronchitis
After commercialisation, the following additional side effects have been rarely reported in people vaccinated with PRIORIX-TETRA:
- chickenpox-like rash
- shingles (herpes zoster)
- mumps like symptoms (including transient, painful swelling of the testicles and swollen glands in the neck)
- temporary lumpy rash that may affect the skin, mouth and other parts of the body
- joint and muscle pains
- infection around the brain or spinal cord (meningitis)
- infection or inflammation of the nervous system resulting in temporary loss of control of bodily movements, walking or sensation changes
- bleeding or bruising more easily than normal which may be associated with skin rashes/peeling or fever
Other side effects not listed above, can also occur during or soon after a dose of PRIORIX-TETRA.
Check with your doctor or nurse if you or your child has any other effects.
Do not be alarmed by this list of possible side effects. You or your child may not experience any of them.
STORAGE
PRIORIX-TETRA is usually stored at the doctor’s clinic or surgery, or at the pharmacy. But if you need to store PRIORIX-TETRA always:
- Keep PRIORIX-TETRA in the refrigerator stored between 2°C and 8°C. DO NOT STORE PRIORIX-TETRA IN THE FREEZER. Do not store it in the bathroom, near the sink, or leave it in the car on hot days. Avoid exposing the vaccine to sunlight.
- Keep the vaccine out of the reach of children.
- Keep PRIORIX-TETRA in the original pack until it is time for it to be given.
Ask your pharmacist what to do with any left over PRIORIX-TETRA that has expired or has not been used.
PRODUCT DESCRIPTION
WHAT IT LOOKS LIKE
PRIORIX-TETRA comes as a whitish to slightly pink powder in a glass vial. This is mixed (reconstituted) with water diluent supplied in a glass syringe. The reconstituted vaccine may vary in colour from clear peach to fuchsia pink.
The vaccine should be injected as soon as possible after reconstitution, and no later than 8 hours after reconstitution.
INGREDIENTS
The active ingredients of PRIORIX-TETRA are live weakened measles, mumps, rubella and varicella viruses in a dry powder. Each 0.5mL dose contains not less than:
- 103.0 CCID50 (cell culture infectious dose 50%) of the Schwarz measles
- 104.4 CCID50 of the RIT 4385 mumps,
- 103.0 CCID50 of the Wistar RA 27/3 rubella and
- 103.3 PFU (plaque forming units) of the OKA varicella-zoster virus strains.
The inactive ingredients in the vaccine are: lactose, amino acids, sorbitol and mannitol. Neomycin sulphate is present as a residual from the manufacturing process. The vaccine is mixed with sterile Water for Injections before use.
The manufacture of this product includes exposure to bovine derived materials. No evidence exists that any case of vCJD (considered to be the human form of bovine spongiform encephalopathy) has resulted from the administration of any vaccine product.
FURTHER INFORMATION
PRIORIX-TETRA is only available if prescribed by a doctor.
PRIORIX-TETRA comes as a whitish to slightly pink powder in a glass vial as:
- individual packs containing one vial of vaccine and one syringe of sterile water diluent (AUST R 107286)
- packs of 10 vials of vaccine and 10 syringes of diluent (AUST R 107286)
MANUFACTURER
GlaxoSmithKline Biologicals s.a.
Rue de l'Institut 89,
1330 Rixensart, Belgium.
DISTRIBUTED IN AUSTRALIA BY
GlaxoSmithKline Australia Pty Ltd
Level 4, 436 Johnston Street
Abbotsford, Victoria 3067, Australia
Date of Preparation:
25 July 2022
Version 7.0
Trade marks are owned by or licensed to the GSK group of companies.
©2022 GSK group of companies or its licensor.
Published by MIMS December 2022

 Always hold the syringe by the barrel, not by the syringe plunger or the Luer Lock Adaptor (LLA), and maintain the needle in the axis of the syringe (as illustrated in Figure 1). Failure to do this may cause the LLA to become distorted and leak.
Always hold the syringe by the barrel, not by the syringe plunger or the Luer Lock Adaptor (LLA), and maintain the needle in the axis of the syringe (as illustrated in Figure 1). Failure to do this may cause the LLA to become distorted and leak. Analysis of the three pivotal studies has shown that redness was the most frequently reported solicited local symptom, in both vaccine treatments. There was no difference between the vaccination regimens, after the first dose.
Analysis of the three pivotal studies has shown that redness was the most frequently reported solicited local symptom, in both vaccine treatments. There was no difference between the vaccination regimens, after the first dose. When summarised according to age stratum for toddlers (15 months-2 years) and children (2-6 years) lower levels of all fever were reported in toddlers who received Priorix-Tetra as a second dose of a measles containing vaccine post MMR vaccine (40.9%), compared to studies where toddlers were administered Priorix-Tetra as a first dose of a measles containing vaccine (61.15%). Fever > 39.5°C was observed in 2.7% of toddlers who received Priorix-Tetra as a second dose of a measles containing vaccine post MMR vaccine, compared with 11.2% in studies where Priorix-Tetra was used as the first dose of a measles containing vaccine.
When summarised according to age stratum for toddlers (15 months-2 years) and children (2-6 years) lower levels of all fever were reported in toddlers who received Priorix-Tetra as a second dose of a measles containing vaccine post MMR vaccine (40.9%), compared to studies where toddlers were administered Priorix-Tetra as a first dose of a measles containing vaccine (61.15%). Fever > 39.5°C was observed in 2.7% of toddlers who received Priorix-Tetra as a second dose of a measles containing vaccine post MMR vaccine, compared with 11.2% in studies where Priorix-Tetra was used as the first dose of a measles containing vaccine.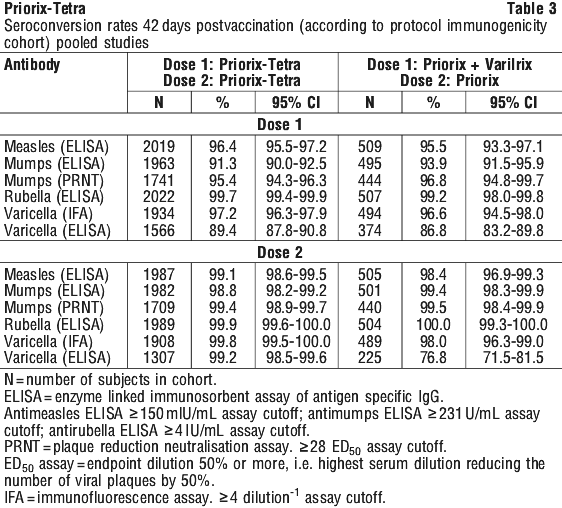 Three years after vaccination with two doses of Priorix-Tetra, 98.5%, 97.4%, 100% and 99.4% of all vaccinees were still seropositive for respectively anti-measles, anti-mumps, anti-rubella and anti-varicella antibodies.
Three years after vaccination with two doses of Priorix-Tetra, 98.5%, 97.4%, 100% and 99.4% of all vaccinees were still seropositive for respectively anti-measles, anti-mumps, anti-rubella and anti-varicella antibodies. In a pivotal clinical study in subjects receiving two-doses of Priorix-Tetra given six weeks apart, at the one year follow-up after the second dose of Priorix-Tetra, no breakthrough cases were reported for measles, mumps and rubella despite reported contacts with wild virus. Exposures to varicella or zoster were reported in 14.5% in the Priorix-Tetra group versus 20.0% in the control group (Priorix + Varilrix for the first dose; Priorix alone for the second dose). Breakthrough cases were reported in 0.34% of Priorix-Tetra recipients, as opposed to 1.9% of children in the control group.
In a pivotal clinical study in subjects receiving two-doses of Priorix-Tetra given six weeks apart, at the one year follow-up after the second dose of Priorix-Tetra, no breakthrough cases were reported for measles, mumps and rubella despite reported contacts with wild virus. Exposures to varicella or zoster were reported in 14.5% in the Priorix-Tetra group versus 20.0% in the control group (Priorix + Varilrix for the first dose; Priorix alone for the second dose). Breakthrough cases were reported in 0.34% of Priorix-Tetra recipients, as opposed to 1.9% of children in the control group.