What is in this leaflet
Please read this leaflet carefully before you start using PROLASTIN® C (prō-‘lǎs-tĭn) LIQUID.
This leaflet answers some common questions about PROLASTIN® C LIQUID, a prescription medicine. It does not contain all the available information. It does not take the place of talking to your doctor.
All medicines have risks and benefits. Your doctor has weighed the risks of you using PROLASTIN® C LIQUID against the benefits they expect it will have for you.
If you have any concerns about using this medicine, ask your doctor.
Keep this leaflet with you. You may need to read it again.
What Prolastin® C Liquid is used for
PROLASTIN® C LIQUID is used to treat Alpha-1-proteinase inhibitor deficiency. This is a genetic disorder. It is characterised by very low amounts of your own Alpha-1-proteinase inhibitor protein in your lungs. Some patients develop a serious lung disease called emphysema.
PROLASTIN® C LIQUID contains human Alpha-1-proteinase inhibitor manufactured from blood products. You take PROLASTIN® C LIQUID to boost Alpha-1-proteinase inhibitor in your bloodstream and lungs.
One of the reasons your lungs have been damaged is because you lack enough of Alpha-1-proteinase inhibitor. Other causes include smoking and infections. Alpha-1-proteinase inhibitor inhibits enzymes that break down lung tissue. Be aware that there are no medical studies in people to prove that taking PROLASTIN® C LIQUID over a long period of time will stop or slow down the symptoms of emphysema.
Before you use Prolastin® C Liquid
When you must not use it
Do not use PROLASTIN® C LIQUID if you know you lack immunoglobulin A (IgA) in your bloodstream and your body has made its own antibodies against IgA. You could have a bad allergic reaction to PROLASTIN® C LIQUID because it contains trace amounts of IgA.
Smoking is not recommended during your treatment with PROLASTIN® C LIQUID.
If you are not sure whether you should use PROLASTIN® C LIQUID, talk to your doctor.
Your doctor should not administer PROLASTIN® C LIQUID if the carton seal has been broken.
Before you start to use it
Tell your doctor if:
- You are pregnant or intend to become pregnant. It is not known whether PROLASTIN® C LIQUID is harmful to an unborn baby when given to a pregnant woman.
- You are breastfeeding. Many medicines are excreted into the human breast milk.
- You do not tolerate medicines made from blood.
- You are concerned that your child may be treated with PROLASTIN® C LIQUID. The safety and effectiveness of PROLASTIN® C LIQUID in children have not been established.
- You are a smoker as the effectiveness of PROLASTIN® C LIQUID in smokers has not been established.
If you have not told your doctor about any of the above, tell your doctor before you are treated with PROLASTIN® C LIQUID.
Your body may not tolerate PROLASTIN® C LIQUID if you have the very rare condition of “IgA deficiency” and your body has already made its own antibodies against immunoglobulin A (IgA). If your doctor suspects that you are experiencing an allergic or anaphylactic reaction during administration of PROLASTIN® C LIQUID, your doctor must immediately discontinue the infusion.
Special safety warning
When medicines are made from human plasma, certain measures are put in place to prevent infections from viruses or the Creutzfeldt-Jakob disease (CJD) agent from being passed on to patients. These include
- Carefully selecting plasma donors to make sure those at risk of carrying infections are excluded.
- Testing of each donation and pools of plasma for signs of virus or virus infections.
- Including steps in the processing of the plasma that can inactivate or remove viruses.
Despite these measures, when medicines prepared from human plasma are administered, the possibility of passing on infection cannot be totally excluded. This applies to any unknown or emerging viruses or other types of infections.
Discuss the risks and benefits of this medicine with your doctor before using it.
Talk to your doctor if you become sick with a virus infection. Your doctor or you may want to report the infection to Grifols Australia Pty Ltd 1800 339 479.
Taking other medicines
Tell your doctor if you are taking any other medicines, including medicines that you buy without a prescription from your pharmacy, supermarket or health food shop. You should also tell any health professional who is prescribing a new medicine for you that you are using PROLASTIN® C LIQUID.
Your doctor has more information on medicines to be careful with or avoid while using PROLASTIN® C LIQUID. Interactions with other medicines are not known.
How to use Prolastin® C Liquid
How much to use
Your doctor will decide the amount of PROLASTIN® C LIQUID that is right for you. A standard dose is 60 milligrams of PROLASTIN® C LIQUID for every 1 kilogram of body weight. PROLASTIN® C LIQUID is administered into a vein with fluid. This is usually done in a health care facility. You will need to be observed for at least 20 minutes after the infusion.
When to use it
Your doctor will determine when your treatments should be given. The standard schedule is weekly. The infusion takes approximately 15 minutes.
How long to use it
You will use PROLASTIN® C LIQUID weekly until your doctor tells you otherwise.
If you forget to use it
If you miss a scheduled infusion of PROLASTIN® C LIQUID, talk to your doctor about rescheduling.
While you are using Prolastin® C Liquid
Things you must do
If you are about to be started on any new medicine tell your doctor that you are using PROLASTIN® C LIQUID.
Things you must not do
Do not give PROLASTIN® C LIQUID to anyone else, even if they have the same condition as you.
Things to be careful of
No effects on ability to drive and use machines have been observed with PROLASTIN® C LIQUID.
In case of overdose
If you use too much (overdose)
There have been no reported cases of overdose for PROLASTIN® C LIQUID. No data are available concerning overdose in humans.
Immediately telephone your doctor or the National Poisons Centre (telephone 0800 POISON or 0800 764 766), or go to accident and emergency at your nearest hospital, if you think that you or anyone else may have taken too much PROLASTIN® C LIQUID.
Do this even if there are no signs of discomfort or poisoning.
Side effects
Tell your doctor as soon as possible if you do not feel well after being treated with PROLASTIN® C LIQUID.
All medicines can have side effects. Sometimes they are serious, most of the time they are not. You may need medical treatment if you get some of the known adverse effects.
Ask your doctor to answer any questions you may have.
Tell your doctor if any of the following side effects happen during or soon after the infusion.
- Headache
- Dizziness
- Fatigue
- Shortness of breath (dyspnoea)
- Hives (urticaria) or rash
Occasionally, you may experience:
- Chills
- Chest discomfort or pain
- Out of sorts feeling (malaise)
- Itching (pruritus)
- Allergic or anaphylactic type reactions including shock
- Influenza-like illness
Rarely, you may experience:
- Racing heartbeat (tachycardia)
There is a possibility that your body could react to PROLASTIN® C LIQUID. Allergic reactions to PROLASTIN® C LIQUID have occurred. If you go into shock, your doctor will treat you by following standard guidelines for shock therapy.
Other adverse effects not listed above may also occur in some patients. Tell your doctor if you notice any other side effects or if any of the above side effects get serious.
Do not be alarmed by this list of possible adverse effects. You may not experience any of them.
After using Prolastin® C Liquid
Storage
Your doctor must store PROLASTIN® C LIQUID unopened vials refrigerated at 2°C to 8°C with no more than 1 month at room temperatures (up to 25°C) after which the product must be used or immediately discarded. PROLASTIN® C LIQUID should never be stored in a freezer. Your doctor must use PROLASTIN® C LIQUID on or before the expiry date stamped on the label. Do not use after the expiration date.
Your doctor must never use PROLASTIN® C LIQUID that was ever frozen, is cloudy, or discoloured. The solution is clear or slightly opalescent, colourless or pale yellow or pale green or pale brown. The solution may have a few particles in it.
All medicines, including PROLASTIN® C LIQUID, must be kept out of reach of children.
Disposal
Your doctor will dispose of any unused or expired medicine or medical waste in accordance with local requirements.
Medicines should not be disposed of into wastewater or household waste. Ask your doctor or pharmacist how to dispose of medicines no longer required. These measures will help to protect the environment.
Product description
What it looks like
PROLASTIN® C LIQUID is a clear or slightly opalescent, colourless or pale yellow or pale green or pale brown solution in a glass vial.
Ingredients
Active ingredient:
- Alpha-1-proteinase inhibitor
Inactive ingredients:
- Alanine
- monobasic sodium phosphate monohydrate
- water for injections
PROLASTIN® C LIQUID is sterile and does not contain an antimicrobial preservative.
Sponsor details
Grifols Australia Pty Ltd
Unit 5/80 Fairbank Road, Clayton South, VIC 3169
Australia
Australian Registration Number
PROLASTIN® C LIQUID
Alpha-1-proteinase inhibitor (human) 500 mg solution for injection for intravenous infusion vial: AUST R 371334
Alpha-1-proteinase inhibitor (human)1000 mg solution for injection for intravenous infusion vial: AUST R 305253
Alpha-1-proteinase inhibitor (human) 4000 mg solution for injection for intravenous infusion vial: AUST R 371339
Date of preparation
This leaflet was prepared on 27 May 2022
Published by MIMS September 2022

 A total of 23 COPD exacerbations were reported for a total of 18 individual subjects. Twelve subjects (12/32, 38%) during Prolastin C Liquid treatment experienced 13 COPD exacerbations, and 9 subjects (9/31, 29%) during Prolastin C treatment had 10 COPD exacerbations. Three COPD exacerbations occurred during the Follow-Up Period after Prolastin C Liquid treatment and 1 COPD exacerbation occurred in the Follow-up period after Prolastin C treatment. The overall rate of pulmonary exacerbations during treatment with either product was 1.9 exacerbations per subject-year. No exacerbation was considered to be serious, except for one event after Prolastin C treatment during the Follow-Up period (due to hospitalization).
A total of 23 COPD exacerbations were reported for a total of 18 individual subjects. Twelve subjects (12/32, 38%) during Prolastin C Liquid treatment experienced 13 COPD exacerbations, and 9 subjects (9/31, 29%) during Prolastin C treatment had 10 COPD exacerbations. Three COPD exacerbations occurred during the Follow-Up Period after Prolastin C Liquid treatment and 1 COPD exacerbation occurred in the Follow-up period after Prolastin C treatment. The overall rate of pulmonary exacerbations during treatment with either product was 1.9 exacerbations per subject-year. No exacerbation was considered to be serious, except for one event after Prolastin C treatment during the Follow-Up period (due to hospitalization).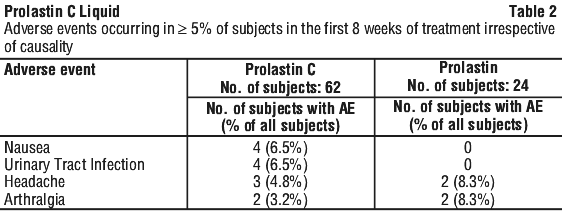 Table 3 displays the overall adverse event rate (> 0.5%), irrespective of causality, as a percentage of infusions received.
Table 3 displays the overall adverse event rate (> 0.5%), irrespective of causality, as a percentage of infusions received. Table 4 displays the overall rates of adverse events (≥ 5%), in the first eight weeks of treatment, that began during or within 72 hours of the end of an infusion of Prolastin C or Prolastin.
Table 4 displays the overall rates of adverse events (≥ 5%), in the first eight weeks of treatment, that began during or within 72 hours of the end of an infusion of Prolastin C or Prolastin. Ten exacerbations of chronic obstructive pulmonary disease were reported by 8 subjects in the 24 week pharmacokinetic crossover study. During the 16 week double-blind crossover phase, 4 subjects (17%) had a total of 4 exacerbations during Prolastin C treatment and 4 subjects (17%) had a total of 4 exacerbations during Prolastin treatment. Two additional exacerbations in 2 subjects (8%) occurred during the 8 week open-label treatment period with Prolastin C. The overall rate of pulmonary exacerbations during treatment with either product was 0.9 exacerbations per subject-year.
Ten exacerbations of chronic obstructive pulmonary disease were reported by 8 subjects in the 24 week pharmacokinetic crossover study. During the 16 week double-blind crossover phase, 4 subjects (17%) had a total of 4 exacerbations during Prolastin C treatment and 4 subjects (17%) had a total of 4 exacerbations during Prolastin treatment. Two additional exacerbations in 2 subjects (8%) occurred during the 8 week open-label treatment period with Prolastin C. The overall rate of pulmonary exacerbations during treatment with either product was 0.9 exacerbations per subject-year.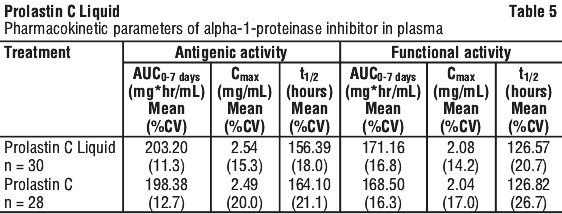 The key pharmacokinetic parameter was the area under the plasma concentration-time curve (AUC0-7 days) following 8 weeks of treatment with Prolastin C Liquid or Prolastin C. The 90% confidence interval (1.03-1.08) for the ratio of AUC0-7 days for Prolastin C Liquid and Prolastin C indicated that the 2 products are comparable, i.e. the entire range falls within the 0.80 - 125% interval.
The key pharmacokinetic parameter was the area under the plasma concentration-time curve (AUC0-7 days) following 8 weeks of treatment with Prolastin C Liquid or Prolastin C. The 90% confidence interval (1.03-1.08) for the ratio of AUC0-7 days for Prolastin C Liquid and Prolastin C indicated that the 2 products are comparable, i.e. the entire range falls within the 0.80 - 125% interval.