1 Name of Medicine
Edaravone.
2 Qualitative and Quantitative Composition
Each clear glass ampoule contains 30 mg of edaravone in 20 mL concentrated injection.
Excipient(s) with known effect.
Each ampoule contains sodium bisulfite.
For the full list of excipients, see Section 6.1 List of Excipients.3 Pharmaceutical Form
Concentrated solution for injection filled in a clear glass ampoule.
4.1 Therapeutic Indications
Radicava is indicated in adults with a diagnosis of amyotrophic lateral sclerosis who are independent in activities of daily living with normal respiratory function and where treatment is initiated within two years of disease onset.
Efficacy has not been demonstrated in patients outside of this defined population.
4.2 Dose and Method of Administration
Radicava concentrated injection must be diluted before use and is for intravenous infusion only. Promptly discontinue the infusion upon the first observation of any signs or symptoms consistent with a hypersensitivity reaction (see Section 4.4 Special Warnings and Precautions for Use).
The recommended dosage of Radicava is 60 mg of edaravone (two ampoules) diluted with 100 mL of 0.9% sodium chloride for infusion and administered as an intravenous infusion over a 60-minute period according to the following schedule:
An initial treatment cycle with daily dosing for 14 days, followed by a 14-day drug-free period.
Subsequent treatment cycles with daily dosing for 10 days out of 14-day periods, followed by 14-day drug-free periods.
Radicava is for single use in one patient only. Discard any residue.
To reduce microbiological hazard, use as soon as practicable after dilution. If storage is necessary, hold at 2°-8°C for not more than 24 hours.
Special populations.
No dosage adjustment is required in patients with hepatic impairment and mild and moderate renal impairment (estimated glomerular filtration rate (eGFR) > 30 mL/min/1.73 m2).
The effects of severe renal impairment on the pharmacokinetics of Radicava have not been studied, however exposure to edaravone is not expected to be significantly affected in patients with eGFR < 30 mL/min/1.73 m2 who do not require renal replacement therapy. Pharmacokinetics of Radicava have not been studied in patients undergoing renal replacement therapy and use of Radicava in this population is not recommended.4.3 Contraindications
Radicava is contraindicated in patients with a history of hypersensitivity to edaravone or any of the excipients listed in Section 6.1 List of Excipients.
4.4 Special Warnings and Precautions for Use
Hypersensitivity reactions.
Hypersensitivity reactions (redness, wheals, and erythema multiforme) and cases of anaphylaxis (urticaria, decreased blood pressure and dyspnoea) have been reported in spontaneous postmarketing reports with edaravone.
Patients should be monitored carefully for hypersensitivity reactions. If hypersensitivity reactions occur, discontinue Radicava, treat per standard of care, and monitor until the condition resolves (see Section 4.3 Contraindications).
Sulfite allergic reactions.
Radicava contains sodium bisulfite, a sulfite that may cause allergic type reactions, including anaphylactic symptoms and life-threatening or less severe asthmatic episodes in susceptible people.
The overall prevalence of sulfite sensitivity in the general population is unknown. Sulfite sensitivity occurs more frequently in asthmatic people.
Sodium content.
Each ampoule of Radicava contains 135 mg sodium chloride, 6.75 mg/mL. This should be taken into consideration by patients on a controlled sodium diet.
Use in the elderly.
Of the 184 patients with ALS who received Radicava in 3 placebo-controlled clinical trials, a total of 53 patients were 65 years of age and older, including 2 patients 75 years of age and older. No overall differences in safety or effectiveness were observed between these patients and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
Paediatric use.
Safety and effectiveness of Radicava in paediatric patients have not been established.
Effects on laboratory tests.
No data available.4.5 Interactions with Other Medicines and Other Forms of Interactions
The pharmacokinetics of edaravone is not expected to be significantly affected by inhibitors of CYP enzymes, or UGTs. The major metabolite of edaravone, edaravone sulfate, is a substrate for OAT1 and OAT3. Inhibitors of these transporters may alter exposures to edaravone sulphate.
In vitro studies demonstrated that, at the recommended clinical dose, edaravone and its metabolites are not expected to significantly inhibit cytochrome P450 enzymes (CYP1A2, CYP2B6, CYP2C8, CYP2C9, CYP2C19, CYP2D6, CYP3A4), uridine diphosphate glucuronosyltransferase (UGT) isoforms UGT1A1, UGT2B7, or the following transporters, P-gp, BCRP, OATP1B1, OATP1B3, OAT1 and OCT2 in humans. The major metabolite of edaravone, edaravone sulfate, is an inhibitor of OAT3. The clinical relevance of this is unknown.
Edaravone and its metabolites are not expected to induce CYP1A2, CYP2B6, or CYP3A4 at the clinical dose level of edaravone.
No interaction studies have been performed in vivo.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility.
There are no data on potential effects of Radicava on human fertility.
In rats, intravenous administration of edaravone (0, 3, 20, or 200 mg/kg) prior to and throughout mating in males and females and continuing in females to gestation day 7 had no effect on fertility; however, disruption of the oestrus cycle and mating behaviour was observed at the highest dose tested.
No effects on reproductive function were observed at the lower doses, which are up to 3 times the maximum recommended human dose (MRHD) of 60 mg, on a body surface area (mg/m2) basis.
(Category B3)
There are no adequate data on the developmental risk associated with the use of Radicava in pregnant women.
In rats, intravenous administration of edaravone throughout the period of organogenesis resulted in lower fetal weights at doses greater than or equal to 30 mg/kg/day. Maternal toxicity was also seen at these doses. A no effect dose for embryofetal developmental toxicity was less than the MRHD of 60 mg, on a body surface area (mg/m2) basis.
In rabbits, intravenous administration of edaravone throughout the period of organogenesis resulted in embryofetal death at 100 mg/kg/day, which was associated with maternal toxicity. The no effect dose for embryofetal developmental toxicity is approximately 6 times the MRHD on a body surface area (mg/m2) basis.
Women should avoid pregnancy while taking edaravone. If edaravone is used during pregnancy or if the patient becomes pregnant while taking edaravone, the patient should be apprised of the potential hazard to the fetus.
There are no data on the presence of edaravone in human milk, the effects on the breastfed infant, or the effects of the drug on milk production. However, edaravone and its metabolites were excreted at high levels in the milk of lactating rats. Because many drugs are excreted in human milk, caution should be exercised. The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for Radicava and any potential adverse effects on the breastfed infant from Radicava or from the underlying maternal condition.
In rats, intravenous administration of edaravone from the end of gestation throughout lactation resulted in an increased incidence of stillbirths and offspring mortality at 200 mg/kg/day, a dose at which maternotoxicity was observed. Additional findings in offspring included: increased locomotor activity in a behavioural study at maternal doses greater than or equal to 20 mg/kg/day; delayed physical development in females; and an increase in embryonic deaths in the next generation. The no effect dose for developmental toxicity is less than the MRHD on a body surface area (mg/m2) basis.4.7 Effects on Ability to Drive and Use Machines
Radicava has no influence on the ability to drive and use machines.
4.8 Adverse Effects (Undesirable Effects)
The tabulated incidence of adverse drug reactions from 3 Phase III clinical studies in patients with amyotrophic lateral sclerosis (ALS) in Japan are provided.
Frequencies are defined as: very common (≥ 1/10), common (≥ 1/100 to < 1/10), uncommon (≥ 1/1000 to < 1/100), rare (≥ 1/10,000 to < 1/1,000), very rare (< 1/10,000) and not known (cannot be estimated from the available data). Within each frequency grouping, adverse reactions are presented in order of decreasing seriousness. See Table 1.
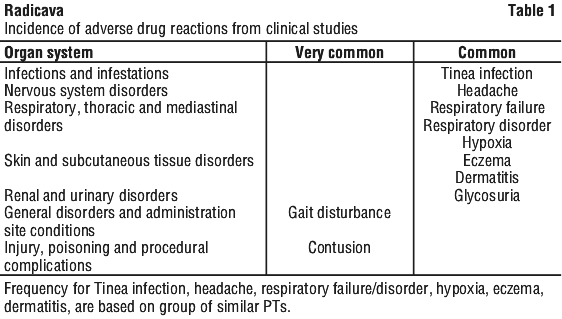 Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
In randomized, placebo-controlled trials, 184 ALS patients were administered Radicava 60 mg in treatment cycles for 6 months. The population consisted of Japanese patients who had a median age of 60 years (range 29-75) and were 59% male. Most (93%) of these patients were living independently at the time of screening.
Most common adverse events observed during clinical studies.
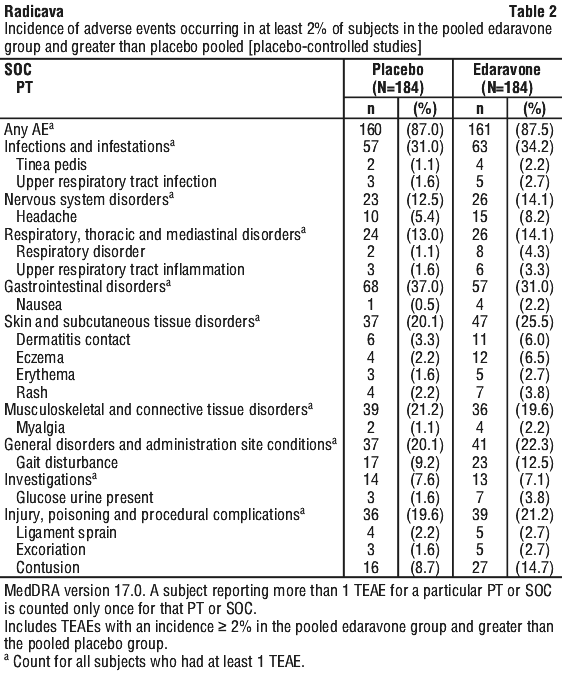
Table 2 summarizes the Adverse Events in randomised, placebo-controlled trials with an incidence ≥ 2% in the pooled edaravone group and at an incidence greater than placebo, by treatment group.
The most frequently reported adverse events with edavarone (≥ 3% and greater than placebo) were contusion (14.7% vs. 8.7%), gait disturbance (12.5% vs. 9.2%), headache (8.2% vs. 5.4%), eczema (6.5% vs. 2.2%), dermatitis contact (6.0% vs. 3.3%), respiratory disorder (4.3% vs. 1.1%), rash (3.8% vs. 2.2%), glucose urine present (3.8% vs. 1.6%), and upper respiratory tract inflammation (3.3% vs. 1.6%).
Postmarketing experience.
The following adverse reactions have been identified during post-approval use of Radicava. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Skin and subcutaneous tissue disorders.
Hypersensitivity reactions and anaphylaxis.
Reporting suspected adverse effects.
Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.4.9 Overdose
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action.
The mechanism by which Radicava exerts its therapeutic effect in patients with ALS is unknown. Edaravone is a free radical scavenger to reduce oxidative stress.
Clinical trials.
Study MCI186-19.
The efficacy of Radicava for the treatment of ALS was established in a 6-month, randomised, placebo-controlled, double-blind study conducted in Japanese patients with ALS who were living independently and met the following criteria at screening:
1. Functionality retained most activities of daily living (defined as scores of 2 points or better on each individual item of the ALS Functional Rating Scale - Revised [ALSFRS-R; described below]).
2. Normal respiratory function (defined as percent-predicted forced vital capacity values of [%FVC] ≥ 80%).
3. Definite or Probable ALS based on El Escorial revised criteria.
4. Disease duration of 2 years or less.
The study enrolled 69 patients in the Radicava arm and 68 in the placebo arm. Baseline characteristics were similar between these groups, with over 90% of patients in each group being treated with riluzole.
Radicava was administered as an intravenous infusion of 60 mg given over a 60-minute period according to the following schedule:
An initial treatment cycle with daily dosing for 14 days, followed by a 14-day drug-free period (Cycle 1).
Subsequent treatment cycles with daily dosing for 10 days out of 14-day periods, followed by 14-day drug-free periods (Cycles 2-6).
The primary efficacy endpoint was a comparison of the change between treatment arms in the ALSFRS-R total scores from baseline to Week 24.
The ALSFRS-R scale consists of 12 questions that evaluate the fine motor, gross motor, bulbar, and respiratory function of patients with ALS (speech, salivation, swallowing, handwriting, cutting food, dressing/hygiene, turning in bed, walking, climbing stairs, dyspnoea, orthopnea, and respiratory insufficiency). Each item is scored from 0-4, with higher scores representing greater functional ability.
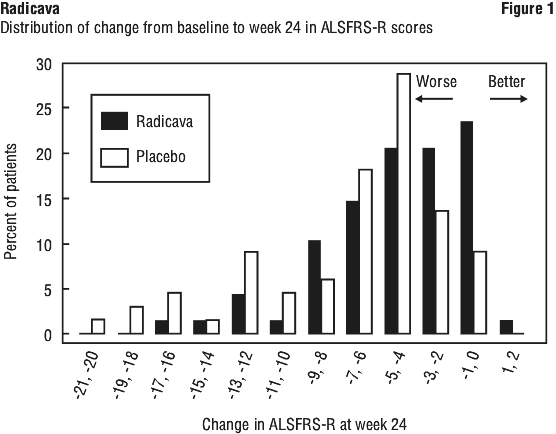
The decline in ALSFRS-R total scores from baseline was significantly less in the Radicava treated patients as compared to placebo (see Table 3). The distribution of change in ALSFRS-R scores from baseline to week 24 by percent of patients is shown in Figure 1.

 No benefit on survival was demonstrated.
No benefit on survival was demonstrated.
Study MCI186-16.
Study MCI186-16 was a randomised, double-blind, parallel-group, placebo-controlled Phase III trial to evaluate the efficacy and safety of 60 mg/day edaravone. The dosing schedule was the same as described for Study MCI186-19.
The primary endpoint was ALSFRS-R score change from Baseline in Cycle 1 to the end of Cycle 6 (24 weeks) or at discontinuation.
The main analyses were performed in the Full Set Analysis (FAS), which consisted of 101 subjects in the edaravone (E) group and 104 subjects in the placebo (P) group, for a total of 205 subjects.
Fourteen subjects in P group and 9 subjects in E group were discontinued before the end of Cycle 6.
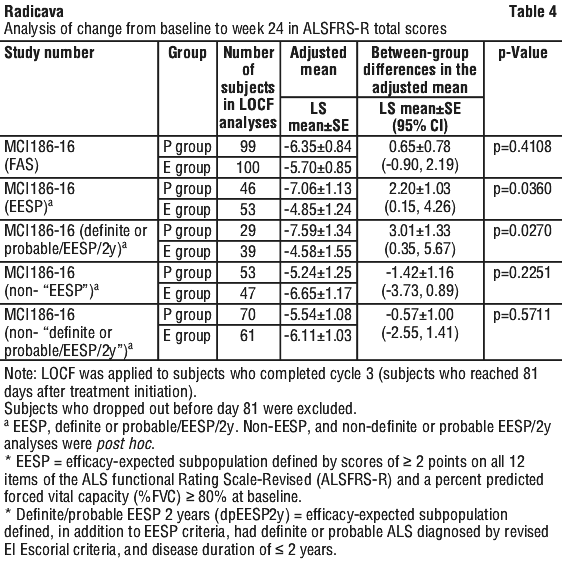
The results of the analysis of the primary efficacy endpoint of change from Baseline to the end of Cycle 6 in ALSFRS-R are shown in Table 4. While a beneficial trend favouring edaravone was observed in the FAS, the prespecified primary analyses did not statistically demonstrate the efficacy of edaravone in comparison to placebo.
In additional exploratory analyses to evaluate the beneficial trend observed with edaravone, the beneficial trend favouring edaravone was mainly driven by data from subjects who had functionality retained in most ADL domains with normal respiratory function and lived independently (see Table 4).
 No benefit on survival was demonstrated.
No benefit on survival was demonstrated.
5.2 Pharmacokinetic Properties
Absorption.
Radicava is administered by intravenous injection with the maximum plasma concentration (Cmax) of edaravone reached by the end of the infusion (60 mg edaravone for 60 min). No gender effect on edaravone pharmacokinetics has been found. There were no significant racial differences in Cmax and the area under the concentration-time curve (AUC) of edaravone between Japanese and Caucasian subjects. There was a trend of more than dose-proportional increase in AUC and Cmax of edaravone. With multiple-dose administration, edaravone does not accumulate in plasma.
Distribution.
Edaravone is bound to human serum proteins (92%), mainly to albumin, with no concentration dependence in the range of 0.1 to 50 micromol/L.
Metabolism.
Edaravone is metabolised to a sulfate conjugate and a glucuronide conjugate, which are not pharmacologically active. The glucuronide conjugation of edaravone involves multiple uridine diphosphate glucuronosyltransferase (UGT) isoforms (UGT1A6, UGT1A9, UGT2B7, and UGT2B17) in the liver and kidney. In human plasma, edaravone is mainly detected as the sulfate conjugate, which is presumed to be formed by sulfotransferases.
Excretion.
In Japanese and Caucasian healthy volunteers, edaravone was excreted mainly in the urine as its glucuronide conjugate form (70-90% of the dose). Approximately 5-10% of the dose was recovered in the urine as sulfate conjugate, and only 1% of the dose or less was recovered in the urine as unchanged form. In vitro studies suggest that the sulfate conjugate of edaravone is hydrolysed back to edaravone, which is then converted to the glucuronide conjugate in the human kidney before excretion into the urine.
The mean terminal elimination half-life of edaravone is 4.5 to 6 hours. The half-lives of its metabolites are 2 to 2.8 hours.
Special populations.
Paediatric population.
Safety and effectiveness of Radicava in paediatric patients have not been established.
Elderly.
No age effect on edaravone pharmacokinetics has been found.
Renal impairment.
Mild and moderate renal impairment have no clinically significant effects on the pharmacokinetics of edaravone. No dose adjustment is needed in these patients. No studies have been performed to characterize the pharmacokinetics of edaravone or establish its safety in patients with severe renal impairment.
Hepatic impairment.
Mild, moderate and severe hepatic impairment have no clinically significant effects on the pharmacokinetics of edaravone. No dose adjustment is needed in these patients.
5.3 Preclinical Safety Data
Genotoxicity.
Edaravone was negative in in vitro (bacterial reverse mutation and Chinese hamster lung chromosomal aberration) and in vivo (mouse micronucleus) assays.
Carcinogenicity.
Edaravone was not carcinogenic at oral doses up to 350 mg/kg/day in transgenic mice and oral doses up to 250 mg/kg/day in rats. Exposures or estimated exposures (AUC) at the highest tested doses were 22 and 133 times in mice and rats, respectively, the AUC at the MRHD of 60 mg.6 Pharmaceutical Particulars
6.1 List of Excipients
Sodium bisulfite, cysteine hydrochloride monohydrate, sodium chloride, sodium hydroxide, phosphoric acid and water for injections.
6.2 Incompatibilities
No incompatibilities between Radicava and commercially available infusion set materials have been observed.
Other medications should not be mixed with Radicava or injected into the infusion bag.
It is not recommended that the Radicava solution for infusion be mixed with total parenteral nutrition preparations and/or with amino-acid infusions before administration and should not be administered through the same intravenous line as those preparations.
It is not recommended that the Radicava solution for infusion be mixed with infusions of potassium canrenoate, or anticonvulsants including diazepam, phenytoin sodium because the solution may become cloudy.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 25°C.
6.5 Nature and Contents of Container
Radicava 30 mg/20 mL concentrated solution for injection is supplied as a clear, colourless, sterile solution in Type I clear glass ampoules and supplied in cartons containing 10 ampoules.
Pack size - 10 ampoules x 20 mL.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of in accordance with local requirements.
6.7 Physicochemical Properties
Edaravone is a white crystalline powder with a melting point of 129.7°C. It is freely soluble in acetic acid, methanol, or ethanol and slightly soluble in water or diethyl ether.
Chemical structure.
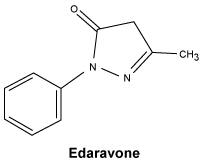 Chemical name - 3-methyl-1-phenyl-2-pyrazolin-5-one.
Chemical name - 3-methyl-1-phenyl-2-pyrazolin-5-one.
Molecular formula - C10H10N2O.
Molecular weight - 174.20.
CAS number.
89-25-8.7 Medicine Schedule (Poisons Standard)
S4 - Prescription only Medicine.
Summary Table of Changes


 Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.

 No benefit on survival was demonstrated.
No benefit on survival was demonstrated. No benefit on survival was demonstrated.
No benefit on survival was demonstrated. Chemical name - 3-methyl-1-phenyl-2-pyrazolin-5-one.
Chemical name - 3-methyl-1-phenyl-2-pyrazolin-5-one.