1 Name of Medicine
Medronic acid.
2 Qualitative and Quantitative Composition
Radpharm MDP Kit consists of sterile, pyrogen free, lyophilised ingredients which need reconstitution with sodium pertechnetate (99mTc) injection to produce a technetium [99mTc] medronate complex suitable for bone scanning. The precise structure of the technetium [99mTc] medronate complex is not known at this time.
Each vial contains the following active ingredient and excipient as a lyophilized powder:
Active ingredient.
10 mg of medronic acid.
Excipient.
1 mg of stannous chloride; 1 mg of ascorbic acid.
The product contains no preservatives.3 Pharmaceutical Form
Radpharm MDP is available as a dry white lyophilised powder in 10 mL vacuum sealed vials for intravenous injection.
4.1 Therapeutic Indications
Technetium [99mTc] medronate may be used as a skeletal imaging pharmaceutical.
4.2 Dose and Method of Administration
Recommended intravenous dose for the normal adult is 740 MBq.
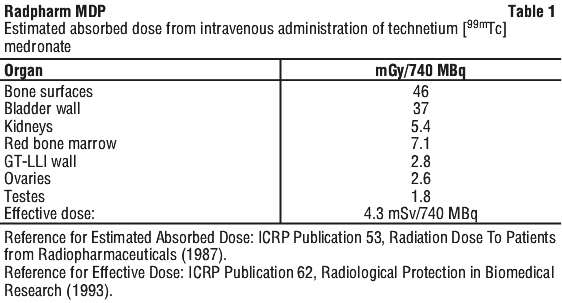
Radiation dosimetry.
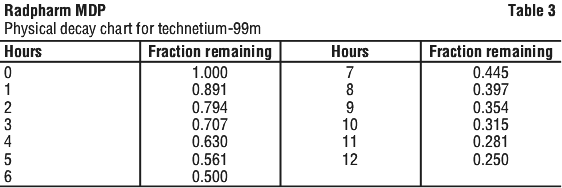
See Table 1.
Procedure.
Note.
If there is no vacuum, discard vial and do not deliver the sodium pertechnetate [99mTc] injection.
1. Place Radpharm MDP vial in a shielding container.
2. Draw a suitable volume (2 to 8 mL) of sodium pertechnetate [99mTc] injection eluted from a technetium-99m generator (1 to 16 GBq), and inject into the Radpharm MDP vial. Mix by inversion for 20 seconds and leave standing at room temperature for 10 minutes before use.
3. Determine the radioactivity per millilitre, label the container and calculate the patient dose.
4. The technetium [99mTc] medronate solution is stable at room temperature and may be used up to 8 hours after preparation.
Note.
In order to reduce the radiation dose to the bladder and other organs, the patient should be encouraged to drink and void as frequently as possible for a period of 4 to 6 hours after the administration of technetium [99mTc] medronate.4.3 Contraindications
None known.
4.4 Special Warnings and Precautions for Use
Radiopharmaceuticals should be used only by physicians who are qualified by specific training in the safe use and handling of radionuclides.
Contents of the vial are intended only for use in the preparation of technetium [99mTc] medronate.
The radioactivity of the dose should be checked with a suitable instrument immediately prior to administration.
Use in the elderly.
No data available.
Paediatric use.
No data available.
Effects on laboratory tests.
No data available.4.5 Interactions with Other Medicines and Other Forms of Interactions
None known.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility.
No data available.
(Category C)
Technetium-99m radiopharmaceuticals should only be given to a pregnant woman if in the judgement of the treating physician the expected benefits outweigh the potential hazards.
Technetium-99m is excreted in human milk. Interruption to breast feeding is not essential after the administration of a technetium [99mTc] medronate (a mother can be reassured by advising an interruption of 4 hr).
(Reference: L.K. Harding, A. Bossuyt, S. Pellet, C. Reiners, J.N. Talbot, "Recommendations for nuclear medicine physicians regarding breastfeeding mothers", Eur. J. Nucl. Med., 1995, 22, BP17).4.7 Effects on Ability to Drive and Use Machines
The effects of this medicine on a person's ability to drive and use machines were not assessed as part of its registration.
4.8 Adverse Effects (Undesirable Effects)
For each patient, exposure to ionising radiation must be justifiable on the basis of likely benefit. The activity administered must be such that the resulting dose is as low as reasonably achievable bearing in mind the need to obtain the intended diagnostic or therapeutic result.
Exposure to ionising radiation is linked with cancer induction and a potential for development of hereditary defects. For diagnostic nuclear medicine investigations the current evidence suggests that these adverse effects will occur with low frequency because of the low radiation doses incurred.
For most diagnostic investigations using a nuclear medicine procedure the radiation dose delivered (EDE) is less than 20 mSv. Higher doses may be justified in some clinical circumstances.
Occasionally (approximately 0.5 out of 100,000 investigations), hypersensitivity reactions may occur following intravenous administration of technetium [99mTc] medronate. Cases of local rash or generalised rash with itching and dermal irritation have been reported. Onset of the reaction is commonly several hours post-injection and it may last up to 48 hours. Treatment with a non-selective histamine H1 antagonist is helpful.
Reporting suspected adverse effects.
Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.4.9 Overdose
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.2 Pharmacokinetic Properties
After intravenous administration of technetium [99mTc] medronate, skeletal uptake appears to be related to bone metabolic activity and skeletal blood flow. Technetium [99mTc] medronate demonstrates specific affinity for areas of altered osteogenesis.
Localised areas of decreased bone uptake may occur after therapeutic external irradiation.
Blood clearance of technetium [99mTc] medronate is multiexponential with an initial fall in blood concentration with half-time (t1/2 = 3.5 minutes), followed by medium (t1/2 = 27 minutes) and slow (t1/2 = 144 minutes). The initial blood clearance represents the relatively rapid movement of technetium [99mTc] medronate from the vascular compartments to other compartments. The other clearance rates are functions of bone uptake and renal clearance and protein binding of tracer. Blood levels fall to 3-5% of the injected dose by 3 hours post injection.
5.3 Preclinical Safety Data
Genotoxicity.
No data available.
Carcinogenicity.
No data available.6 Pharmaceutical Particulars
6.1 List of Excipients
See Section 2 Qualitative and Quantitative Composition.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life of the Radpharm MDP kit can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
After reconstitution of Radpharm MDP with sodium pertechnetate [99mTc] injection, (1 to 16 GBq), the technetium [99mTc] medronate complex is stable at room temperature for 8 hours.
6.4 Special Precautions for Storage
The Radpharm MDP kit must be stored at 2°C to 8°C. (Refrigerate. Do not freeze.)
6.5 Nature and Contents of Container
Radpharm MDP kit is supplied as a carton of 10 sterile, pyrogen free, vacuum sealed multidose 10 mL glass Type 1 coloured vials with 1888 grey chlorobutyl lyophilisation stopper and aluminium seal with green flip off.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or radioactive waste material should be disposed off in accordance with Code for the disposal of radioactive wastes by the user which is published on the Australian Radiation Protection and Nuclear Safety Agency (ARPANSA) website as Radiation Protection Series (RPS C-6).
6.7 Physicochemical Properties
Chemical structure.
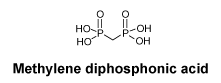
CAS number.
1984-15-2.7 Medicine Schedule (Poisons Standard)
Unscheduled.
Summary Table of Changes