Notes
Distributed by Clinect Pty Ltd
1 Name of Medicine
Cariprazine.
2 Qualitative and Quantitative Composition
Reagila contains cariprazine, a novel atypical antipsychotic agent. Each Reagila hard capsule contains cariprazine hydrochloride corresponding to 1.5 mg, 3 mg, 4.5 mg or 6 mg of cariprazine.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Hard capsule that is filled with white to yellowish white powder mixture.
Reagila 1.5 mg.
Hard gelatin capsule with white opaque cap and white opaque body imprinted with "GR 1.5" on the capsule body with black ink.
Reagila 3 mg.
Hard gelatin capsule with green opaque cap and white opaque body imprinted with "GR 3" on the capsule body with black ink.
Reagila 4.5 mg.
Hard gelatin capsule with green opaque cap and green opaque body imprinted with "GR 4.5" on the capsule body with white ink.
Reagila 6 mg.
Hard gelatin capsule with purple opaque cap and white opaque body imprinted with "GR 6" on the capsule body with black ink.4.1 Therapeutic Indications
Reagila is indicated for the treatment of schizophrenia in adult patients.
4.2 Dose and Method of Administration
Reagila hard capsules are intended for oral administration only.
Reagila is to be taken once daily at the same time of the day with or without food.
Alcohol should be avoided when taking cariprazine (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
The recommended starting dose of Reagila is 1.5 mg once daily. Thereafter the dose can be increased in 1.5 mg increments according to efficacy and tolerability to a maximum dose of 6 mg/day, if needed. The lowest effective dose should be maintained according to the clinical judgement of the treating physician.
Because of the long half-life of cariprazine and its active metabolites, changes in dose will not be fully reflected in plasma for several weeks. Patients should be monitored for adverse reactions and treatment response for several weeks after starting Reagila and after each dosage change (see Section 5.2 Pharmacokinetic Properties).
Switching from other antipsychotics to Reagila.
When switching from another antipsychotic to Reagila, gradual cross-titration should be considered with gradual discontinuation of the previous treatment while Reagila treatment is initiated.
Discontinuation of treatment.
Reagila can be stopped immediately. If discontinuing treatment with cariprazine, because of the long half-life of Reagila and its active metabolites, it may take 3 to 4 weeks for Reagila to be excreted from the body.
Missed dose.
If the patient misses a dose, the patient should take the missed dose as soon as possible. However, if it is almost time for the next dose, the missed dose should be skipped and the next dose should be taken according to the regular schedule. It is not recommended to take a double dose to make up for the forgotten dose.
Switching to another antipsychotic from Reagila.
When switching to another antipsychotic from Reagila, no gradual cross-titration is needed. The new antipsychotic should be initiated in its lowest dose while Reagila is discontinued.
Renal impairment.
No dose adjustment is required in patients with mild to moderate renal impairment (creatinine clearance (CrCl) ≥ 30 mL/min and < 89 mL/min). Safety and efficacy of cariprazine have not been evaluated in patients with severe renal impairment (CrCl < 30 mL/min). Use of Reagila is not recommended in patients with severe renal impairment (see Section 5.2 Pharmacokinetic Properties).
Hepatic impairment.
No dose adjustment is required in patients with mild to moderate hepatic impairment (Child-Pugh score between 5-9). Safety and efficacy of cariprazine have not been evaluated in patients with severe hepatic impairment (Child-Pugh score between 10 and 15). Use of Reagila is not recommended in patients with severe hepatic impairment (see Section 5.2 Pharmacokinetic Properties).
Elderly patients.
Available data in elderly patients aged ≥ 65 years treated with Reagila are not sufficient to determine whether or not they respond differently from younger patients (see Section 5.2 Pharmacokinetic Properties). Dose selection for an elderly patient should be more cautious.
Paediatric patients.
The safety and efficacy of Reagila in children and adolescents aged less than 18 years have not been established. No data are available.4.3 Contraindications
Reagila (cariprazine) is contraindicated in any patient with a known hypersensitivity to cariprazine or any component in the formulation (listed, see Section 6.1 List of Excipients).
Reagila is contraindicated with concomitant administration of strong or moderate CYP3A4 inhibitors (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions) and strong or moderate CYP3A4 inducers (see Section 4.5 Interactions
with Other Medicines and Other Forms of Interactions).
4.4 Special Warnings and Precautions for Use
Identified precautions.
Suicidal ideation and behaviour. The possibility of suicidality (suicidal ideation, suicide attempt and completed suicide) is inherent in psychotic illnesses and, generally, it is reported early after initiation or switch of antipsychotic therapy. Close supervision of high-risk patients should accompany antipsychotic therapy.
Akathisia, restlessness. Akathisia and restlessness is a frequently occurring adverse reaction of antipsychotics. Akathisia is a movement disorder characterised by a feeling of inner restlessness and a compelling need to be in constant motion, as well as by actions such as rocking while standing or sitting, lifting the feet as if marching on the spot, and crossing and uncrossing the legs while sitting. As Reagila causes akathisia and restlessness, it should be used cautiously in patients who are prone to or already exhibit symptoms of akathisia. Akathisia develops early in treatment. Therefore, close monitoring in the first phase of treatment is important. Prevention includes slow up-titration; treatment measures include slight down-titration of Reagila or anti-EPS medication. The dose can be modified based on individual response and tolerability (see Section 4.8 Adverse Effects (Undesirable Effects)).
Tardive dyskinesia. Tardive dyskinesia is a syndrome consisting of potentially irreversible, rhythmical, involuntary movements, predominantly of the tongue and/or face that can develop in patients treated with antipsychotics. If signs and symptoms of tardive dyskinesia appear in a patient treated with Reagila, discontinuation should be considered.
Parkinson's disease. If prescribed to patients with Parkinson's disease, antipsychotic medicinal products may exacerbate the underlying disease and worsen symptoms of Parkinson's disease. Physicians should, therefore, weigh the risks versus the benefits when prescribing Reagila to patients with Parkinson's disease.
Ocular symptoms/ cataract. In the preclinical studies of Reagila lens opacity/cataract was detected in dogs (see Section 4.8 Adverse Effects (Undesirable Effects); Section 5.3 Preclinical Safety Data). However, a causal relationship between lenticular changes/ cataracts observed in human studies and Reagila use has not been established. Nevertheless, patients who would develop symptoms potentially related to cataract should be advised to have an ophthalmologic examination and be re-evaluated for treatment continuation.
Neuroleptic malignant syndrome (NMS). A potentially fatal symptom complex referred to as NMS has been reported in association with antipsychotic treatment. Clinical manifestations of NMS are hyperpyrexia, muscle rigidity, elevated serum creatine phosphokinase levels, altered mental status and evidence of autonomic instability (irregular pulse or blood pressure, tachycardia, diaphoresis and cardiac dysrhythmia). Additional signs may include myoglobinuria (rhabdomyolysis) and acute renal failure. If a patient develops signs and symptoms indicative of NMS, or presents with unexplained high fever without additional clinical manifestations of NMS, Reagila must be discontinued immediately.
Seizures. Reagila should be used cautiously in patients with history of seizures or with conditions that potentially lower the seizure threshold.
Risk of cerebrovascular accidents (CVA). An approximately 3-fold increased risk of CVA has been seen in randomised placebo controlled clinical trials in the dementia population with some atypical antipsychotics. The mechanism for this increased risk is not known. An increased risk cannot be excluded for other antipsychotics or other patient populations. Reagila should be used with caution in patients with risk factors for stroke.
Cardiovascular disorders. Blood pressure changes.
Reagila can cause orthostatic hypotension as well as hypertension (see Section 4.8 Adverse Effects (Undesirable Effects)). Reagila should be used with caution in patients with known cardiovascular disease predisposing to blood pressure changes. Blood pressure should be monitored.
Electrocardiogram (ECG) changes.
QT prolongation can develop in patients treated with antipsychotics.
With Reagila no QT interval prolongation was detected compared to placebo in a clinical trial designed to assess QT prolongation (see Section 5.1 Pharmacodynamic Properties). In clinical trials, only a few, non-serious, QT-prolongations have been reported with Reagila (see Section 4.8 Adverse Effects (Undesirable Effects)). Therefore, Reagila should be used cautiously in patients with known cardiovascular disease or a family history of QT prolongation and in patients treated with medicinal products that might cause QT prolongation (see Section 5.1 Pharmacodynamic Properties).
Venous thromboembolism (VTE).
Cases of VTE have been reported with antipsychotic medicinal products. Since patients treated with antipsychotics often present with acquired risk factors for VTE, all possible risk factors for VTE should be identified before and during treatment with Reagila and preventive measures undertaken.
Hyperglycaemia and diabetes mellitus. Patients with an established diagnosis of diabetes mellitus or patients with risk factors for diabetes mellitus (e.g. obesity, family history of diabetes) who are starting treatment with atypical antipsychotics should be assessed for fasting plasma glucose before or soon after initiation of antipsychotic medication, and monitored periodically during long-term treatment. In clinical studies, glucose-related adverse reactions have been reported with Reagila (see Section 4.8 Adverse Effects (Undesirable Effects)).
In clinical studies with Reagila, changes from baseline to endpoint in fasting serum glucose for cariprazine were comparable to placebo.
Weight change. Weight gain has been observed with atypical antipsychotic use. Clinical monitoring of weight is recommended (see Section 4.8 Adverse Effects (Undesirable Effects)).
Excipients. Reagila 3 mg, 4.5 mg and 6 mg hard capsules contain Allura red AC (E 129), which may cause allergic reactions.
Use in the elderly.
Available data in elderly patients aged ≥ 65 years treated with Reagila are not sufficient to determine whether or not they respond differently from younger patients (see Section 5.2 Pharmacokinetic Properties).
Reagila has not been studied in elderly patients with dementia and is not recommended to treat elderly patients with dementia due to increased risk of overall mortality.
Paediatric use.
The safety and efficacy of Reagila in children and adolescents aged less than 18 years have not been established. No data are available.
Effects on laboratory tests.
The use of Reagila has been associated with increases in levels of liver enzymes and creatine phosphokinase and abnormal amounts of lipids (e.g. cholesterol and/or fat). Regular monitoring of lipids and liver function tests is recommended.
Elevated prolactin levels have been reported with other medicines that antagonise dopamine D2 receptors. In clinical trials, Reagila did not cause hyperprolactinaemia.
Undesirable alterations in lipids have been observed in patients treated with atypical antipsychotics. In clinical trials of Reagila, changes in metabolic profile and hyperlipidaemia, were comparable to placebo.4.5 Interactions with Other Medicines and Other Forms of Interactions
Potential for other medicinal products to affect Reagila.
Metabolism of cariprazine and its major active metabolites, desmethyl cariprazine (DCAR) and didesmethyl cariprazine (DDCAR), is mediated mainly by CYP3A4 with a minor contribution of CYP2D6.
CYP3A4 inhibitors.
Ketoconazole, a strong CYP3A4 inhibitor, caused two-fold increase in plasma exposure for total cariprazine (sum of cariprazine and its active metabolites) during short-term (4 days) co-administration, either if unbound or unbound + bound moieties considered.
Due to the long half-life of the active moieties of Reagila, a further increase in plasma exposure of total cariprazine can be expected during longer co-administration. Therefore, co-administration of Reagila with strong or moderate inhibitors of CYP3A4 (e.g. boceprevir, clarithromycin, cobicistat, indinavir, itraconazole, ketoconazole, nefazodone, nelfinavir, posaconazole, ritonavir, saquinavir, telaprevir, telithromycin, voriconazole, diltiazem, erythromycin, fluconazole, verapamil) is contraindicated (see Section 4.3 Contraindications).
Consumption of grapefruit juice should be avoided.
CYP3A4 inducers.
Co-administration of Reagila with strong and moderate inducers of CYP3A4 may result in a significant decrease in total cariprazine exposure, therefore the co-administration of Reagila and strong or moderate CYP3A4 inducers (e.g. carbamazepine, phenobarbital, phenytoin, rifampicin, St. John's wort (Hypericum perforatum), bosentan, efavirenz, etravirine, modafinil, nafcillin) is contraindicated (see Section 4.3 Contraindications).
CYP2D6 inhibitors.
CYP2D6 mediated pathway plays a minor role in the metabolism of Reagila, the major pathway is via CYP3A4 (see Section 5.2 Pharmacokinetic
Properties). Therefore, CYP2D6 inhibitors are unlikely to have a clinically relevant effect on Reagila metabolism.
P-gp, OATP1B1, OATP1B3 and BCRP.
Reagila and its major active metabolites are not substrates of P-glycoprotein (P-gp), the organic anion transporting polypeptide 1B1 and 1B3 (OATP1B1 and OATP1B3), or the breast cancer resistance protein (BCRP). This suggests that an interaction of cariprazine with inhibitors of P-gp, OATP1B1, OATP1B3 and BCRP is unlikely.
Potential for Reagila to affect other medicinal products.
P-glycoprotein (P-gp) substrates.
Cariprazine is a P-gp inhibitor in vitro at its theoretical maximum intestinal concentration. The clinical consequences of this effect are not fully understood, however the use of P-gp substrates with narrow therapeutic index such as dabigatran and digoxin could require extra monitoring and dose adjustment.
Cytochrome P450 (CYP450) substrates.
Based on the results of in vitro studies, cariprazine and its major metabolites are not expected to inhibit the metabolism of co-administered medicines that are substrates for CYP1A2, CYP2A6, CYP2B6, CYP2D6, CYP2C8, CYP2C9 or CYP3A4, CYP2C19 or CYP2E1.
Neither cariprazine, DCAR nor DDCAR showed potential to induce the activities of CYP1A2, CYP2B6 or CYP3A4.
Hormonal contraceptives.
In a drug interaction study, 28 days of treatment with cariprazine at 6 mg daily had no clinically relevant effect on the pharmacokinetics of oral contraceptives (ethinylestradiol and levonorgestrel).
Pharmacodynamic interactions.
Given the primary central nervous system effects of cariprazine, Reagila should be used with caution in combination with other centrally acting medicinal products and alcohol.4.6 Fertility, Pregnancy and Lactation
Effects on fertility.
The effect of Reagila on human fertility has not been evaluated.
Animal data.
Fertility was reduced in female rats given oral daily doses of 1 mg/kg (1.5 times the human dose in mg/m2) from 4 weeks prior to mating through gestation day 7. Reductions in conception indices were evident at 10 mg/kg/day (15 times the human dose in mg/m2).
Cariprazine had no effect on fertility in male rats given oral daily doses up to 10 mg/kg/day from 70 days prior to mating (up to 4 times the maximal recommended human dose (MRHD) of 6 mg/day based on AUC of total cariprazine, i.e. sum of AUC values of cariprazine, DCAR and DDCAR).
(Category D)
There are no or limited amount of data from the use of cariprazine in pregnant women.
Reagila is not recommended during pregnancy and in women of childbearing potential not using effective contraception. After discontinuation of Reagila treatment contraception should be used for at least 10 weeks due to the slow elimination of active moieties.
Non-teratogenic class effect.
Neonates exposed to antipsychotics (including cariprazine) during the third trimester of pregnancy are at risk of adverse reactions including extrapyramidal and/or withdrawal symptoms that may vary in severity and duration following delivery. There have been reports of agitation, hypertonia, hypotonia, tremor, somnolence, respiratory distress or feeding disorder. These complications have varied in severity; while in some cases symptoms have been self-limited, in other cases, neonates have required intensive care unit support and prolonged hospitalisation. Consequently, newborns should be monitored carefully.
Animal data.
Oral administration of cariprazine to rats at doses of 0.5 mg/kg/day and above during the period of organogenesis caused malformations, lower pup survival, and developmental delays at drug exposures 0.2 times the MRHD of 6 mg/day based on AUC of total cariprazine, i.e. sum of AUC values of cariprazine, DCAR and DDCAR.
Oral administration of cariprazine to pregnant rats during the period of organogenesis, throughout pregnancy and lactation at 1 mg/kg/day (0.4 times the MRHD based on AUC of total cariprazine) decreased postnatal survival, birth weight, and post-weaning body weight of first generation pups. In addition, pale, cold bodies and developmental delays (renal papillae not developed/underdeveloped and decreased auditory startle response in males) were observed in the absence of maternal toxicity. Reproductive performance of the first generation pups was unaffected; however, second generation pups also had similar clinical signs and lower body weight.
In rabbits, cariprazine caused maternal toxicity, but no fetal toxicity, developmental effects or malformations at oral doses of 5 mg/kg/day (exposures 5 times the clinical exposure at the MRHD based on AUC of total cariprazine).
Women of childbearing potential/contraception.
Women of childbearing potential must be advised to avoid pregnancy while on Reagila. Female patients of child-bearing potential must use highly effective contraceptive methods during treatment and for at least 10 weeks following the last dose of Reagila.
It is unknown whether Reagila or its major active metabolites are excreted in human milk. Cariprazine and its metabolites are excreted in milk of rats during lactation. A risk to the newborns/infants cannot be excluded. Breast-feeding should be discontinued during treatment with Reagila.4.7 Effects on Ability to Drive and Use Machines
Reagila has minor or moderate influence on the ability to drive and use machines. Patients should be cautioned about operating hazardous machinery, including motor vehicles, until they are reasonably certain that therapy with Reagila does not affect them adversely.
4.8 Adverse Effects (Undesirable Effects)
Reporting suspected adverse effects.
Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
Summary of the safety profile.
The most frequently reported adverse drug reactions (ADRs) with Reagila in the dose range (1.5-6 mg) were akathisia (19%) and parkinsonism (17.5%). Most events were mild to moderate in severity.
Tabulated list of adverse reactions.
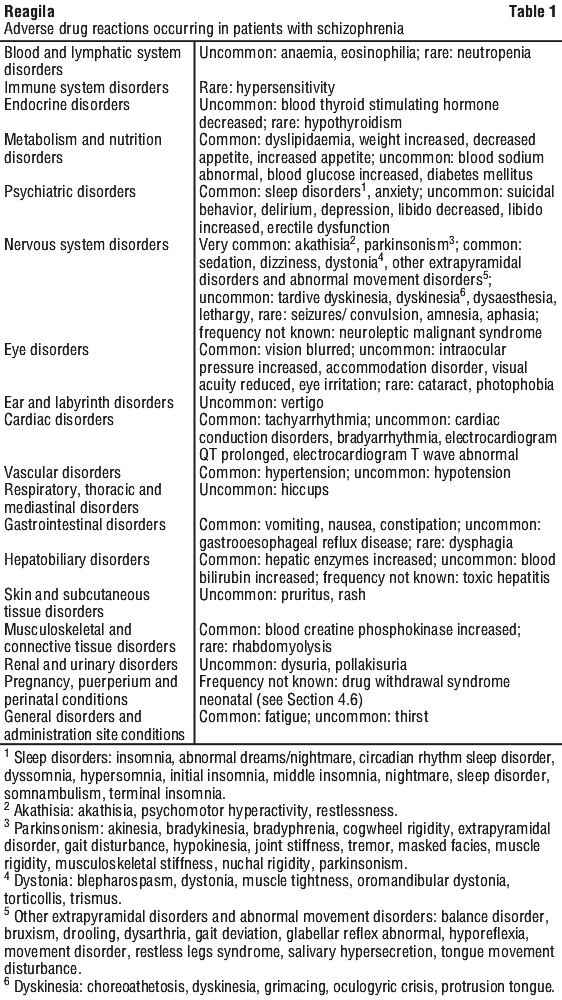
ADRs based upon pooled data from cariprazine schizophrenia studies are shown by system organ class and by preferred term (see Table 1).
Adverse reactions are ranked by frequency, the most frequent first, using the following convention: very common (≥ 1/10); common (≥ 1/100 to < 1/10); uncommon (≥ 1/1,000 to < 1/100); rare (≥ 1/10,000 to < 1/1,000); very rare (< 1/10,000); not known (cannot be estimated from the available data). Within each frequency grouping, adverse reactions are presented in order of decreasing seriousness.
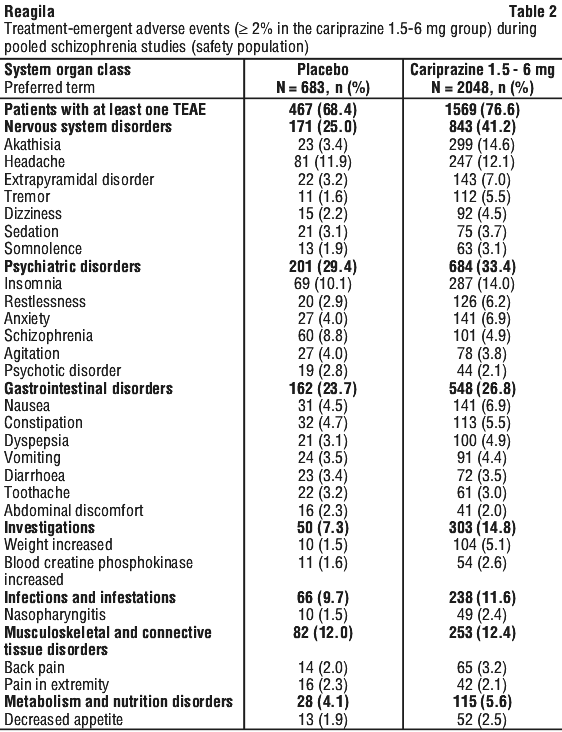
 Adverse events reported with the use of Reagila (incidence of 2% or greater) that occurred during pooled schizophrenia studies (safety population) are presented in Table 2.
Adverse events reported with the use of Reagila (incidence of 2% or greater) that occurred during pooled schizophrenia studies (safety population) are presented in Table 2.
Description of selected adverse reactions.
Lens opacity/ cataract.
Development of cataracts was observed in cariprazine non-clinical studies (see Section 5.3 Preclinical Safety Data). Therefore, cataract formation was closely monitored with slit lamp examinations in the clinical studies and patients with existing cataracts were excluded. During the schizophrenia clinical development program of cariprazine, few cataract cases were reported, characterised with minor lens opacities with no visual impairment (13/3192; 0.4%). Some of these patients had confounding factors. The most commonly reported ocular adverse event was blurred vision (placebo: 1/683; 0.1%, cariprazine: 22/2048; 1.1%).
Extrapyramidal symptoms (EPS).
In the short term studies EPS was observed in 27%; 11.5%; 30.7% and 15.1% of patients treated with cariprazine, placebo, risperidone and aripiprazole respectively. Akathisia was reported in 13.6%; 5.1%; 9.3% and 9.9% of patients treated with cariprazine, placebo, risperidone and aripiprazole, respectively. Parkinsonism was experienced in 13.6%; 5.7%; 22.1% and 5.3% of patients treated with cariprazine, placebo, risperidone and aripiprazole respectively. Dystonia was observed in 1.8%; 0.2%; 3.6% and 0.7% of patients on cariprazine, placebo, risperidone and aripiprazole, respectively.
In the placebo-controlled part of the long-term maintenance of effect study EPS was 13.7% of the cariprazine group compared to 3.0% of the placebo treated patients. Akathisia was reported in 3.9% of patients treated with cariprazine, versus 2.0% of the placebo group. Parkinsonism was experienced in 7.8% and 1.0% in cariprazine and placebo group, respectively.
In the negative symptom study EPS was reported in 14.3% of the cariprazine group and 11.7% of the risperidone treated patients. Akathisia was reported in 10.0% of patients treated with cariprazine and 5.2% of the risperidone group. Parkinsonism was experienced in 5.2% and 7.4% in cariprazine and risperidone treated patients respectively. Most EPS cases were mild to moderate in intensity and could be handled with common anti-EPS medicinal products. The rate of discontinuation due to EPS related ADRs was low.
Venous thromboembolism (VTE).
Cases of VTE, including cases of pulmonary embolism and cases of deep vein thrombosis have been reported with antipsychotics - Frequency unknown.
Elevated liver transaminases.
Elevated liver transaminases (Alanine Aminotransferase [ALT], Aspartate Aminotransferase [AST]) are frequently observed with antipsychotic treatment. In the cariprazine clinical studies, the incidence of ALT and AST elevation ADRs occurred in 2.2% of cariprazine-, 1.6% of risperidone- and 0.4% of placebo-treated patients. None of the cariprazine-treated patients had any liver damage.
Weight changes.
In the short-term studies, within the recommended dose range of 1.5-6 mg/day there were slightly greater mean increases in body weight in the cariprazine group compared to the placebo group: 1 kg and 0.3 kg, respectively. The proportion of patients with potentially clinically significant (PCS) weight gain (defined as increase ≥ 7%) was 7.6 and 7.7% for the 1.5-3 and 4.5-6 mg/day cariprazine groups respectively, compared with 16.7% in the risperidone 4 mg group, 6.0% in the aripiprazole 10 mg group and 4.7% in the placebo group.
In the long term maintenance of effect study, there was no clinically relevant difference in change of body weight from baseline to end of treatment in the total study population (1.1 kg for cariprazine 3-9 mg and 0.9 kg for placebo). At the dose range of 3-6 mg/day cariprazine, during the open-label phase of the study during 20 weeks cariprazine treatment, 9.0% of patients developed PCS weight gain, while during the double-blind phase, 9.8% of the patients who continued with cariprazine treatment had PCS weight gain versus 7.1% of the patients who were randomised to placebo after the 20 week open-label cariprazine treatment.
In the negative symptom study, the mean change of body weight was -0.3 kg for cariprazine and +0.6 kg for risperidone and PCS weight gain was observed in 6% of the cariprazine group while 7.4% of the risperidone group.
QT-prolongation.
With cariprazine no QT interval prolongation was detected compared to placebo in a clinical study designed to assess QT prolongation (see Section 5.1 Pharmacodynamic Properties). In other clinical studies, only a few, non-serious, QT-prolongations have been reported with cariprazine. During the long-term, open-label treatment period, 3 patients (0.4%) had QTcB > 500 msec, one of whom also had QTcF > 500 msec. A > 60 msec increase from baseline was observed in 7 patients (1%) for QTcB and in 2 patients (0.3%) for QTcF. In the long-term, maintenance of effect study, during the open-label phase, > 60 msec increase of from baseline was observed in 12 patients (1.6%) for QTcB and in 4 patients (0.5%) for QTcF. During the double-blind treatment period, > 60 msec increases from baseline in QTcB were observed in 3 cariprazine-treated patients (3.1%) and 2 placebo-treated patients (2%).
Post-marketing experience.
The following adverse reaction has been identified with unknown frequency during post approval use of cariprazine:
Skin and subcutaneous tissue disorders.
Stevens-Johnson syndrome.4.9 Overdose
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
Symptoms.
Accidental acute overdose (48 mg/day) was reported in one patient. This patient experienced orthostasis and sedation. The patient fully recovered the same day.
Management of overdose.
There is no specific antidote to Reagila, therefore, management of overdose should concentrate on supportive therapy including maintenance of an adequate airway, oxygenation and ventilation and management of symptoms.
Cardiovascular monitoring should commence immediately, including continuous electrocardiographic monitoring for possible arrhythmias. In case of severe extrapyramidal symptoms, anticholinergic medicinal products should be administered. Since Reagila is highly bound to plasma proteins, haemodialysis is unlikely to be useful in the management of overdose. Close medical supervision and monitoring should continue until the patient recovers.5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Pharmacotherapeutic group: Psycholeptics, other antipsychotics, ATC code: N05AX15.
Reagila is an atypical antipsychotic. It is an orally active and potent dopamine D3/D2 receptor partial agonist with preferential binding to D3 receptors and partial agonist at serotonin 5-HT1A receptors.
Mechanism of action.
The mechanism of action of cariprazine, as with other medicines having efficacy in schizophrenia, is not fully understood. However, the therapeutic effect of cariprazine may be mediated through a combination of partial agonist activity at dopamine D3, D2L and D2S receptors and serotonin 5-HT1A receptors, and antagonist activity at serotonin 5-HT2B, 5-HT2A and histamine H1 receptors.
In vitro receptor binding studies revealed that cariprazine binds with high affinity (Ki < 1 nanoM) at dopamine D3 (Ki = 0.085-0.3 nanoM), D2L (Ki = 0.49-0.71 nanoM), D2S (Ki = 0.69 nanoM) (i.e. cariprazine displays 2-8 fold selectivity for D3 over D2 receptors) and serotonin 5-HT2B (Ki values of 0.58-1.1 nanoM) receptors, and with medium affinity (1 nanoM < Ki < 20 nanoM) at serotonin 5-HT1A (Ki = 1.4-2.6 nanoM) and 5-HT2A (Ki = 18.8 nanoM) receptors.
Cariprazine has low affinity (20 nanoM < Ki < 200 nanoM) for histamine H1 receptors (Ki = 23.3 nanoM), serotonin 5-HT2C (Ki = 134 nanoM) and adrenergic α1 receptors (Ki = 155 nanoM).
Cariprazine has no appreciable affinity for cholinergic muscarinic receptors (IC50 > 1000 nanoM). The two major active metabolites, desmethyl cariprazine and didesmethyl cariprazine have a similar in vitro receptor binding and functional activity profile as the parent active substance.
Pharmacodynamic effects.
In vivo non-clinical studies demonstrated that cariprazine occupies D3 receptors to a similar extent as D2 receptors at pharmacologically effective doses. There was a dose-dependent occupancy of brain dopamine D3 and D2 receptors (with preferential occupancy in regions with higher D3 expression) in patients with schizophrenia within the therapeutic dose range of cariprazine for 15 days.
The effects of cariprazine on the QT interval were evaluated in patients with schizophrenia or schizoaffective disorder. Holter monitor-derived electrocardiographic assessments were obtained in 129 patients over a twelve-hour period at baseline and steady state. No QT interval prolongation was detected following supratherapeutic doses (9 mg/day or 18 mg/day). No patients treated with cariprazine experienced QTc increases ≥ 60 msec from baseline, nor did any patient experience a QTc of > 500 msec in the study.
Clinical trials.
Efficacy with short-term use.
The efficacy of Reagila for the treatment of acute schizophrenia was studied in three multi-centre, multinational, randomised, double-blind, placebo-controlled 6-week studies including 1,754 patients with the age of 18 to 60 years. The primary endpoint was change from baseline to week 6 in the Positive and Negative Syndrome Scale (PANSS) total score and the secondary endpoint was change from baseline to week 6 in the Clinical Global Impressions-Severity (CGI-S) score in all acute schizophrenia studies.
In a multinational placebo controlled study using fixed doses of 1.5 mg, 3.0 mg and 4.5 mg Reagila and 4.0 mg risperidone for assay sensitivity, all Reagila doses and the active-control showed statistically significant improvement in both primary as well as secondary endpoint compared to placebo.
In another multinational placebo controlled study using fixed doses of 3.0 mg, and 6.0 mg Reagila and 10 mg aripiprazole for assay sensitivity, both cariprazine doses and the active-control showed statistically significant improvement in both primary as well as secondary endpoint compared to placebo.
In a third multinational placebo controlled study using fixed/flexible doses of 3.0-6.0 mg and 6.0-9.0 mg Reagila, both Reagila doses groups showed statistically significant improvement in both primary as well as secondary endpoint compared to placebo.
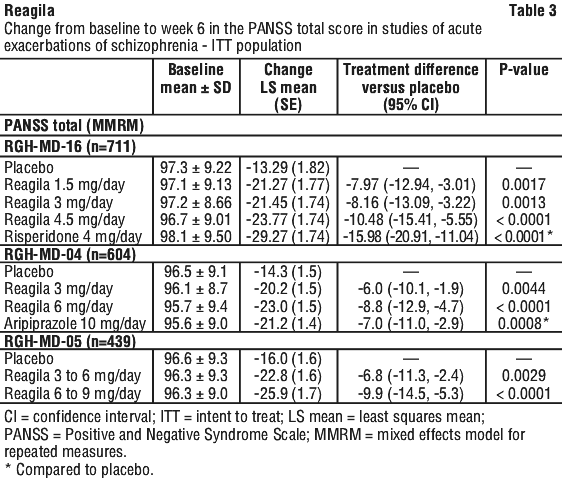
Results for the primary outcome parameter, analysed using mixed effects model for repeated measures (MMRM), are summarised in Table 3: Change from baseline to week 6 in the PANSS total score in studies of acute exacerbations of schizophrenia-ITT population and are comparable to analysis using analysis of covariance (ANCOVA) with last observation carried forward (LOCF). Results for the secondary outcome parameter (CGI) and additional endpoints were supportive of the primary endpoint.
Efficacy with long-term use.
The efficacy of Reagila for maintaining antipsychotic effect was investigated in a randomized withdrawal, long-term clinical study. In total, 751 patients with acute symptoms of schizophrenia were treated with Reagila 3-9 mg/d during 20-week open-label treatment consisting of an 8-week, flexible-dose run-in phase and a 12-week fixed-dose stabilization phase. 337 of these patients received Reagila in the dose-range of 3 or 6 mg/day.
Stable patients who completed open-label treatment could be randomised to continue Reagila (3, 6, or 9 mg/d) or placebo for double-blind treatment (up to 72 weeks). A total of 264 patients completed open label treatment; 200 eligible patients were randomised to double-blind placebo (n=99) or Reagila (n=101). The primary efficacy parameter was time to relapse (worsening of symptom scores, psychiatric hospitalisation, aggressive/ violent behaviour, or suicidal risk) in the pooled Reagila group (3-9 mg) vs placebo.
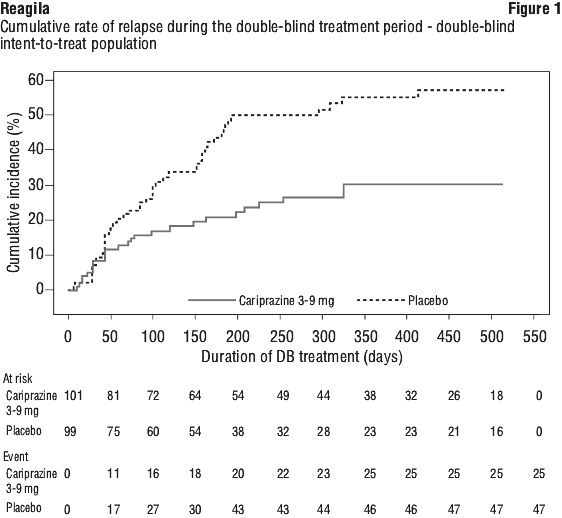
By the end of the double-blind treatment, time to relapse was significantly longer in the Reagila group (3-9 mg) than in the placebo group (p=0.0010). The 25th percentile for time to relapse was 92 days in the placebo group, compared to 224 days in the Reagila group. 47.5% (47/99) of placebo-treated patients and 24.8% (25/101) of Reagila-treated patients had a relapse of schizophrenia symptoms. The hazard of relapse for Reagila-treated patients was estimated to be less than half that of placebo-treated patients (hazard ratio [95% CI] = 0.45 [0.28, 0.73]) (Figure 1). For Reagila 3-6 mg, time to relapse was also significantly longer in the Reagila group than in the placebo group (p=0.0091). The 25th percentile for time to relapse was 92 days in the placebo group, compared to 326 days in the Reagila group. 49.0% (25/51) of placebo-treated patients versus 21.6% (11/51) of Reagila-treated patients had a relapse of schizophrenic symptoms (hazard ratio [95% CI] = 0.40 [0.20, 0.82]) (Figure 2).
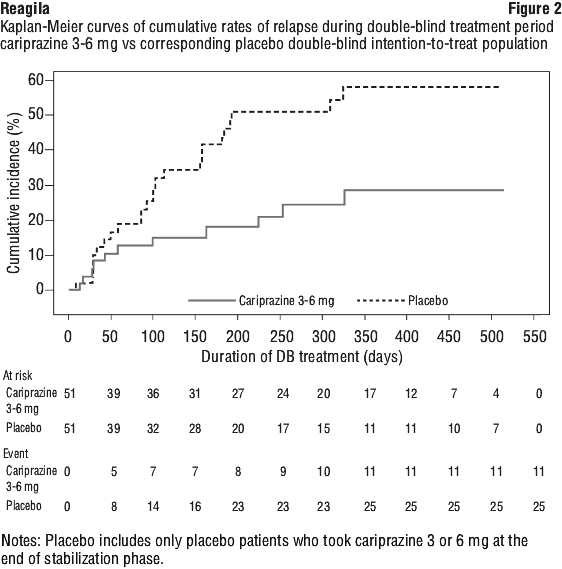
Efficacy in predominantly negative symptoms of schizophrenia.
The efficacy of Reagila for the treatment of predominantly negative symptoms of schizophrenia was investigated in a 26-week, multi-centre, double-blind, and active-controlled clinical study. Reagila (dose range 3-6 mg, target dose 4.5 mg) was investigated compared to risperidone (dose range 3-6 mg, target dose 4 mg) in patients with persistent, predominant negative symptoms of schizophrenia (n=461). 86% of patients were less than 55 years old, 54% of them were male.
Persistent predominant negative symptoms were defined as symptoms lasting for a period of at least 6 months with high level of negative symptoms and low level of positive symptoms [(PANSS factor score for negative symptoms ≥ 24, a score of ≥ 4 on a minimum 2 of the 3 PANSS items (N1: flat affect, N4: avolition, and N6: poverty of speech) and PANSS factor score for positive symptoms ≤ 19]. Patients with secondary negative symptoms, such as moderate to severe depressive symptoms and clinically relevant parkinsonism (EPS) were excluded.
Both Reagila- and risperidone-treated patient groups have shown statistically significant improvement in the change from baseline for the primary efficacy parameter, PANSS factor score for negative symptoms (PANSS-FSNS) (p = 0.002). However, a statistically significant difference (p = 0.008) in favour of Reagila over risperidone was observed from Week 14 onward (see Table 4: Summary of results in study RGH-188-005 (MMRM analysis)).
Both Reagila- and risperidone-treated patient groups have shown statistically significant improvement in the change from baseline for the secondary efficacy parameter, Personal and Social Performance (PSP) total score (p < 0.001). However, a statistically significant difference (p = 0.005) in favour of Reagila over risperidone was observed from Week 10 onward (see Table 4: Summary of results in study RGH-188-005 (MMRM analysis)).
Differences on the Clinical Global Impression Severity (p = 0.005) and Improvement (p < 0.001) scales, as well as PANSS-FSNS response rates (PANSS FSNS ≥ 30% improvement at Week 26; p = 0.003) were supportive of findings on the primary and secondary efficacy parameters. The results were analysed using MMRM and were consistent with the analysis using ANCOVA with LOCF.

5.2 Pharmacokinetic Properties
Cariprazine has two pharmacologically active metabolites with similar activities as cariprazine, desmethyl cariprazine (DCAR) and didesmethyl cariprazine (DDCAR). Total cariprazine (sum of cariprazine + DCAR and DDCAR) exposure approaches 50% of steady state exposure in ~1 week of daily dosing while 90% of steady state is achieved in 3 weeks. At steady state, exposure to DDCAR is approximately two to three-fold higher than to cariprazine, and exposure to DCAR is approximately 30% of cariprazine exposure.
Absorption.
Absolute bioavailability of Reagila is unknown. Reagila is well absorbed after oral administration. Following multiple-dose administration, peak plasma concentrations for cariprazine and the major active metabolites generally occur at approximately 3-8 hours post dose.
Administration of a single dose of 1.5 mg Reagila with a high-fat meal (900 to 1,000 calories) did not significantly affect the Cmax or AUC of cariprazine (AUC0-∞ increased by 12%, Cmax decreased by < 5% under fed condition versus fasting). The effect of food on the exposure of the metabolites DCAR and DDCAR was also minimal.
Reagila can be administered with or without food.
Distribution.
Based on a population pharmacokinetic analysis, the apparent volume of distribution (V/F) was 916 L for cariprazine, 475 L for DCAR and 1,568 L for DDCAR, indicating extensive distribution of cariprazine and its major active metabolites. Cariprazine and its major active metabolites are highly bound (96 to 97% for CAR, 94% to 96% for DCAR and 92% to 94% for DDCAR) to plasma proteins.
Metabolism.
The metabolism of cariprazine involves demethylation (DCAR and DDCAR), hydroxylation (hydroxy cariprazine, HCAR) and a combination of demethylation and hydroxylation (hydroxy desmethyl cariprazine, HDCAR and hydroxy didesmethyl cariprazine, HDDCAR). The metabolites of HCAR, HDCAR, and HDDCAR are subsequently biotransformed to their corresponding sulfate and glucuronide conjugates. An additional metabolite, desdichlorophenyl piperazine cariprazine (DDCPPCAR) acid, is produced by dealkylation and subsequent oxidation of cariprazine.
Reagila is metabolised by CYP3A4 and, to a lesser extent, by CYP2D6, to DCAR and HCAR. DCAR is further metabolised by CYP3A4 and to a lesser extent by CYP2D6 into DDCAR and HDCAR. DDCAR is further metabolised to HDDCAR by CYP3A4.
Excretion.
Elimination of cariprazine and its major active metabolites is mainly through hepatic metabolism. Following administration of 12.5 mg/day Reagila to patients with schizophrenia, 20.8% of the dose was excreted in urine as cariprazine and its metabolites.
Unchanged cariprazine is excreted by 1.2% of the dose in urine and 3.7% of the dose in feces.
The mean terminal half-life (1 to 3 days for cariprazine and DCAR and 13 to 19 days for DDCAR) is not predictive of time to reach steady state or plasma concentration decline after treatment discontinuation. For the management of patients treated with Reagila, the effective half-life is more relevant than the terminal half-life. The effective (functional) half-life is ~2 days for cariprazine and DCAR, 8 days for DDCAR and is ~1 week for total cariprazine. The plasma concentration of total cariprazine will gradually decline following dose discontinuation or interruption. The plasma concentration of total cariprazine decreases by 50% in ~1 week and greater than 90% decline in total cariprazine concentration occurs in ~3 weeks.
Linearity.
After repeated administration plasma exposure of cariprazine and its two major active metabolites, desmethyl cariprazine (DCAR) and didesmethyl cariprazine (DDCAR), increases proportionally over the therapeutic dose range of 1.5 to 6 mg.
Special populations.
Renal impairment.
Population pharmacokinetic modelling was performed using data from patients enrolled in the schizophrenia cariprazine clinical program with differing levels of renal function, including normal renal function (creatinine clearance (CrCl) ≥ 90 mL/min), as well as mild (CrCl 60 to 89 mL/min) and moderate (CrCl 30 to 59 mL/min) renal impairment. No significant relationship was found between cariprazine plasma clearance and creatinine clearance.
Reagila has not been evaluated in patients with severe (CrCl < 30 mL/min) renal impairment (see Section 4.2 Dose and Method of Administration).
Hepatic impairment.
A 2-part study (a single dose of 1 mg Reagila [Part A] and a daily dose of 0.5 mg Reagila for 14 days [Part B]) was conducted in patients with varying degrees of impaired hepatic function (Child-Pugh Classes A and B). Compared to healthy subjects, patients with either mild or moderate hepatic impairment had up to approximately 25% higher exposure (Cmax and AUC) for cariprazine and up to approximately 45% lower exposure for the major active metabolites, desmethyl cariprazine and didesmethyl cariprazine, following the single dose of 1 mg Reagila or 0.5 mg Reagila for 14 days.
The total active moiety (CAR+DCAR+DDCAR) exposure (AUC and Cmax) decreased by 21-22% and 13-15% in mild or moderate hepatic impairment (HI), respectively, compared to healthy subjects if unbound + bound concentrations were considered, while for unbound total moiety a decrease of 12-13% and an increase of 20-25% were calculated in mild HI patients and in moderate HI patients, respectively, after multiple dosing of Reagila.
Reagila has not been evaluated in patients with severe hepatic impairment (Child-Pugh Class C) (see Section 4.2 Dose and Method of Administration).
Age, gender and race.
In the population PK analysis there were no clinically relevant differences in the PK parameters (AUC and Cmax of the sum of cariprazine and its major active metabolites) based on age, gender and race.
This analysis included 2,844 patients of different races, involving 536 patients between the ages of 50 and 65. Of the 2,844 patients 933 were female (see Section 4.2 Dose and Method of Administration). In elderly patients above 65 years of age data are limited.
Smoking status.
Because cariprazine is not a substrate for CYP1A2, smoking is not expected to have an effect on the pharmacokinetics of cariprazine.
Potential for Reagila to affect other medicinal products.
Cariprazine and its major active metabolites did not induce CYP1A2, CYP2B6 and CYP3A4 enzymes and were not inhibitors of CYP1A2, CYP2A6, CYP2B6, CYP2C8, CYP2C9, CYP219, CYP2D6, CYP2E1 and CYP3A4 in vitro. Cariprazine and its major active metabolites are not inhibitors of transporters OATP1B1, OATP1B3, BCRP, organic cation transporter 2 (OCT2), and organic anion transporters 1 and 3 (OAT1 and OAT3) in vitro. DCAR and DDCAR were not inhibitors of transporter P-gp although cariprazine could be a P-gp inhibitor in the intestine (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
5.3 Preclinical Safety Data
Genotoxicity.
Cariprazine was not mutagenic in the in vitro bacterial reverse mutation assay, nor clastogenic in the in vitro human lymphocyte chromosomal aberration assay or in the in vivo mouse bone marrow micronucleus assay. However, cariprazine increased the mutation frequency in the in vitro mouse lymphoma assay under conditions of metabolic activation at cytotoxic concentrations.
The major human metabolite DDCAR was not mutagenic in the in vitro bacterial reverse mutation assay, but induced a small increase in chromosomal aberrations in the in vitro human lymphocyte chromosomal aberration assay at cytotoxic concentrations in the presence of metabolic activation.
Based on the weight of evidence, cariprazine and its major metabolite DDCAR have negligible genotoxic potential.
Carcinogenicity.
There was no increase in the incidence of tumours following daily oral administration of cariprazine to rats for 2 years and to Tg.rasH2 mice for 6 months at doses that are up to 4 and 19 times respectively, the MRHD based on AUC of total cariprazine, (i.e. sum of AUC values of cariprazine, DCAR and DDCAR).
Rats were administered cariprazine at oral doses of 0.25, 0.75, and 2.5 (males)/ 1, 2.5, and 7.5 mg/kg/day (females) which are 0.2 to 1.8 (males)/ 0.8 to 4.1 (females) times the MRHD based on AUC of total cariprazine.
Tg.rasH2 mice were administered cariprazine at oral doses of 1, 5, and 15 (males)/ 5, 15, and 50 mg/kg/day (females) which are 0.6 to 7.9 (males)/ 2.6 to 19 (females) times the MRHD based on AUC of total cariprazine.
Animal toxicology.
Retinal pathology.
Reagila caused bilateral cataract and secondary retinal changes (retinal detachment and cystic degeneration) in the dog. The exposure (AUC of total cariprazine) at the no-observed-adverse-effect-level (NOAEL) for ocular toxicity (2 mg/kg/day) was 4.2-fold the clinical AUC exposure for total cariprazine at the MRHD. Increased incidence and severity of retinal degeneration/atrophy was observed in albino rats in the 2-year study at 0.75 mg/kg/day (0.6 times the clinical AUC exposure for total cariprazine at the MRHD). Cataracts were not observed in other repeat dose studies in pigmented mice or albino rats.
Phospholipidosis.
Phospholipidosis was observed in the lungs of rats, dogs, and mice (with or without inflammation) and in the adrenal gland cortex of dogs at clinically relevant exposures. Inflammation was observed in the lungs of dogs dosed for 1 year with a NOAEL of 1 mg/kg/day (approximately 2 times the MRHD based on AUC of total cariprazine). No inflammation was observed at the end of 2-month drug-free period at an exposure 4 times the clinical exposure at the MRHD; however, inflammation was still present at higher doses.
Effects on adrenal gland.
Hypertrophy of the adrenal gland cortex was observed in rats (females only) and in mice following daily oral administration of cariprazine for 2 years and 6 months, respectively. Reversible hypertrophy/hyperplasia and vacuolation/ vesiculation of the adrenal gland cortex were observed following daily oral administration of cariprazine to dogs for 1 year. The NOAEL was 2 mg/kg/day which is 4 times the MRHD based on AUC of total cariprazine.
The relevance of these findings to human risk is unknown.6 Pharmaceutical Particulars
6.1 List of Excipients
This medicine does not contain lactose, sucrose, gluten or tartrazine.
Capsule contents.
Pregelatinised maize starch, magnesium stearate.
Capsule shell (1.5 mg capsule).
Titanium dioxide, gelatin.
Capsule shell (3 mg capsule).
Allura red AC, brilliant blue FCF, titanium dioxide, iron oxide yellow, gelatin.
Capsule shell (4.5 mg capsule).
Allura red AC, brilliant blue FCF, titanium dioxide, iron oxide yellow, gelatin.
Capsule shell (6 mg capsule).
Brilliant blue FCF, allura red AC, titanium dioxide, gelatin.
Printing ink (black: 1.5 mg, 3 mg and 6 mg capsules).
Shellac, iron oxide black, propylene glycol, potassium hydroxide.
Printing ink (white: 4.5 mg capsule).
Shellac, titanium dioxide, propylene glycol, simethicone.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 30°C. Keep the blister in the outer carton in order to protect from light.
6.5 Nature and Contents of Container
Transparent hard PVC/PE/PVDC blister heat-sealed with hard aluminum foil backing packed in folded carton box.
Reagila 1.5 mg and Reagila 3 mg hard capsules.
Cartons contain 10 (starter pack), 30, 60 or 90 hard capsules. Not all pack sizes may be marketed.
Reagila 4.5 mg and Reagila 6 mg hard capsules.
Cartons contain 30, 60 or 90 hard capsules. Not all pack sizes may be marketed.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of in accordance with local requirements.
6.7 Physicochemical Properties
Cariprazine HCl (active entity) is a white or almost white crystalline powder. It is freely soluble in methanol, slightly soluble in dichloromethane and, ethanol, very slightly soluble in acetone, acetonitrile and water, and practically insoluble in isopropyl alcohol and N,N-dimethylformamide and has a pKa of 8.2.
Chemical structure.
CAS number.
Cariprazine: [839712-12-8].
Cariprazine hydrochloride: [1083076-69-0].7 Medicine Schedule (Poisons Standard)
Prescription Only Medicine (S4).
Summary Table of Changes






 Adverse events reported with the use of Reagila (incidence of 2% or greater) that occurred during pooled schizophrenia studies (safety population) are presented in Table 2.
Adverse events reported with the use of Reagila (incidence of 2% or greater) that occurred during pooled schizophrenia studies (safety population) are presented in Table 2.





