What is in this leaflet
This leaflet answers some common questions about REKOVELLE. It does not contain all the available information. It does not take the place of talking to your doctor or pharmacist.
All medicines have risks and benefits. Your doctor has weighed the risks of you using REKOVELLE against the benefits they expect it will have for you.
If you have any concerns about taking this medicine, ask your doctor or pharmacist.
Keep this leaflet with the medicine. You may need to read it again.
What REKOVELLE is used for
REKOVELLE contains follitropin delta, which is a recombinant human follicle-stimulating hormone (FSH) produced in a human cell line (PER.C6®) by recombinant DNA technology.
REKOVELLE belongs to a class of hormones called gonadotropins. FSH is necessary for the growth and development of egg sacs (follicles) in women.
This medicine is used for controlled ovarian stimulation for the development of multiple follicles in women undergoing assisted reproductive technologies (ART) such as an in vitro fertilisation (IVF) or intracytoplasmic sperm injection (ICSI) cycle.
Ask your doctor if you have any questions about why this medicine has been prescribed for you. Your doctor may have prescribed it for another reason.
This medicine is not addictive.
It is available only with a doctor's prescription.
This medicine is not expected to affect your ability to drive a car or operate machinery.
Before you use REKOVELLE
When you must not use it
Do not use REKOVELLE if you have an allergy to:
- any medicine containing the active substance
- any of the ingredients listed at the end of this leaflet.
Some of the symptoms of an allergic reaction may include:
- shortness of breath
- wheezing or difficulty breathing
- swelling of the face, lips, tongue or other parts of the body
- rash, itching or hives on the skin.
Do not use REKOVELLE if any of the following apply to you:
- tumours of the hypothalamus or pituitary gland
- ovarian enlargement or ovarian cyst not due to polycystic ovarian syndrome
- bleeding from the vagina without a known cause
- ovarian, uterine or breast cancer
- experienced early menopause
- your ovaries have failed.
Do not use this medicine if you are already pregnant. It may affect your developing baby if you use it during pregnancy.
Do not breast-feed if you are taking this medicine.
Do not take this medicine after the expiry date printed on the pack or if the packaging is torn or shows signs of tampering. If it has expired or is damaged, return it to your pharmacist for disposal.
If you are not sure whether you should start using this medicine, talk to your doctor.
Before you start to use it
Your doctor will assess you and your partner's fertility. This may include tests for other medical conditions, which may interfere with your ability to become pregnant. If necessary, other medical conditions may be treated before starting infertility treatments including REKOVELLE.
Tell your doctor if you have allergies to any other medicines, foods, preservatives or dyes.
Tell your doctor if you have or have had any of the following medical conditions:
- disorders of the thyroid gland
- disorders of the adrenal glands
- high prolactin levels in the blood
- porphyria or a family history of porphyria.
Tell your doctor if you are already pregnant or are breast-feeding.
Treatment with REKOVELLE may increase your risk of developing a condition called ovarian hyperstimulation syndrome (OHSS). This is when the ovaries over-react to the hormonal treatment and become larger.
The most common symptoms are lower abdominal pain, discomfort or swelling. During stimulation your doctor will monitor your treatment by the use of ultrasound and/or blood tests to help determine if you are likely to develop OHSS. If necessary, your doctor will delay or cancel your REKOVELLE injection. You may also be advised to refrain from sexual intercourse until the end of the cycle if this occurs.
Compared to natural conception, the frequency of multiple pregnancies and births is higher in patients receiving treatments that stimulate follicle growth for ovulation induction. The majority of these are twins. Your doctor will monitor your response to treatment to minimise the chance of multiple pregnancies, because of the greater risks they carry for mothers and babies.
Compared to natural conception, the frequency of pregnancy loss is higher in patients undergoing treatments to stimulate follicle growth for ovulation induction.
There may be a slightly increased risk of birth defects in women using assisted reproductive technologies. This may be due to increased maternal age, genetic factors, multiple pregnancies or the procedures. An effect of medicines used to induce ovulation has not been excluded.
Tell your doctor if you or a family member have or have had signs of blood clots (e.g. pain, warmth, redness, numbness or tingling in the arm or leg). Treatment with REKOVELLE may increase the risk of blood clots forming in your blood vessels.
Talk to your doctor about any concerns you may have before undergoing treatment or before you start using REKOVELLE.
Taking other medicines
Tell your doctor or pharmacist if you are taking any other medicines, including any that you get without a prescription from your pharmacy, supermarket or health food shop.
Some medicines and REKOVELLE may interfere with each other.
Your doctor and pharmacist have more information on medicines to be careful with or avoid while taking this medicine.
How REKOVELLE is given
Follow all directions given to you by your doctor or pharmacist carefully. They may differ from the information contained in this leaflet.
Treatment with REKOVELLE should be initiated under the supervision of a physician experienced in the treatment of fertility problems. You must be educated on how to use the REKOVELLE injection pen and to perform injections
If you do not understand the instructions on the box, ask your doctor or pharmacist for help.
How much to inject
The dosage of REKOVELLE is individualised for each patient. The dosage requirement can be influenced by a woman's body weight and her ovarian reserve, which is reflected in her serum anti- Müllerian hormone (AMH) concentration. Your doctor may measure and record your body weight and serum AMH concentration to assist in determining your REKOVELLE dosage requirement.
Women with low AMH levels are likely to have low ovarian reserve.
REKOVELLE is dosed in micrograms and not in international units (IU) of biological activity. The dosing regimen is specific for REKOVELLE and the microgram dose cannot be applied to other gonadotropins.
Your doctor will decide on the dose of REKOVELLE to be given. Your doctor will individualise your treatment cycle of REKOVELLE.
How to use it
REKOVELLE is to be injected under the skin, preferably in the abdominal wall. Supervision by a trained health practitioner is required for the first injection.
Self-administration of REKOVELLE should only be performed if you are well motivated, adequately trained and have access to expert advice.
For instructions on the administration with the REKOVELLE injection pen, see the 'Instructions for Use' in the pack.
It is important that you do not try to give yourself the injection unless you have received special training from your doctor or nurse.
When to use it
Dosing with REKOVELLE should be initiated day 2 or 3 after start of menstrual bleeding, and continue until adequate follicular development has been achieved as assessed by monitoring with ultrasound alone or in combination with measurement of serum estradiol levels.
How long to use it
Adequate follicular development is achieved on average by the ninth day of treatment (range 5 to 20 days).
Continue using your medicine for as long as your doctor tells you.
If you forget to use REKOVELLE
If you forget an injection or are not sure what to do, contact your doctor or nurse immediately for advice.
If you inject too much (overdose)
Immediately telephone your doctor or the Poisons Information Centre (telephone 13 11 26) for advice, or go to Accident and Emergency at the nearest hospital, if you think that you or anyone else may have used too much REKOVELLE.
While you are using REKOVELLE
Things you must do
See your doctor regularly so you can be monitored closely throughout your treatment.
Tell your doctor, nurse and pharmacist that you are using REKOVELLE if you are about to be started on any new medicine.
Tell any other doctors who treat you that you are taking this medicine.
Tell your doctor immediately if you become pregnant while taking this medicine.
Things you must not do
Do not use REKOVELLE to treat any other complaints unless your doctor tells you to.
Do not give your medicine to anyone else, even if they have the same condition as you.
Do not stop taking your medicine or lower the dosage without checking with your doctor.
Side effects
Tell your doctor or pharmacist as soon as possible if you do not feel well while you are using REKOVELLE.
All medicines can have side effects. Sometimes they are serious, most of the time they are not. You may need medical attention if you get some of the side effects.
Do not be alarmed by the following lists of side effects. You may not experience any of them.
Ask your doctor or pharmacist to answer any questions you may have.
Tell your doctor or pharmacist if you notice any of the following and they worry you:
- headache
- nausea
- pelvic discomfort
- pelvic pain
- fatigue.
The above list includes the more common side effects of your medicine. They are usually mild and short-lived.
Tell your doctor as soon as possible if you notice any of the following:
- mood swings
- sleepiness/drowsiness
- dizziness
- diarrhoea
- constipation
- abdominal discomfort
- vaginal bleeding
- breast pain
- breast tenderness.
If any of the following happen, tell your doctor immediately or go to Accident and Emergency at your nearest hospital, even if they develop some days after the final injection:
- signs of an allergic reaction such as shortness of breath, wheezing or difficulty breathing; swelling of the face, lips, tongue or other parts of the body; rash, itching or hives on the skin
A complication with FSH treatment is unwanted overstimulation of the ovaries. Contact your doctor if you experience any symptoms of overstimulation of the ovaries. The first symptoms of ovarian overstimulation may be noticed as pain in the stomach (abdomen), feeling sick or diarrhoea.
Ovarian overstimulation may develop into a serious medical condition called ovarian hyperstimulation syndrome (OHSS).
Signs and symptoms of severe OHSS may include:
- acute stomach pain, weight gain (due to the accumulation of fluid in the abdomen and/or chest), shortness of breath and passing less urine
- pain, warmth, redness, numbness, or tingling in your arm or leg; confusion, extreme dizziness or severe headache (signs of a blood clot).
If you get any side effects, talk to you doctor, pharmacist or nurse.
This also includes any possible side effects not listed above.
Tell your doctor if you notice anything that worries you or that is making you feel unwell.
After using REKOVELLE
Storage
- Keep REKOVELLE in a refrigerator at a temperature of 2°C to 8°C.
- Do not freeze.
- REKOVELLE may be removed from the refrigerator and stored at or below 25°C for up to 3 months and must be discarded afterwards.
- Before use: store in the original package in order to protect from light.
- Do not use this medicine if you notice that it is cloudy or there are particles in it.
- After the first injection: the pre- filled pen can be stored at or below 25°C and it must be discarded after 28 days.
- Keep REKOVELLE out of sight and reach of children.
Disposal
Once you have injected REKOVELLE, do not re-use the needle.
Discard the used needle into an approved, puncture-resistant sharps container and keep it out of the reach of children.
Never put a used needle into your normal household rubbish bin.
Dispose of this medicine as instructed by your doctor, nurse or pharmacist.
Product description
What it looks like
REKOVELLE is a clear and colourless solution for injection presented in a pre-filled multidose pen with a dose selection knob, display window and cap. Each pen contains an integrated non- replaceable cartridge containing the solution for injection. Each pack contains 1 pre-filled multidose pen and a number of injection needles.
REKOVELLE is available in 3 strengths.
REKOVELLE 12 micrograms pack contains:
- 1 x pre-filled multidose pen containing 12 micrograms follitropin delta (rhu) in 0.36 mL of solution for injection
- 3 x injection needles
REKOVELLE 36 micrograms pack contains:
- 1 x pre-filled multidose pen containing 36 micrograms follitropin delta (rhu) in 1.08 mL of solution for injection
- 6 x injection needles
REKOVELLE 72 micrograms pack contains:
- 1 x pre-filled multidose pen containing 72 micrograms follitropin delta (rhu) in 2.16 mL of solution for injection
- 9 x injection needles
Each mL of the solution contains 33.3 micrograms of follitropin delta.
Ingredients
The active ingredient in REKOVELLE is follitropin delta.
Other ingredients are:
- phenol
- polysorbate 20
- methionine
- sodium sulfate
- dibasic sodium phosphate dodecahydrate
- phosphoric acid
- sodium hydroxide
- Water for Injections.
This medicine does not contain lactose, sucrose, gluten, tartrazine or any other azo dyes.
Sponsor
REKOVELLE is supplied in Australia by:
Ferring Pharmaceuticals Pty Ltd
Suite 2, Level 1, Building 1
20 Bridge Street
Pymble, NSW 2073, Australia.
AUST R 289310 - REKOVELLE
12 micrograms/0.36 mL solution for injection in a pre-filled pen.
AUST R 289311 - REKOVELLE
36 micrograms/1.08 mL solution for injection in a pre-filled pen.
AUST R 289312 - REKOVELLE
72 micrograms/2.16 mL solution for injection in a pre-filled pen.
This leaflet was prepared in April 2017. #45471-v1A
REKOVELLE® is a registered trademark of Ferring B.V.
® = Registered Trademark
Published by MIMS June 2018

 Dosing with Rekovelle should be initiated on day 2 or 3 after start of menstrual bleeding in a protocol using a gonadotropin-releasing hormone (GnRH) antagonist, or approximately 2 weeks after the start of agonist treatment in a protocol using down-regulation with a GnRH agonist. Treatment should continue until adequate follicular development has been achieved as assessed by monitoring with ultrasound alone or in combination with measurement of serum oestradiol levels. Adequate follicular development is achieved on average by the ninth or tenth day of treatment (range 5 to 20 days). As soon as ≥ 3 follicles ≥ 17 mm are observed, a single injection of 250 micrograms recombinant human chorionic gonadotropin (hCG) or 5,000 IU hCG is administered to induce final follicular maturation.
Dosing with Rekovelle should be initiated on day 2 or 3 after start of menstrual bleeding in a protocol using a gonadotropin-releasing hormone (GnRH) antagonist, or approximately 2 weeks after the start of agonist treatment in a protocol using down-regulation with a GnRH agonist. Treatment should continue until adequate follicular development has been achieved as assessed by monitoring with ultrasound alone or in combination with measurement of serum oestradiol levels. Adequate follicular development is achieved on average by the ninth or tenth day of treatment (range 5 to 20 days). As soon as ≥ 3 follicles ≥ 17 mm are observed, a single injection of 250 micrograms recombinant human chorionic gonadotropin (hCG) or 5,000 IU hCG is administered to induce final follicular maturation.

 The clinical value of the AMH-based dosing regimen of Rekovelle was also assessed in secondary endpoints, such as ovarian response, OHSS risk management and gonadotropin consumption.
The clinical value of the AMH-based dosing regimen of Rekovelle was also assessed in secondary endpoints, such as ovarian response, OHSS risk management and gonadotropin consumption.
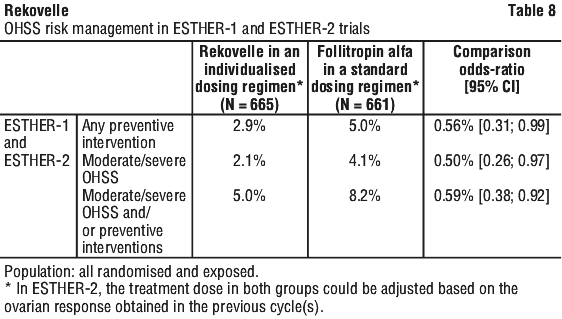
 The risk of requiring OHSS preventive interventions or experiencing OHSS increased with increasing AMH and this relationship differed between treatment groups (p < 0.05); see Figure 1.
The risk of requiring OHSS preventive interventions or experiencing OHSS increased with increasing AMH and this relationship differed between treatment groups (p < 0.05); see Figure 1.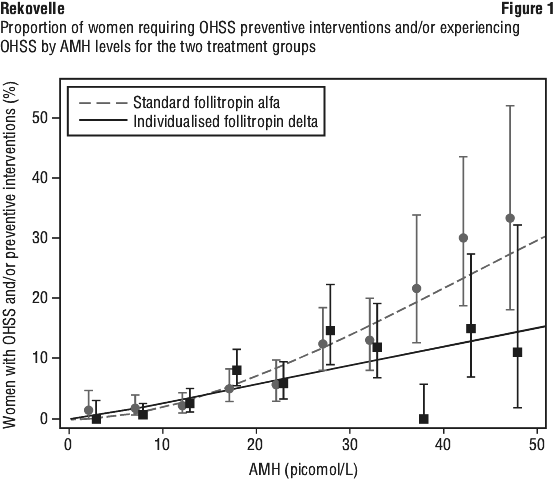 In ovulatory patients with polycystic ovaries undergoing a GnRH antagonist cycle, the incidence of early moderate/severe OHSS and/or preventive interventions for early OHSS was 7.7% with Rekovelle in an individualised dosing regimen and 26.7% with follitropin alfa in a standard dosing regimen.
In ovulatory patients with polycystic ovaries undergoing a GnRH antagonist cycle, the incidence of early moderate/severe OHSS and/or preventive interventions for early OHSS was 7.7% with Rekovelle in an individualised dosing regimen and 26.7% with follitropin alfa in a standard dosing regimen. The cumulative incidence of moderate/severe OHSS and/or preventive interventions for early OHSS across three stimulation cycles were significantly lower for Rekovelle in an individualised dosing regimen compared to follitropin alfa in a standard dosing regimen (p < 0.05). See Table 8.
The cumulative incidence of moderate/severe OHSS and/or preventive interventions for early OHSS across three stimulation cycles were significantly lower for Rekovelle in an individualised dosing regimen compared to follitropin alfa in a standard dosing regimen (p < 0.05). See Table 8.