1 Name of Medicine
Budesonide.
Formoterol fumarate dihydrate.
2 Qualitative and Quantitative Composition
Rilast Rapihaler is a pressurised metered dose inhaler (pMDI).
The following strengths are registered:
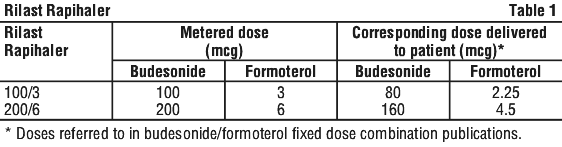
Rilast Rapihaler 100/3.
Each delivered dose (the dose that leaves the mouthpiece) contains as active constituents: budesonide 80 microgram/inhalation and formoterol 2.25 microgram/inhalation.
Rilast Rapihaler 200/6.
Each delivered dose (the dose that leaves the mouthpiece) contains as active constituents: budesonide 160 microgram/inhalation and formoterol 4.5 microgram/inhalation.
To avoid confusion, Rilast Rapihaler is labelled as metered dose like Rilast Turbuhaler. Table 1 gives the corresponding dose delivered to the patient.
 For the full list of excipients, see Section 6.1 List of Excipients.
For the full list of excipients, see Section 6.1 List of Excipients.3 Pharmaceutical Form
Inhalation, pressurised.
4.1 Therapeutic Indications
Asthma.
Rilast Rapihaler is indicated in adults and adolescents (12 years and older), for the treatment of asthma to achieve overall asthma control, including the relief of symptoms and the reduction of the risk of exacerbations (see Section 4.2 Dose and Method of Administration).
Chronic obstructive pulmonary disease (COPD).
Rilast Rapihaler 200/6 is indicated for the symptomatic treatment of moderate to severe COPD (FEV1 ≤ 50% predicted normal) in adults with frequent symptoms despite long-acting bronchodilator use, and/or a history of recurrent exacerbations. Rilast Rapihaler is not indicated for the initiation of bronchodilator therapy in COPD.4.2 Dose and Method of Administration
Asthma.
Rilast Rapihaler can be used according to different treatment approaches:
A. Anti-inflammatory reliever therapy (patients with mild disease).
B. Anti-inflammatory reliever plus maintenance therapy.
C. Maintenance therapy (fixed dose).
Anti-inflammatory reliever therapy (patients with mild disease). Rilast Rapihaler 100/3 is taken as needed for the relief of asthma symptoms when they occur, and as preventative treatment of symptoms in those circumstances recognised by the patient to precipitate an asthma attack. Patients should be advised to always have Rilast Rapihaler 100/3 available for relief of symptoms.
Preventative use of Rilast Rapihaler 100/3 for allergen- or exercise-induced bronchoconstriction (AIB/EIB) should be discussed between physician and patient; the recommended dose frequency should take into consideration both allergen exposure and exercise patterns.
Adults and adolescents (12 years and older).
Patients should take 2 inhalations of Rilast Rapihaler 100/3 as needed in response to symptoms. If symptoms persist after a few minutes, an additional 2 inhalations should be taken. No more than 12 inhalations should be taken on any single occasion.
A total daily dose of more than 16 inhalations is normally not needed, however a total daily dose of up to 24 inhalations can be used temporarily. If the patient experiences a 3-day period of deteriorating symptoms after taking additional as needed inhalations, the patient should be reassessed for alternative explanations of persisting symptoms.
Anti-inflammatory reliever plus maintenance therapy. When maintenance treatment with a combination of inhaled corticosteroid (ICS) and long acting β2-agonist (LABA) is required, patients take anti-inflammatory reliever therapy and in addition take a daily maintenance dose of Rilast Rapihaler. The as-needed inhalations provide both rapid relief of symptoms and improved overall asthma control. Patients should be advised to have Rilast Rapihaler available for relief of symptoms at all times.
Preventative use of Rilast Rapihaler 100/3 for allergen- or exercise-induced bronchoconstriction (AIB/EIB) should be discussed between physician and patient; the recommended dose frequency should take into consideration both allergen exposure and exercise patterns.
Adults and adolescents (12 years and older).
Patients should take 2 inhalations of Rilast Rapihaler 100/3 as needed in response to symptoms to control asthma. If symptoms persist after a few minutes, 2 additional inhalations should be taken. No more than 12 inhalations should be taken on any single occasion.
Patients also take the recommended maintenance dose of Rilast Rapihaler 100/3, 4 inhalations per day, given as either 2 inhalations in the morning and evening or as 4 inhalations in either the morning or evening. For some patients, a maintenance dose of Rilast Rapihaler 100/3 4 inhalations twice daily may be appropriate. The maintenance dose should be titrated to the lowest dose at which effective control of asthma is maintained.
A total daily dose of more than 16 inhalations is normally not needed, however a total daily dose of up to 24 inhalations can be used temporarily. If the patient experiences a 3-day period of deteriorating symptoms after taking the appropriate maintenance therapy and additional as needed inhalations, the patient should be reassessed for alternative explanations of persisting symptoms.
Maintenance therapy (fixed dose). When maintenance treatment with a combination of ICS and LABA is required, Rilast Rapihaler is taken as a fixed daily dose treatment, with a separate short-acting bronchodilator for relief of symptoms. Patients should be advised to have their separate short-acting bronchodilator available for relief of symptoms at all times.
Increasing use of short-acting bronchodilators indicates a worsening of the underlying condition and warrants reassessment of the asthma therapy. The dosage of Rilast Rapihaler should be individualised according to disease severity. When control of asthma has been achieved, the maintenance dose should be titrated to the lowest dose at which effective asthma control is maintained.
Adults and adolescents (12 years and older).
Rilast Rapihaler 100/3.
2 or 4 inhalations of Rilast Rapihaler 100/3 twice daily. The maximum recommended daily maintenance dose is 8 inhalations (4 inhalations twice daily corresponding to 800 microgram budesonide/24 microgram formoterol).
Rilast Rapihaler 200/6.
2 inhalations of Rilast Rapihaler 200/6 twice daily. The maximum recommended daily maintenance dose is 4 inhalations (2 inhalations twice daily corresponding to 800 microgram budesonide/24 microgram formoterol).
For adults 18 years and over who require a higher daily maintenance dose, the maximum recommended maintenance dose may be increased to 4 inhalations of Rilast Rapihaler 200/6 twice daily (corresponding to 1600 microgram budesonide/48 microgram formoterol).
COPD.
Adults.
2 inhalations of Rilast Rapihaler 200/6 twice daily. The maximum recommended daily dose is 4 inhalations (corresponding to 800 microgram budesonide/ 24 microgram formoterol).
Special patient populations.
Renal impairment.
There are no data available for use of Rilast Rapihaler in patients with renal impairment.
Hepatic impairment.
There are no data available for use of Rilast Rapihaler in patients with hepatic impairment. As budesonide and formoterol are primarily eliminated via hepatic metabolism, an increased systemic availability can be expected in patients with severe liver disease.
Use in the elderly.
There is no need to adjust the dose in elderly patients.
Use in paediatric patients.
Rilast Rapihaler is not recommended for children below 12 years of age.
General information.
If patients take Rilast Rapihaler as an anti-inflammatory reliever (either alone or in combination with maintenance therapy) physicians should discuss allergen exposure and exercise patterns with the patients and take these into consideration when recommending the dose frequency for asthma treatment.
If patients take Rilast Rapihaler as a maintenance therapy, they should be instructed that Rilast Rapihaler must be used even when asymptomatic for optimal benefit.
Instruction for correct use of Rilast Rapihaler.
On actuation of the Rilast Rapihaler, a volume of the suspension is expelled from the canister at high velocity. When the patient inhales through the mouthpiece at the same time as actuating the inhaler, the substance will follow the inspired air into the airways.
Note.
It is important to instruct the patient to:
Check the expiry date.
Carefully read the instructions for use in the patient information leaflet that are provided with each pack of Rilast Rapihaler.
Shake the inhaler well prior to each use to mix its contents properly.
Prime the inhaler when the inhaler is new, if it has not been used for more than one week or if it has been dropped. See Instructions for use leaflet for priming instructions.
Place the mouthpiece into the mouth. While breathing in slowly and deeply, press the inhaler firmly to release the medication. Continue to breathe in and hold the breath for approximately 10 seconds or as long as is comfortable. Shake the inhaler again and repeat this step for the second inhalation.
Rinse the mouth with water after inhaling the maintenance dose to minimise the risk of oropharyngeal thrush.
Clean the mouthpiece of the inhaler regularly, at least once a week with a clean dry cloth. Do not put the inhaler into water.4.3 Contraindications
Hypersensitivity to budesonide, formoterol or any other ingredients present in this formulation.
4.4 Special Warnings and Precautions for Use
Treatment of asthma or COPD should be in accordance with physician recommendations or current national treatment guidelines.
Patients with asthma should have a personal asthma action plan designed in association with their healthcare professional. This plan should incorporate a stepwise treatment regime which can be instituted if the patient's asthma improves or deteriorates.
Patients should be advised to have their reliever available at all times, either Rilast Rapihaler (for asthma patients on anti-inflammatory reliever therapy and anti-inflammatory reliever plus maintenance therapy) or a separate short-acting bronchodilator (for other patients using Rilast Rapihaler as fixed dose maintenance therapy only and for COPD patients).
Sudden and progressive deterioration in control of asthma or COPD is potentially life threatening and the patient should undergo urgent medical assessment. In this situation, consideration should be given to the need for increased therapy with corticosteroids (e.g. a course of oral corticosteroids) or antibiotic treatment if a bacterial infection is present. For treatment of severe exacerbations, a combination product of ICS and LABA alone is not sufficient. Patients should be advised to seek medical attention if they find the treatment ineffective or they have exceeded the prescribed dose of Rilast Rapihaler.
It is recommended that the maintenance dose be tapered when long-term treatment is discontinued, and the dosing should not be stopped abruptly. Complete withdrawal of ICS should not be considered unless it is temporarily required to confirm the diagnosis of asthma.
Oral corticosteroid usage.
Rilast Rapihaler should not be used to initiate treatment with inhaled steroids in patients being transferred from oral steroids. Care should be taken when commencing Rilast Rapihaler treatment, particularly if there is any reason to suspect that adrenal function is impaired from previous systemic steroid therapy.
Potential systemic effects of ICS.
ICS are designed to direct glucocorticoid delivery to the lungs in order to reduce overall systemic glucocorticoid exposure and side effects. However, in higher than recommended doses, ICS may have adverse effects; possible systemic effects of ICS include Cushing's syndrome, cushingoid features, depression of the HPA axis, reduction of bone density, cataract and glaucoma, and retardation of growth rate in children and adolescents. These effects are much less likely to occur with ICS treatments than with oral corticosteroids. In steroid-dependent patients, prior systemic steroid usage may be a contributing factor, but such effects may occur amongst patients who use only ICS regularly.
HPA axis suppression and adrenal insufficiency.
Dose-dependent HPA axis suppression (as indicated by 24-hour urinary and/or plasma cortisol AUC) has been observed with inhaled budesonide, although the physiological circadian rhythms of plasma cortisol were preserved. This indicates that the HPA axis suppression represents a physiological adaption in response to inhaled budesonide, not necessarily adrenal insufficiency. The lowest dose that results in clinically relevant adrenal insufficiency has not been established. Very rare cases of clinically relevant adrenal dysfunction have been reported in patients using inhaled budesonide at recommended doses.
Clinically important disturbances of the HPA axis and/or adrenal insufficiency induced by severe stress (e.g. trauma, surgery, infection in particular gastroenteritis or other conditions associated with severe electrolyte loss) may be related to inhaled budesonide in specific patient populations. These are patients switched from oral corticosteroids (see Section 4.4 Special Warnings and Precautions for Use, Oral corticosteroid usage) and patients administering concomitant medication metabolised by CYP3A4 (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions, Pharmacokinetic interactions). Monitoring for signs of adrenal dysfunction is advisable in these patient groups. For these patients additional systemic glucocorticosteroid treatment should be considered during periods of stress, a severe asthma attack or elective surgery.
Bone density.
Whilst corticosteroids may have an effect on bone mass at high doses, long-term follow up (3 to 6 years) studies of budesonide treatment in adults at recommended doses, have not demonstrated a negative effect on bone mass compared to placebo, including a study conducted in patients with a high risk of osteoporosis. The lowest dose that does effect bone mass has not been established.
Bone-mineral density measurements in children should be interpreted with caution as an increase in bone area in growing children may reflect an increase in bone volume. In 3 large, medium-to-long-term (12 months to 6 years) studies in children (5 to 16 years), no effects on bone-mineral density were observed after treatment with budesonide (189 to 1322 microgram/day) compared to nedocromil, placebo or age matched controls. However, in a randomised 18-month paediatric study (n=176; 5 to 10 years), bone-mineral density was significantly decreased by 0.11 g/cm2 (p=0.023) in the group treated with inhaled budesonide via Turbuhaler, compared with the group treated with inhaled disodium cromoglycate. The dose of budesonide was 400 microgram twice daily for 1 month, 200 microgram twice daily for 5 months, and 100 microgram twice daily for 12 months, and the dose of disodium cromoglycate 10 mg three times daily. The clinical significance of this result remains uncertain.
Growth.
Limited data from long-term studies suggest that most children and adolescents treated with inhaled budesonide will ultimately achieve their adult target height. However, an initial small but transient reduction of growth (approximately 1 cm) has been observed. This generally occurs within the first year of treatment. Physicians should closely follow the growth of children and adolescents taking long-term corticosteroids.
Rare individuals may be exceptionally sensitive to ICS. Height measurements should be performed to identify patients with increased sensitivity. The potential growth effects of prolonged treatment should be weighed against the clinical benefit. To minimise the systemic effects of ICS, each patient should be titrated to his/her lowest dose at which effective control of symptoms is maintained (see Section 4.2 Dose and Method of Administration).
Visual disturbance.
Visual disturbance may be reported with systemic and topical corticosteroid use. If a patient presents with symptoms such as blurred vision or other visual disturbances, the patient should be considered for referral to an ophthalmologist for evaluation of possible causes which may include cataract, glaucoma or rare diseases such as central serous chorioretinopathy (CSCR) which have been reported after use of systemic and topical corticosteroids.
Infections/tuberculosis.
Signs of existing infection may be masked by the use of high doses of glucocorticosteroids and new infections may appear during their use. Special care is needed in patients with active or quiescent pulmonary tuberculosis or fungal, bacterial or viral infections of the respiratory system.
Sensitivity to sympathomimetic amines.
In patients with increased susceptibility to sympathomimetic amines (e.g. inadequately controlled hyperthyroidism), formoterol should be used with caution.
Cardiovascular disorders.
β2-agonists have an arrhythmogenic potential that must be considered before commencing treatment for bronchospasm.
The effects of formoterol in acute as well as chronic toxicity studies were seen mainly on the cardiovascular system and consisted of hyperaemia, tachycardia, arrhythmias and myocardial lesions. These are known pharmacological manifestations seen after administration of high doses of β2-adrenoceptor agonists.
Patients with pre-existing cardiovascular conditions may be at greater risk of developing adverse cardiovascular effects following administration of β2-adrenoreceptor agonists. Caution is advised when formoterol is administered to patients with severe cardiovascular disorders such as ischaemic heart disease, tachyarrhythmias or severe heart failure.
Hypokalaemia.
High doses of β2-agonists can lower serum potassium by inducing a redistribution of potassium from the extracellular to the intracellular compartment, via stimulation of Na+/K+-ATPase in muscle cells.
Potentially serious hypokalaemia may result. Particular caution is advised in acute exacerbation as the associated risk may be augmented by hypoxia. The hypokalaemic effect may be potentiated by concomitant treatments (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions, Xanthine derivatives, mineralocorticosteroids and diuretics). Patients receiving digoxin are particularly sensitive to hypokalaemia. Serum potassium levels should therefore be monitored in such situations.
Diabetes.
Due to the blood-glucose increasing effects of β2-stimulants, extra blood glucose controls are initially recommended when diabetic patients are commenced on formoterol.
Pneumonia.
Physicians should remain vigilant for the possible development of pneumonia in patients with COPD as the clinical features of pneumonia and exacerbations frequently overlap. Pneumonia has been reported following the administration of inhaled corticosteroids. See Section 4.8 Adverse Effects (Undesirable Effects).
Use in renal impairment.
The effect of decreased kidney function on the pharmacokinetics of formoterol and budesonide are not known.
Use in hepatic impairment.
The effect of decreased liver function on the pharmacokinetics of formoterol and budesonide are not known. As budesonide and formoterol are primarily eliminated via hepatic metabolism, an increased exposure can be expected in patients with severe liver disease.
Use in the elderly.
See Section 5.1 Pharmacodynamic Properties, Clinical trials.
Paediatric use.
Rilast Rapihaler is not recommended for children under the age of 12 years because of lack of data on efficacy and safety.
Effects on laboratory tests.
No data available.4.5 Interactions with Other Medicines and Other Forms of Interactions
Pharmacokinetic interactions.
The metabolism of budesonide is primarily mediated by the enzyme CYP3A4. Potent CYP3A inhibitors may therefore increase systemic exposure to budesonide. This is of limited clinical importance for short-term (1 to 2 weeks) treatment with potent CYP3A4 inhibitors but should be taken into consideration during long-term treatment.
If a patient requires long-term concomitant treatment with Rilast Rapihaler and a potent CYP3A4 inhibitor, the benefit should be weighed against the increased risk of systemic corticosteroid side effects, patients should be monitored for corticosteroid side effects and/or a reduction of the ICS dose could be considered.
Pharmacodynamic interactions.
Neither budesonide nor formoterol have been observed to interact with any other drug used in the treatment of asthma or COPD.
β-receptor blocking agents.
β-receptor blocking agents, especially those that are non-selective, may partially or totally inhibit the effect of β2-agonists. These drugs may also increase airway resistance, therefore the use of these drugs in asthma patients is not recommended.
Other sympathomimetic agents.
Other β-adrenergic stimulants or sympathomimetic amines such as ephedrine should not be given concomitantly with formoterol, since the effects will be cumulative. Patients who have already received large doses of sympathomimetic amines should not be given formoterol.
Xanthine derivatives, mineralocorticosteroids and diuretics.
Hypokalaemia may result from β2-agonist therapy and may be potentiated by concomitant treatment with xanthine derivatives, mineralocorticosteroids, and diuretics (see Section 4.4 Special Warnings and Precautions for Use, Hypokalaemia).
Monoamine oxidase inhibitors, tricyclic antidepressants, quinidine, disopyramide, procainamide, phenothiazines and antihistamines.
The adverse cardiovascular effects of formoterol may be exacerbated by concurrent administration of drugs associated with QT-interval prolongation and increased risk of ventricular arrhythmia. For this reason, caution is advised when formoterol is administered to patients already taking monoamine oxidase inhibitors, tricyclic antidepressants, quinidine, disopyramide, procainamide, phenothiazines, or antihistamines associated with QT-interval prolongation (e.g. terfenadine, astemizole).4.6 Fertility, Pregnancy and Lactation
Effects on fertility.
There are no animal studies on the effect of the budesonide/formoterol combination on fertility.
Long-term treatment of female mice and rats with formoterol fumarate causes ovarian stimulation, the development of ovarian cysts, and hyperplasia of granulosa/theca cells as a result of the β-agonist properties of the compound. A study by another company showed no effect on fertility of female rats dosed orally with formoterol fumarate at 60 mg/kg/day for two weeks. This finding was repeated in an AstraZeneca study where no effect was seen on the fertility of female rats dosed orally with formoterol fumarate at 15 mg/kg/day for two weeks.
Testicular atrophy was observed in mice given formoterol fumarate in the diet at 0.2 to 50 mg/kg/day for 2 years, but no effect on male fertility was observed in rats dosed orally at 60 mg/kg/day for 9 weeks, in studies undertaken by another company.
(Category B3)
For the concomitant treatment with budesonide and formoterol, no clinical data on exposed pregnancies are available. Fetal malformations (umbilical hernia and cleft palate), typical of glucocorticoid toxicity in animals, occurred in rats dosed with the budesonide/formoterol Rapihaler formulation at the inhaled dose of 12 microgram/kg/day budesonide and 0.66 microgram/kg/day formoterol, with plasma AUC values for both drugs below that expected in patients at the maximum recommended clinical dose. No teratogenic effect was detected at 2.5 microgram/kg/day of budesonide and 0.14 microgram/kg/day of formoterol.
Rilast Rapihaler should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus. Only after special consideration should Rilast Rapihaler be used during the first 3 months and shortly before delivery.
Because β-agonists, including formoterol, may potentially interfere with uterine contractility due to a relaxant effect on uterine smooth muscle, Rilast Rapihaler should be used during labour only if the potential benefit justifies the potential risk.
Budesonide.
Results from a large prospective epidemiological study and from world-wide post marketing experience indicate no adverse effects of inhaled budesonide during pregnancy on the health of the fetus/newborn child.
If treatment with glucocorticosteroids during pregnancy is unavoidable, ICS such as budesonide should be considered due to their lower systemic effect. The lowest effective dose of budesonide to maintain asthma control should be used.
Formoterol.
No teratogenic effects were observed in rats receiving formoterol fumarate at doses up to 60 mg/kg/day orally or 1.2 mg/kg/day by inhalation. Foetal cardiovascular malformations were observed in one study in which pregnant rabbits were dosed orally at 125 or 500 mg/kg/day during the period of organogenesis, but similar results were not obtained in another study at the same dose range. In a third study, an increased incidence of subcapsular hepatic cysts was observed in foetuses from rabbits dosed orally at 60 mg/kg/day. Decreased birth weight and increased perinatal/postnatal mortality were observed when formoterol fumarate was given to rats at oral doses of 0.2 mg/kg/day or greater during late gestation.
Budesonide is excreted in breast milk. However, due to the relatively low doses used via the inhalational route the amount of drug present in the breast milk, if any, is likely to be low.
It is not known whether formoterol passes into human breast milk. In rats, formoterol was excreted into breast milk. There are no studies in lactating animals using the budesonide/formoterol combination. Increased postnatal mortality at maternal formoterol doses of 0.2 mg/kg/day PO or greater, and retardation of pup growth at 15 mg/kg/day PO were observed in a rat study. There are no well-controlled human studies using Rilast Rapihaler in nursing mothers. Because many drugs are excreted in human breast milk, administration of Rilast Rapihaler to women who are breastfeeding should only be considered if the expected benefit to the mother is greater than any possible risk to the child.4.7 Effects on Ability to Drive and Use Machines
Driving or using machinery should be undertaken with caution until the effect of Rilast Rapihaler on the individual is established. Rilast Rapihaler does not generally affect the ability to drive or use machinery.
4.8 Adverse Effects (Undesirable Effects)
Since Rilast Rapihaler contains both budesonide and formoterol, the same adverse effects as reported for these substances may be expected. No increased incidence of adverse reactions has been seen following concurrent administration of the two compounds. The most common drug related adverse reactions are pharmacologically predictable side-effects of β2-agonist therapy, such as tremor and palpitations. These tend to be mild and usually disappear within a few days of commencing treatment.
In the clinical program comparing budesonide/formoterol Rapihaler with budesonide/formoterol Turbuhaler, 679 adults and adolescents (Study 681 and Study 715) were exposed to budesonide/formoterol Rapihaler 800/24 microgram daily with a median duration of 359 days and a range of 1 to 427 days.
There were no apparent differences in the overall pattern of AE's between the budesonide/formoterol Rapihaler and budesonide/formoterol Turbuhaler groups in the clinical program. The AEs were generally mild to moderate in intensity and the pattern was that usually seen in a population with persistent asthma and dominated by symptoms of upper respiratory events.
Overall, the AE profile was similar for patients receiving budesonide/formoterol Rapihaler and budesonide/formoterol Turbuhaler with regard to total daily dose, age, sex and ethnic group and no new safety concerns were identified with budesonide/formoterol Rapihaler.
If oropharyngeal candidiasis develops, it may be treated with appropriate anti-fungal therapy whilst still continuing with Rilast Rapihaler therapy. The incidence of candidiasis can generally be held to a minimum by having patients rinse their mouth out with water after inhaling their maintenance dose.
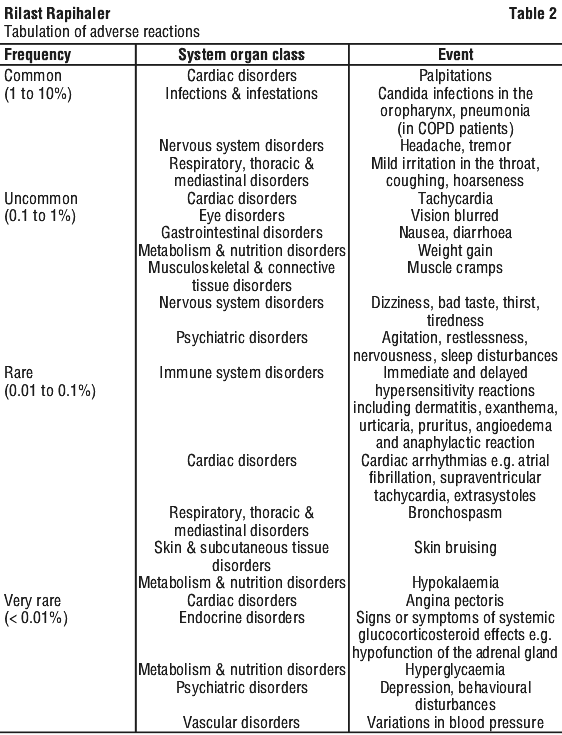
Adverse reactions, which have been associated with budesonide, formoterol, and budesonide/formoterol Turbuhaler and Rapihaler combination products, are given in Table 2.
 As with other inhalation therapy, paradoxical bronchospasm may occur in very rare cases.
As with other inhalation therapy, paradoxical bronchospasm may occur in very rare cases.
Treatment with β-sympathomimetics may result in an increase in blood levels of insulin, free fatty acids, glycerol and ketone bodies.
Pneumonia.
Table 3 provides the incidence of pneumonia observed in the four-pivotal phase III COPD studies (see Section 5.1 Pharmacodynamic Properties, Clinical trials, COPD) for the budesonide/formoterol (as Turbuhaler or Rapihaler 200/6) and comparative placebo arms.
 In these placebo-controlled studies, the incidence of pneumonia was low.
In these placebo-controlled studies, the incidence of pneumonia was low.
Anti-inflammatory reliever therapy.
Overall, anti-inflammatory reliever therapy is generally well tolerated, based on the frequency and nature of adverse effects. No new safety concerns were identified for the use of Rilast Rapihaler 100/3 as needed in a mild asthma population.
Reporting suspected adverse effects.
Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.4.9 Overdose
An overdose of formoterol may lead to effects that are typical for β2-adrenergic agonists: tremor, headache, palpitations, and tachycardia. Monitoring of serum potassium concentrations may be warranted. Hypotension, metabolic acidosis, hypokalaemia, and hyperglycaemia may also occur. Supportive and symptomatic treatment may be indicated. β-blockers should be used with care because of the possibility of inducing bronchospasm in sensitive individuals. A metered dose of 120 microgram administered during 3 hours in patients with acute bronchial obstruction raised no safety concerns.
Acute overdosage with budesonide, even in excessive doses, is not expected to be a clinical problem. However, the plasma cortisol level will decrease and number and percentage of circulating neutrophils will increase. The number and percentage of lymphocytes and eosinophils will decrease concurrently. When used chronically in excessive doses, systemic glucocorticosteroid effects, such as hypercorticism and adrenal suppression, may appear.
Withdrawing Rilast Rapihaler or decreasing the dose of budesonide will abolish these effects, although the normalisation of the HPA-axis may be a slow process.
For information on the management of overdose, contact the Poison Information Centre on 131126 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action.
Rilast Rapihaler contains budesonide and formoterol, which have different modes of action and show additive effects in terms of reduction of asthma and COPD exacerbations. The specific properties of budesonide and formoterol allow the combination to be used either as an anti-inflammatory reliever or as maintenance treatment for asthma, and for symptomatic treatment of patients with moderate to severe COPD.
Budesonide is a non-halogenated glucocorticosteroid structurally related to 16α hydroxyprednisolone with a high local anti-inflammatory effect. Budesonide has shown anti-anaphylactic and anti-inflammatory effects in provocation studies in animals and humans, manifested as decreased bronchial obstruction in the immediate as well as the late phase of an allergic reaction. Budesonide has also been shown to decrease airway reactivity to both direct (histamine, methacholine) and indirect (exercise) challenge in hyper-reactive patients. Budesonide, when inhaled, has a rapid (within hours) and dose-dependent anti-inflammatory action in the airways, resulting in reduced symptoms and fewer exacerbations. Inhaled budesonide has less severe adverse effects than systemic corticosteroids. The exact mechanism responsible for the anti-inflammatory effect of glucocorticosteroids is unknown.
Formoterol is a potent selective β2-adrenergic agonist that produces relaxation of bronchial smooth muscle. Therefore, it has a bronchodilating effect in patients with reversible airways obstruction and in patients with bronchospasm induced by direct (methacholine) and indirect (e.g. exercise) stimuli. The bronchodilating effect is dose dependent with an onset of effect within 1 to 3 minutes after inhalation. The duration of effect is at least 12 hours after a single dose.
Clinical trials.
Asthma.
Therapeutic equivalence between budesonide/formoterol Rapihaler and budesonide/formoterol Turbuhaler was demonstrated in three clinical efficacy and safety studies in adults and adolescents with asthma. They included two randomised, double-blind, active controlled, parallel-group studies, Studies 681 (12 weeks duration) and 003 (6 weeks duration); and one randomised, open-label, parallel group, long-term (12 months) study, Study 715.
No clinical studies have been conducted to directly compare the efficacy and safety of budesonide/formoterol Rapihaler 100/3 with budesonide/formoterol Turbuhaler 200/6.
In Study 681, budesonide/formoterol Rapihaler 200/6 (2 inhalations twice daily) was compared with the corresponding dose of budesonide pMDI (200 microgram; 2 inhalations twice daily), or budesonide/formoterol Turbuhaler (200/6; 2 inhalations twice daily) in adults and adolescents (≥ 12 years) with moderate to severe asthma [e.g. mean forced expiratory volume during the first second (FEV1) ≥ 50% and ≤ 90% of predicted normal (PN) and FEV1 reversibility ≥ 12%]. Budesonide/formoterol Rapihaler was shown to significantly improve morning peak expiratory flow rate (primary efficacy variable), other lung function parameters, symptom scores and use of reliever medication compared to budesonide and was equivalent to budesonide/formoterol Turbuhaler (see Table 4).
 Study 715 investigated primarily the safety of budesonide/formoterol Rapihaler 200/6 (2 inhalations twice daily) during 12 months. The reference treatment was the corresponding dose of budesonide/formoterol Turbuhaler 200/6 and in a population consisting of adults and adolescents (≥ 12 years) with moderate to severe asthma (e.g. mean FEV1 of ≥ 50% of PN and FEV1 reversibility ≥ 12%). The study was of an open-label design.
Study 715 investigated primarily the safety of budesonide/formoterol Rapihaler 200/6 (2 inhalations twice daily) during 12 months. The reference treatment was the corresponding dose of budesonide/formoterol Turbuhaler 200/6 and in a population consisting of adults and adolescents (≥ 12 years) with moderate to severe asthma (e.g. mean FEV1 of ≥ 50% of PN and FEV1 reversibility ≥ 12%). The study was of an open-label design.
There was no statistically significant difference between budesonide/formoterol Rapihaler and budesonide/formoterol Turbuhaler regarding FEV1 and FVC (forced vital capacity). The percentage of patients experiencing one or more severe asthma exacerbations did not differ between the two budesonide/formoterol groups: 11% in the budesonide/formoterol Rapihaler group and 13% in the budesonide/formoterol Turbuhaler group. The maximum number of severe exacerbations per patient was 6 in the budesonide/formoterol Rapihaler group and 4 in the budesonide/formoterol Turbuhaler group. There was no statistical significant difference in time to first severe asthma exacerbation between the two treatment groups.
COPD.
The efficacy and safety of budesonide/formoterol as a fixed dose combination in the treatment of patients with moderate to severe COPD (pre-bronchodilator FEV1 ≤ 50% predicted normal) has been evaluated in four randomised, double-blind, placebo and active controlled, parallel-group, multi-centre clinical studies. Two 12-month studies were performed with the dry powder inhaler budesonide/formoterol Turbuhaler (studies 629 and 670), and one 12-month and one 6-month study were performed with the pressurised metered dose inhaler (pMDI) budesonide/formoterol Rapihaler (studies 001 and 002, respectively).
Studies 629 and 670. In both studies, budesonide/formoterol Turbuhaler 200/6 was compared with placebo and the corresponding mono-products (budesonide Turbuhaler 200 microgram and formoterol Turbuhaler 6 microgram), all taken as two inhalations twice daily. A total of 812 and 1022 patients with moderate to severe COPD were randomised, of which 208 and 254 were treated with budesonide/formoterol Turbuhaler. Patients in both studies had a mean age of 64 years and FEV1 of 0.99 L or 36% of predicted normal at baseline.
Studies 001 and 002. The study plans were similar. Both studies used budesonide/formoterol Rapihaler.
For Study 001, after a screening visit (visit 1), subjects entered a two weeks run-in period after which they were randomly assigned (visit 2) to one of the four following treatments:
1. Budesonide/formoterol Rapihaler 200/6, fixed combination of 200 microgram budesonide and 6 microgram formoterol per actuation, administered as 2 actuations twice daily.
2. Budesonide/formoterol Rapihaler 100/6 (never supplied in Australia; results not provided in this PI), fixed combination of 100 microgram budesonide and 6 microgram formoterol per actuation, administered as 2 actuations twice daily.
3. Formoterol Turbuhaler, 6 microgram per inhalation, administered as 2 actuations twice daily.
4. Placebo.
Study 002 had two additional treatment groups:
5. Budesonide pMDI 200 microgram per actuation, administered as 2 actuations twice daily.
6. Free combination of budesonide pMDI 200 microgram per actuation plus formoterol Turbuhaler 6 microgram per actuation, administered as 2 actuations of each twice daily.
A total of 1964 (Study 001) and 1704 (Study 002) patients with moderate to severe COPD were randomised, of which 494 and 277 were treated with budesonide/formoterol Rapihaler 200/6. The study populations had a mean age of 63 years and mean FEV1 of 1.04-1.05 L or 34% of predicted normal at baseline.
Study 629. In Study 629, efficacy was evaluated over 12 months using the co-primary endpoints of post-dose FEV1 and number of severe COPD exacerbations (defined as intake of a course of oral steroids and/or antibiotics and/or hospitalisation due to respiratory symptoms).
Budesonide/formoterol Turbuhaler significantly improved mean FEV1 compared with placebo and budesonide by 15% (p < 0.001) and 9% (p < 0.001) respectively.
Budesonide/formoterol Turbuhaler significantly reduced the number of severe exacerbations compared with placebo and formoterol by 24% (p=0.035) and 23% (p=0.043) respectively. The number needed to treat (NNT) to prevent one severe COPD exacerbation in a year for budesonide/formoterol Turbuhaler compared with formoterol was 2.4.
Study 670. In Study 670, efficacy was evaluated over 12 months using the co-primary endpoints of post dose-FEV1 and time to first severe COPD exacerbation (defined as intake of a course of oral steroids and/or antibiotics and/or hospitalisation due to respiratory symptoms).
Budesonide/formoterol Turbuhaler significantly improved mean FEV1 compared with placebo, budesonide and formoterol by 14% (p < 0.001), 11% (p < 0.001), and 5% (p=0.002) respectively.
Budesonide/formoterol Turbuhaler significantly prolonged the time to first severe COPD exacerbation compared to all comparator treatments. The instantaneous risk of experiencing a severe COPD exacerbation compared to placebo, budesonide and formoterol was reduced by 29% (p=0.006), 23% (p=0.033), and 30% (p=0.003) respectively.
Budesonide/formoterol Turbuhaler also significantly reduced the number of severe COPD exacerbations compared to placebo and formoterol by 24% (p=0.029) and 26% (p=0.015) respectively. The NNT to prevent one COPD exacerbation in a year compared to formoterol was 2.1.
Study 001. In Study 001, efficacy was evaluated over 12 months using the co-primary efficacy variables of change from baseline in average pre-dose and 1-hour post-dose FEV1 over the treatment period.
Primary endpoints.
Budesonide/formoterol Rapihaler 200/6 significantly improved 1-hour pre-dose FEV1 compared with formoterol and placebo by 0.04 L (p=0.008) and 0.09 L (p < 0.001) respectively.
Budesonide/formoterol Rapihaler 200/6 significantly improved post-dose FEV1 over the treatment period compared with formoterol and placebo by 0.03 L (p=0.023) and 0.18 L (p < 0.001) respectively.
Serial FEV1 measures over 12 hours were obtained in a subset of patients (N=491). The median time to onset of bronchodilation (> 15% improvement in FEV1) was seen within 5 minutes at the end of treatment time point in patients receiving budesonide/formoterol Rapihaler 200/6 (N=121). Maximum improvement in FEV1 occurred at approximately 2 hours post-dose and post-dose bronchodilator effect was maintained over 12 hours.
Exacerbations (secondary variable).
Budesonide/formoterol Rapihaler reduced the number of severe COPD exacerbations (defined as a worsening of COPD requiring oral steroid use and/or hospitalisation) to a statistically significant degree. Overall 34.1% of subjects experienced 1159 exacerbations: budesonide/formoterol Rapihaler 200/6, 30.8%; placebo 37.2%. The majority of exacerbations were treated with oral glucocorticosteroids: budesonide/formoterol Rapihaler 200/6, 96.5% of exacerbations; placebo 97.4%. Treatment comparisons were by means of rate ratios (RR) estimates, CIs and p-values derived from a Poisson regression adjusted for treatment, country and differential treatment exposure. Budesonide/formoterol Rapihaler 200/6 demonstrated a statistically significant reduction of 37% (p < 0.001) and 25% (p=0.004) in the rate of exacerbations per subject-treatment year compared with placebo and formoterol respectively.
Budesonide/formoterol Rapihaler 200/6 significantly prolonged the time to first severe COPD exacerbation compared to placebo, reducing the instantaneous risk of experiencing a severe COPD exacerbation by 26% (p=0.009). The NNT to prevent one severe COPD exacerbation in a year for budesonide/formoterol Rapihaler compared with formoterol was 5.4.
Study 002. In Study 002, efficacy was evaluated over 6 months using the co-primary efficacy variables of change from baseline in average pre-dose and 1-hour post-dose FEV1 over the treatment period.
Budesonide/formoterol Rapihaler 200/6 significantly improved pre-dose FEV1 compared with formoterol by 0.04 L (p=0.026) and compared with placebo and budesonide by 0.08 L (p < 0.001) for both comparators.
Budesonide/formoterol Rapihaler 200/6 significantly improved 1-hour post-dose FEV1 compared with formoterol by 0.04 L (p=0.039) and compared with placebo and budesonide by 0.17 L (p < 0.001) for both comparators.
Study 002 was not powered for showing effect on severe COPD exacerbations.
Serial FEV1 measures over 12 hours were obtained in subsets of patients (n=618). The median time to onset of bronchodilation (> 15% improvement in FEV1) was seen within 5 minutes at the end of treatment in patients receiving budesonide/formoterol Rapihaler 200/6 (N=101). Maximal improvement in FEV1 occurred at approximately 2 hours post-dose and post-dose bronchodilator effect was generally maintained over 12 hours.
5.2 Pharmacokinetic Properties
The budesonide and formoterol bioavailability of budesonide/formoterol Rapihaler and budesonide/formoterol Turbuhaler were similar after single doses containing 1280 microgram budesonide and 36 microgram formoterol (8 inhalations) in healthy adult volunteers. The budesonide and formoterol bioavailability from budesonide/formoterol Rapihaler was also comparable with that from similar doses of the component products, Pulmicort (budesonide) Turbuhaler, Oxis (formoterol) Turbuhaler and a specially prepared budesonide HFA pressurised inhalation suspension.
There was no evidence of pharmacokinetic interactions between budesonide and formoterol.
Absorption.
Inhaled budesonide is rapidly absorbed and the maximum plasma concentration is reached within 30 minutes after inhalation.
Inhaled formoterol is rapidly absorbed and the maximum plasma concentration is reached within 10 minutes after inhalation.
Distribution.
Plasma protein binding is approximately 90% for budesonide and volume of distribution is 3 L/kg.
Plasma protein binding is approximately 50% for formoterol and volume of distribution is about 4 L/kg.
Metabolism.
Budesonide undergoes an extensive degree (approximately 90%) of biotransformation on first passage through the liver to metabolites of low glucocorticosteroid activity. The glucocorticosteroid activity of the major metabolites, 6β-hydroxy-budesonide and 16α-hydroxy-prednisolone is less than 1% of that of budesonide.
Formoterol is inactivated via conjugation reactions (active-0-demethylated and deformylated metabolites are formed, but they are seen mainly as inactivated conjugates).
Excretion.
Budesonide is eliminated via metabolism mainly catalysed by the enzyme CYP3A4. The metabolites of budesonide are excreted in urine as such or in conjugated form. Only negligible amounts of unchanged budesonide have been detected in the urine. Budesonide has a high systemic clearance (approximately 1.2 L/min) and the plasma elimination half-life after i.v. dosing averages 4 hours.
The major part of a dose of formoterol is eliminated by metabolism in the liver followed by renal excretion. After inhalation of formoterol via a Turbuhaler, 8% to 13% of the delivered dose of formoterol is excreted unmetabolised in the urine. Formoterol has a high systemic clearance (approximately 1.4 L/min) and terminal elimination half-life averages 17 hours.
Special patient populations - children, elderly patients, renal and/or hepatic impairment.
The pharmacokinetics of budesonide or formoterol in children, elderly patients, and in patients with renal failure is unknown. The systemic availability of budesonide and formoterol may be increased in patients with liver disease.
5.3 Preclinical Safety Data
Genotoxicity.
Individually, budesonide and formoterol were not genotoxic in a series of assays for gene mutations (except for a slight increase in reverse mutation frequency in Salmonella typhimurium at high concentrations of formoterol), chromosomal damage and DNA repair. The combination of budesonide and formoterol has not been tested in genotoxicity assays.
Carcinogenicity.
The carcinogenic potential of the budesonide/formoterol combination has not been investigated in animal studies.
In formoterol carcinogenicity studies performed by AstraZeneca, there was a dose dependent increase in the incidence of uterine leiomyomas in mice dosed orally at 0.1, 0.5, and 2.5 mg/kg/day for 2 years, and a mesovarian leiomyoma was observed in a female rat dosed by inhalation at 0.13 mg/kg/day for 2 years. The effects observed are expected findings with high-dose exposure to β2-agonists.
Formoterol carcinogenicity studies performed by other companies used systemic exposure levels 800 to 4800-fold higher than those expected upon clinical use of formoterol (based on an 18 microgram daily dose).
Some carcinogenicity activity was observed in rats and mice. However, in view of the dose levels at which these effects were observed and the fact that formoterol is not mutagenic (except for very weak activity at high concentrations in one test system), it is concluded that the cancer risk in patients treated with formoterol fumarate is no greater than for other β2-adrenoceptor agonists.
The carcinogenic potential of budesonide has been evaluated in the mouse and rat at oral doses up to 200 and 50 microgram/kg/day respectively. In male rats dosed with 10, 25, and 50 microgram budesonide/kg/day, those receiving 25 and 50 microgram/kg/day showed an increased incidence of primary hepatocellular tumours. In a repeat study, this effect was observed in a number of steroid groups (budesonide, prednisolone, triamcinolone acetonide), thus indicating a class effect of corticosteroids.6 Pharmaceutical Particulars
6.1 List of Excipients
Rilast Rapihaler contains the inactive ingredients povidone (polyvinylpyrolidone K25), macrogol (polyethylene glycol) 1000 and apaflurane (known as hydrofluroalkane (HFA)-227).
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 30°C.
The inhaler should be discarded within 3 months after removal from the foil pouch.
6.5 Nature and Contents of Container
Rilast Rapihaler is a pressurised metered dose inhaler with an actuation counter. The inhaler is comprised of a pressurised aluminium canister with an attached actuation counter, a red plastic actuation body with a white mouthpiece and attached grey mouthpiece cover. Each inhaler is individually wrapped in a foil laminate pouch with desiccant sachet.
Two strengths of Rilast Rapihaler are registered (100/3 and 200/6 - see Section 2 Qualitative and Quantitative Composition) in packs of one inhaler containing 120 inhalations.
*Not all presentations may be available in Australia.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
Always be sure to dispose of Rilast Rapihaler responsibly, since some of the medicine may remain inside it. The canister in Rilast Rapihaler contains a pressurised liquid. The canister should not be broken, punctured or burnt, even when it seems empty.
6.7 Physicochemical Properties
Chemical structure.
Budesonide.
 Chemical name: 16α, 17α-22 R, S-propylmethylenedioxypregna-1, 4-diene-1β, 21-diol-3, 20-dione.
Chemical name: 16α, 17α-22 R, S-propylmethylenedioxypregna-1, 4-diene-1β, 21-diol-3, 20-dione.
Formoterol fumarate dihydrate.
 Chemical name: (R*R*)-(±)-N-[2-hydroxy-5-[1-hydroxy-2-[[2-(4-methoxyphenyl)-1-methylethyl]amino]ethyl]phenyl]formamide, (E)-2-butendioate(2:1), dihydrate.
Chemical name: (R*R*)-(±)-N-[2-hydroxy-5-[1-hydroxy-2-[[2-(4-methoxyphenyl)-1-methylethyl]amino]ethyl]phenyl]formamide, (E)-2-butendioate(2:1), dihydrate.
CAS number.
Budesonide.
51333-22-3.
Formoterol fumarate dihydrate.
183814-30-4.7 Medicine Schedule (Poisons Standard)
Prescription only medicine (Schedule 4).
Summary Table of Changes


 For the full list of excipients, see Section 6.1 List of Excipients.
For the full list of excipients, see Section 6.1 List of Excipients. As with other inhalation therapy, paradoxical bronchospasm may occur in very rare cases.
As with other inhalation therapy, paradoxical bronchospasm may occur in very rare cases. In these placebo-controlled studies, the incidence of pneumonia was low.
In these placebo-controlled studies, the incidence of pneumonia was low. Study 715 investigated primarily the safety of budesonide/formoterol Rapihaler 200/6 (2 inhalations twice daily) during 12 months. The reference treatment was the corresponding dose of budesonide/formoterol Turbuhaler 200/6 and in a population consisting of adults and adolescents (≥ 12 years) with moderate to severe asthma (e.g. mean FEV1 of ≥ 50% of PN and FEV1 reversibility ≥ 12%). The study was of an open-label design.
Study 715 investigated primarily the safety of budesonide/formoterol Rapihaler 200/6 (2 inhalations twice daily) during 12 months. The reference treatment was the corresponding dose of budesonide/formoterol Turbuhaler 200/6 and in a population consisting of adults and adolescents (≥ 12 years) with moderate to severe asthma (e.g. mean FEV1 of ≥ 50% of PN and FEV1 reversibility ≥ 12%). The study was of an open-label design. Chemical name: 16α, 17α-22 R, S-propylmethylenedioxypregna-1, 4-diene-1β, 21-diol-3, 20-dione.
Chemical name: 16α, 17α-22 R, S-propylmethylenedioxypregna-1, 4-diene-1β, 21-diol-3, 20-dione. Chemical name: (R*R*)-(±)-N-[2-hydroxy-5-[1-hydroxy-2-[[2-(4-methoxyphenyl)-1-methylethyl]amino]ethyl]phenyl]formamide, (E)-2-butendioate(2:1), dihydrate.
Chemical name: (R*R*)-(±)-N-[2-hydroxy-5-[1-hydroxy-2-[[2-(4-methoxyphenyl)-1-methylethyl]amino]ethyl]phenyl]formamide, (E)-2-butendioate(2:1), dihydrate.