What is in this leaflet
This leaflet answers some common questions about Sabril.
It does not contain all the available information. It does not take the place of talking to your doctor or pharmacist.
All medicines have risks and benefits.
Your doctor has weighed the risks of you taking Sabril against the benefits this medicine is expected to have for you.
If you have any concerns about taking this medicine, ask your doctor or pharmacist.
Keep this leaflet with the medicine. You may need to read it again.
What Sabril is used for
Sabril is a medicine used to help control epilepsy, a condition where you have repeated fits or convulsions. Sabril is used in addition to other medicines to treat epilepsy.
Sabril is an anticonvulsant. It works by stopping the breakdown of an important chemical transmitter ("messenger") in the brain and this helps reduce seizure activity.
Your doctor, however, may have prescribed Sabril for another reason.
Ask your doctor if you have any questions about why it has been prescribed for you.
This medicine is available only with a doctor's prescription.
Before you take it
When you must not take it
Do not take Sabril if you are allergic to it or any of the ingredients listed at the end of this leaflet.
Some symptoms of an allergic reaction include skin rash, itching, shortness of breath or swelling of the face, lips or tongue, which may cause difficulty in swallowing or breathing.
Do not take it if you are pregnant or intend to become pregnant. It may affect your developing baby if you take it during pregnancy.
Do not take it if you are breast-feeding or planning to breast-feed. Sabril passes into breast milk and there is a possibility your baby may be affected.
Do not take it after the expiry date (EXP) printed on the pack. If you take this medicine after the expiry date has passed, it may not work as well.
Do not take it if the packaging is torn or shows signs of tampering.
Before you start to take it
Tell your doctor if you have allergies to:
- any of the ingredients listed at the end of this leaflet
- any other medicines
- any other substances, such as foods, preservatives or dyes
Tell your doctor if you have or have had any medical conditions, especially the following:
- kidney problems
- nervous or mental illness
- depression
- psychosis, a severe mental condition in which the person is unable to think and judge clearly.
Tell your doctor if you are pregnant or intend to become pregnant. Like most medicines for epilepsy, Sabril may affect your unborn baby. However, it is very important to control your epilepsy while you are pregnant, since seizures may also affect your baby. Your doctor will help you decide whether or not you should take Sabril during pregnancy.
Tell your doctor if you are breast-feeding or plan to breast-feed. Sabril passes into breast milk. Your doctor will discuss the risks and benefits of using it if you are breast-feeding or planning to breast-feed.
Tell your doctor or pharmacist if you plan to have surgery.
If you have not told your doctor about any of the above, tell them before you take Sabril.
Your doctor may want to test your eyesight before you start taking Sabril and during the course of your treatment so that any changes in your sight can be detected. Some anti-epilepsy medicines may affect eyesight. In rare cases, patients treated with Sabril have experienced changes to their peripheral vision (i.e., they had trouble seeing things that were not directly in front of them).
Please ask your doctor if you want more information about this.
Taking other medicines
Tell your doctor or pharmacist if you are taking any other medicines, including any that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interfere with the absorption of Sabril. These include phenytoin, a medicine used to treat seizures.
Other medicines can increase the drowsiness that Sabril causes. These include clonazepam, a medicine used as a sedative or to treat anxiety.
These medicines may be affected by Sabril. You may need to use different amounts of your medicine, or take different medicines.
Your doctor will advise you.
Your doctor or pharmacist has more information about medicines to be careful with or avoid while taking Sabril.
How to take it
Follow all directions given to you by your doctor and pharmacist carefully.
These directions may differ from the information contained in this leaflet.
How much to take
Sabril tablets and sachets are taken by mouth either once or twice a day.
The dose of Sabril varies between patients. Your doctor may have prescribed a different dose.
Ask your doctor or pharmacist if you are unsure of the correct dose for you. They will tell you how much to take.
Follow the instructions they give you. If you take the wrong dose, Sabril may not work as well and your problem may not improve.
How to take it
Tablets
Swallow your tablet(s) whole with plenty of water. Do not crush or chew the tablet(s).
Sachets
The contents of the sachets should be dissolved in half a glass of water, juice or soft drink just before you plan to take it. Once dissolved, the solution should be taken immediately. It will dissolve easily and has no taste or smell.
Once mixed, any remaining solution should be discarded within 24 hours.
When to take it
You can take Sabril with or without food.
Take Sabril at about the same time each day. Taking Sabril at the same time each day will have the best effect. It will also help you remember when to take it.
If you are not sure when to take it, ask your doctor or pharmacist.
How long to take it
Do not stop taking Sabril until your doctor tells you to.
Ask your doctor or pharmacist if you are not sure how long to take the medicine for.
If you forget to take it
If it is almost time for your next dose, skip the dose you missed and take your next dose when you are meant to.
If there is still a long time to go before your next dose, take it as soon as you remember, and then go back to taking your dose as you would normally.
Do not take a double dose to make up for the dose that you missed.
If you are not sure what to do, ask your doctor or pharmacist.
If you have trouble remembering when to take your medicine, ask your pharmacist for hints.
If you take too much (overdose)
Immediately telephone your doctor or Poisons Information Centre (telephone Australia 13 11 26 or New Zealand 0800 POISON or 0800 764766) for advice, or go to Accident and Emergency at your nearest hospital if you think that you or anyone else may have taken too much Sabril. Do this even if there are no signs of discomfort or poisoning. You may need urgent medical attention.
While you are taking it
Things you must do
Tell all the doctors, dentists and pharmacists that are treating you that you are taking Sabril.
If you are about to start taking any new medicine, tell your doctor and pharmacist that you are taking Sabril.
If you plan to have surgery that needs a general anaesthetic, tell your doctor or dentist that you are taking this medicine.
If you become pregnant while you are taking Sabril, tell your doctor immediately.
Things you must not do
Do not stop taking Sabril until your doctor tells you to.
Do not give this medicine to anyone else, even if they have the same condition as you.
Do not take more than the recommended dose unless your doctor or pharmacist tells you to.
Do not use this medicine to treat any other complaints unless your doctor tells you to.
Things to be careful of
Be careful driving or operating machinery until you know how Sabril affects you. Sabril may cause dizziness or light-headedness in some people. Make sure you know how you react to Sabril before you drive a car, operate machinery or do anything else that could be dangerous if you are dizzy or light-headed. This effect decreases with time.
Side Effects
All medicines can cause side effects and this may occur with the normal use of Sabril. Sometimes they are serious, most of the time they are not.
Your doctor or pharmacist has weighed the risks of using this medicine against the benefits they expect it will have for you.
Do not be alarmed by this list of possible side effects. You may not experience any of them.
Tell your doctor or pharmacist as soon as possible if you do not feel well while you are taking Sabril.
It helps most people with epilepsy, but it may have unwanted side effects in a few people.
You may need medical treatment if you get some of the side-effects.
Tell your doctor if you notice any of the following and they worry you:
- drowsiness
- nervousness, irritability, depression
- dizziness
- confusion
- headache
- tiredness
- difficulty sleeping
- joints pain
- impaired concentration
- weight gain
- difficulty in speaking or slurred speech
- lack of coordination
- agitation, excitation
- aggression
- tremors
- nausea
- vomiting
- abdominal pain
- unusual hair loss or thinning
These are the most common side effects of this medicine.
Tell your doctor immediately if you notice any of the following:
- an increase in the number of seizures
- changes in your usual behaviour or mood
- severe sedation or confusion
- suicidal thoughts (thoughts of self-harm)
- suicide attempts
- changes in your eyesight (eg:blurred vision, tunnel vision)
Tell your Doctor immediately or go to the Accident and Emergency department of your nearest hospital if you have any thoughts of harming yourself or committing suicide.
Do not stop taking Sabril without consulting your doctor first.
If any of the following happen, stop taking this medicine and tell your doctor immediately, or go to Accident and Emergency at your nearest hospital:
- swelling of the face, lips, mouth or throat, which may cause difficultly in swallowing or breathing
- hives
These are very serious side effects. If you have them, you may have had a serious allergic reaction to Sabril. You may need urgent medical attention or hospitalisation.
Other side effects not listed above may occur in some patients.
Tell your doctor or pharmacist if you notice anything else that is making you feel unwell.
Ask your doctor or pharmacist to answer any questions you may have.
After taking it
If you have any queries about any aspect of your medicine, or any questions regarding the information in this leaflet, discuss them with your doctor or pharmacist.
Storage
Keep your tablets or sachets in the container until it is time to take them.
Keep Sabril in a cool dry place where the temperature stays below 30°C.
Do not store Sabril or any other medicine in the bathroom or near a sink.
Do not leave it in the car or on window sills. Heat and dampness can destroy some medicines.
Keep Sabril where children cannot reach it.
Disposal
If your doctor tells you to stop taking Sabril tablets or sachets or the expiry date has passed, ask your pharmacist what to do with any tablets or sachets that are left over.
Return any unused medicine to your pharmacist.
Product description
What it looks like
Tablets
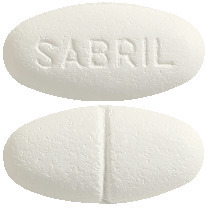
Sabril 500 mg are white to off-white, biconvex oval shaped tablets with a single breakline on one face and Sabril inscribed in the other. They come in a blister pack of 100 tablets.
Sachets
Sabril sachets contain 500 mg of vigabatrin. They come in a pack of 60 sachets.
Ingredients
Sabril does not contain gluten, sucrose, tartrazine or any other azo dyes.
Tablets
Active ingredient:
Each tablet contains 500 mg of vigabatrin.
Inactive ingredients:
Each tablet also contains povidone, microcrystalline cellulose, sodium starch glycollate, magnesium stearate, hypromellose, titanium dioxide, macrogol 8000, and Opadry White OY-S-7298
Sachets
Active ingredient:
Each sachet contains 500 mg of vigabatrin.
Inactive ingredients:
Each sachet also contains povidone.
Manufacturer
Sabril is supplied in Australia by:
sanofi-aventis australia pty ltd
12-24 Talavera Road
Macquarie Park NSW 2113
Sabril is supplied in New Zealand by:
sanofi-aventis new zealand limited
Level 8, 56 Cawley Street
Ellerslie
Auckland 1051
This leaflet was prepared in August 2019
Australian Register Numbers
Sabril tablets: AUST R 150021
Sabril sachets: AUST R 52985
sabril-ccdsv13-cmiv8-19aug19
Published by MIMS October 2019

 The relative risk for suicidal thoughts or behaviour was higher in clinical trials for epilepsy than in clinical trials for psychiatric or other conditions, but the absolute risk differences were similar for the epilepsy and psychiatric indications. Anyone considering prescribing vigabatrin or any other AED must balance this risk with the risk of untreated illness. Epilepsy and many other illnesses for which AEDs are prescribed are themselves associated with morbidity and mortality and an increased risk of suicidal thoughts and behaviour. Should suicidal thoughts and behaviour emerge during treatment, the prescriber needs to consider whether the emergence of these symptoms in any given patient may be related to the illness being treated. Patients, their caregivers, and families should be informed that AEDs increase the risk of suicidal thoughts and behaviour and should be advised of the need to be alert for the emergence or worsening of the signs and symptoms of depression, any unusual changes in mood or behaviour, or the emergence of suicidal thoughts, behaviour, or thoughts about self harm. Behaviours of concern should be reported immediately to the treating doctor.
The relative risk for suicidal thoughts or behaviour was higher in clinical trials for epilepsy than in clinical trials for psychiatric or other conditions, but the absolute risk differences were similar for the epilepsy and psychiatric indications. Anyone considering prescribing vigabatrin or any other AED must balance this risk with the risk of untreated illness. Epilepsy and many other illnesses for which AEDs are prescribed are themselves associated with morbidity and mortality and an increased risk of suicidal thoughts and behaviour. Should suicidal thoughts and behaviour emerge during treatment, the prescriber needs to consider whether the emergence of these symptoms in any given patient may be related to the illness being treated. Patients, their caregivers, and families should be informed that AEDs increase the risk of suicidal thoughts and behaviour and should be advised of the need to be alert for the emergence or worsening of the signs and symptoms of depression, any unusual changes in mood or behaviour, or the emergence of suicidal thoughts, behaviour, or thoughts about self harm. Behaviours of concern should be reported immediately to the treating doctor.
