What is in this leaflet
What Saxenda® is and what it is used for
Before you use Saxenda®
How to use Saxenda®
While you are using Saxenda®
Things to be careful of
Side effects
After using Saxenda®
Product description
Further information
Instructions for use
This leaflet answers some common questions about Saxenda®. It does not contain all the available information. It does not take the place of talking to your doctor, nurse or pharmacist.
All medicines have risks and benefits. You should discuss with your doctor the risks of using Saxenda® against the expected benefits.
If you have any concerns about using this medicine, ask your doctor or pharmacist.
Keep this leaflet with the medicine. You may need to read it again.
What Saxenda® is and what it is used for
Saxenda® is a weight loss medicine that contains the active ingredient liraglutide. It is similar to a naturally occurring hormone called GLP-1 that is released from the intestine after a meal. Saxenda® is an injection that is used once a day.
Saxenda® is used for weight loss, in addition to diet and exercise, in adults aged 18 years and above who have:
- a BMI of 30 or more (obese), or
- a BMI of more than 27 and less than 30 (overweight) and weight related health problems (such as diabetes, high blood pressure, abnormal levels of fats in the blood or breathing problems during sleep, called ‘obstructive sleep apnoea’).
You should only continue using Saxenda® if you have lost at least 5% of your initial body weight after 12 weeks on the 3.0 mg/day dose. Consult your doctor before you continue.
Saxenda® should be used with a reduced calorie diet and increased physical activity.
BMI (Body Mass Index) is a simple measure of your weight in relation to your height. It is calculated by dividing your weight in kg by your height in metres squared.
Saxenda® works by acting on receptors in the brain that control your appetite, causing you to feel fuller and less hungry. This may help you eat less food and reduce your body weight.
Weight loss normally starts within 2 weeks and continues for 9 to 12 months on Saxenda® treatment. Saxenda® may then help you to maintain this new lower weight and help to prevent weight being regained.
Saxenda® should not be used in children and adolescents under 18 years. This is because the effects of this medicine have not been studied in this age group.
Saxenda® is not addictive.
Saxenda® is available only with a doctor’s prescription.
Ask your doctor if you have any questions about why Saxenda® has been prescribed for you.
Before you use Saxenda®
When you must not use it
Do not use Saxenda® if you are allergic to liraglutide or any of the ingredients listed at the end of this leaflet.
Some of the symptoms of an allergic reaction may include:
- redness, swelling, rash and itching at the injection site
- rash, itching or hives on the skin
- shortness of breath
- wheezing or difficulty breathing
- swelling of the face, lips, tongue or other parts of the body.
Do not use this medicine after the expiry date printed on the pack or if the packaging is torn or shows signs of tampering. If it has expired or is damaged, return it to your pharmacist for disposal.
Before you start to use it
Tell your doctor if:
- you have palpitations (you feel aware of your heartbeat) or if you have feelings of a racing heartbeat while at rest during Saxenda® treatment.
- you have diabetes. Do not use Saxenda® as a replacement for insulin. Saxenda® and insulin should not be used together.
- you have heart failure (disease of the heart with shortness of breath, and swelling of the feet or legs due to fluid build-up). The use of Saxenda® is not recommended if you have severe heart failure.
- you are taking any other weight loss medications, including prescription, over the counter or herbal medicines.
- you have inflammatory bowel disease, such as Crohn's disease.
- you have gastroparesis (a condition in which your stomach has difficulty in emptying food properly).
- you currently have or have had thyroid disease, including thyroid nodules and enlargement of the thyroid gland.
- you have kidney disease or are on dialysis (a treatment where your blood is filtered and purified using a machine).
- you have liver problems.
- you have or have had a disease of the pancreas.
- you have or have had depression, suicidal thoughts or any other major mental illness.
- you have other medical conditions or have taken other medicines that may have caused you to gain weight.
Drink plenty of fluids to avoid dehydration, particularly if you experience vomiting or diarrhoea when beginning treatment with Saxenda®. Dehydration can cause kidney problems that sometimes require dialysis.
Contact your doctor if you have any questions or concerns.
Stop taking Saxenda® and contact a doctor immediately if you experience severe pain in your upper abdomen, usually worse on the right side under the ribs. The pain may be felt through to your back or right shoulder. If you lose substantial weight, you are at risk of gallstones and thereby inflamed gallbladder.
There is little experience with Saxenda® in patients ≥ 75 years old. It is not recommended if you are 75 years or older.
Tell your doctor if you have allergies to any other medicines, foods, preservatives or dyes.
Tell your doctor if you are pregnant or plan to become pregnant. Use of this medicine in pregnancy has not been studied. Saxenda® should not be used during pregnancy. It is not known if Saxenda® may harm your unborn child.
Tell your doctor if you are breast-feeding or plan to breast-feed. It is not known if Saxenda® passes into breast milk. Do not use Saxenda® if you are breast-feeding.
If you have not told your doctor about any of the above, tell them before you use Saxenda®.
Taking other medicines
Tell your doctor or pharmacist if you are taking any other medicines, including any that you buy without a prescription from your pharmacy, supermarket, naturopath or health food shop.
In particular, tell your doctor or pharmacist if you are using medicines:
- for the treatment of type 2 diabetes, such as a sulfonylurea (including glimepiride or glibenclamide). You may get low blood sugar (hypoglycaemia) when you use these medicines with Saxenda®. Your doctor may adjust the dose of your diabetes medicine to prevent you from getting low blood sugar.
- to thin your blood (anticoagulants), such as warfarin or other types of medicines called ‘coumarin derivatives’. Your doctor may need to monitor you more closely.
- for high blood pressure, such as amlodipine, atenolol or bisoprolol or for heart disease, such as digoxin.
- for HIV, such as atazanavir or ritonavir.
Tell your doctor about any other medicines that you are taking. This is very important. Your doctor will advise you if it is alright to keep taking them or if you should stop taking them.
Your doctor and pharmacist have more information on medicines to be careful with or avoid while using Saxenda®.
How to use Saxenda®
Your doctor or pharmacist will have given you advice on how to use your medicine. Carefully follow all the directions. They may differ from the information contained in this leaflet.
If you do not understand the instructions, ask your doctor, nurse or pharmacist for help.
Your doctor will start you on a diet and exercise programme. Stay on this programme while you are using Saxenda®.
Saxenda® is supplied as a pre-filled pen containing liraglutide. The Saxenda® pen can give doses of 0.6 mg, 1.2 mg, 1.8 mg, 2.4 mg or 3.0 mg.
How much to use
Your doctor will tell you how much of this medicine you need to use each day.
- The usual starting dose is 0.6 mg once a day.
- Your doctor will tell you how long to keep taking this dose. It will be for at least one week.
- Your dose may then be increased by 0.6 mg each week until you reach the recommended dose of 3.0 mg once a day.
- Your doctor will tell you how much Saxenda® to use each week. Usually, you will be told to follow the table below:
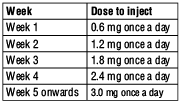
Once you reach the recommended dose of 3.0 mg in Week 5 of treatment, keep using this dose until your treatment period ends. Do not increase your dose further.
Visit your doctor on a regular basis to assess your need for continued treatment with Saxenda®.
Do not change your dose unless your doctor has told you to.
When to use it
You can use Saxenda® at any time of day, with or without food and drinks. It is preferable to use Saxenda® at about the same time each day, at a time that is most convenient for you.
How to use it
Before you use the pen for the first time, your doctor or nurse will show you how to use the pen.
- Saxenda® pre-filled pen is designed to be used with NovoFine® needles, up to a length of 8 mm. Needles are not included with the pen.
- Ask your doctor or nurse which needle width (gauge) and length is best for you.
- Use a new needle for each injection and dispose of it after use.
- Saxenda® is given as an injection under the skin (subcutaneous injection) as shown to you by your doctor or nurse. Never inject Saxenda® into a vein or muscle.
- Saxenda® may be injected into the front of your waist (abdomen), the front of your thigh, or your upper arm.
- Change the place within the area where you inject each day to reduce the risk of developing lumps under the skin. See ‘Side effects’.
Checking your Saxenda® pen:
Saxenda® should be clear and colourless.
Do not use this medicine if it is thickened, coloured, or has solid bits in it.
Read the instructions printed later in this leaflet carefully in order to prepare and handle your Saxenda® pen correctly.
How long to use it
Only continue using Saxenda® if you have lost at least 5% of yourinitial body weight after 12 weeks on the 3.0 mg/day dose (see ‘How much to use,’ above). Consult your doctor before you continue. Your doctor will assess your treatment on a regular basis.
If you use too much (overdose)
If you use more Saxenda® than you should, talk to your doctor or go to a hospital straight away. Take the medicine pack with you. You may need medical treatment. Using too much Saxenda® may cause nausea (feeling sick), vomiting (being sick), or low blood sugar (hypoglycaemia). See 'Side effects' for the early warning signs of hypos.
If you forget to use it
If you forget a dose and remember it within 12 hours from when you usually take the dose, inject Saxenda® as soon as you remember.
However, if more than 12 hours have passed since you should have used Saxenda®, skip the missed dose and inject your next dose the following day at the usual time.
Do not use an extra dose or increase the dose on the following day to make up for the missed dose.
If you are not sure what to do, talk to your doctor, nurse or pharmacist.
While you are using Saxenda®
Things you must do
Tell your doctor if you have diabetes. They may adjust the dose of your diabetes medicine to prevent you getting ‘hypos’ (low blood sugar levels).
- Do not mix Saxenda® with other medicines that you inject (e.g. insulins).
- Do not use Saxenda® in combination with other medicines that contain GLP-1 receptor agonists (e.g. exenatide or lixisenatide).
Tell your doctor, nurse or pharmacist if you are travelling. Ask them for a letter explaining why you are taking injecting devices with you. Each country you visit will need to see this letter, so you should take several copies.
You may not be able to get Saxenda® in the country you are visiting.
Your doctor, nurse or pharmacist can provide you with some helpful information.
Things you must not do
Do not stop using your medicine unless your doctor tells you to.
Do not use this medicine if you think it has been frozen or exposed to excessive heat.
Do not use this medicine to treat any other complaints unless your doctor tells you to.
Do not give your medicine to anyone else, even if they have the same condition as you.
Do not share needles or pens.
Things to be careful of
Be careful driving or operating machinery until you know how Saxenda® affects you. For further information talk to your doctor or pharmacist.
Side effects
Tell your doctor, nurse or pharmacist as soon as possible if you do not feel well while you are using Saxenda®.
All medicines can have side effects. Sometimes they are serious, most of the time they are not. You may need medical treatment if you get some of the side effects.
Do not be alarmed by the following lists of side effects. You may not experience any of them.
Ask your doctor, nurse or pharmacist to answer any questions you may have.
The most common side effects when using Saxenda® are:
- feeling sick (nausea)
- being sick (vomiting)
- diarrhoea
- constipation
- headache.
These side effects are usually mild and normally decrease with continued use.
Tell your doctor if you notice any of the following and they worry you:
- changed sense of taste
- dizziness
- indigestion (dyspepsia)
- feeling bloated, burping, or wind (flatulence)
- dry mouth
- heartburn
- painful or swollen abdomen
- low blood sugar (a ‘hypo’)
- injection site reactions (such as bruising, pain, irritation, itching and rash)
- fast heartbeat
- feeling weak or tired
- difficulty sleeping (insomnia).
This usually occurs during the first 3 months of treatment - increase of pancreatic enzymes (such as lipase and amylase). This can only be detected with a blood test.
The above list includes the more common side effects of your medicine. They are usually mild and short-lived.
Other, less common side effects include:
- feeling generally unwell.
- urticaria (a type of skin rash).
- loss of fluids (dehydration). This is more likely to occur at the start of treatment and may be due to you being sick (vomiting), feeling sick (nausea), and having diarrhoea.
Hypos are more likely to occur if you have diabetes and are also taking a sulfonylurea. A hypo may come on suddenly. The warning signs of a hypo can include:
- cold sweat, cool pale skin
- headache
- fast heartbeat
- feeling sick
- feeling very hungry
- changes in vision
- feeling sleepy
- feeling weak, nervous, anxious, or confused
- difficulty concentrating
- shaking (tremor)
Your doctor can provide you with further information about how to treat low blood sugar levels and what to do if you notice these warning signs. If you are already taking a sulfonylurea when you start using Saxenda® your doctor may tell you to reduce the dose of the sulfonylurea.
Lumps under the skin may be caused by build-up of a protein called amyloid (cutaneous amyloidosis; how often this occurs is not known). Saxenda® may not work very well if you inject into a lumpy area. Change the injection site with each injection to help prevent this skin change.
Tell your doctor immediately if you:
- experience severe and persistent pain in the abdomen (stomach area) which might reach through to your back, as well as nausea and vomiting. This could be a sign of an inflamed pancreas (pancreatitis). Pancreatitis is a serious, potentially life-threatening medical condition. Stop taking Saxenda® if you experience this.
- get a lump or swelling in your neck, hoarseness, trouble swallowing, or shortness of breath. These can be symptoms of a condition called ‘goitre’.
- experience the signs of reduced kidney function, which may include reduction in urine volume, a metallic taste in your mouth and bruising easily.
- experience upper right side abdominal pain, yellowing of the skin and eyes or clay coloured stools, which may be signs of gallbladder problems.
- experience depression or have any mental changes (especially sudden changes in mood, behaviours, thoughts or feelings) that are new, worse or worry you.
Tell your doctor if you notice anything else that is making you feel unwell.
Tell your doctor immediately or go to Accident and Emergency at your nearest hospital if you notice any of the following:
- skin rashes over a large part of the body
- shortness of breath, wheezing
- swelling of the face, lips or tongue
- fast heartbeat
- sweating.
This list includes very serious side effects that could be signs of a severe allergic reaction. You may need urgent medical attention or hospitalisation. These side effects are rare.
Other side effects not listed above may also occur in some people.
Ask your doctor, nurse or pharmacist to answer any questions you have.
After using Saxenda®
Storage
Store Saxenda® pens that are not being used between 2°C and 8°C in a refrigerator (not in or too near the freezer section or cooling element). Do not freeze. When Saxenda® is being used, you can keep the pen for 1 month either at room temperature (below 30˚C) or in a refrigerator (2˚C to 8˚C), away from the cooling element. Store the pen without a needle attached.
Discard Saxenda® after 1 month even if there is still some medicine left in it.
The medicine in Saxenda® must not be frozen, or exposed to heat or direct sunlight.
When you are not using the pen, keep the pen cap on in order to protect it from light.
Do not use Saxenda® after the expiry date printed on the pen label and carton. The expiry date refers to the last day of that month.
Do not use Saxenda® if the solution is not clear and colourless.
Keep it where children cannot reach it.
Disposal
Dispose of used needles safely into a yellow plastic sharps container.
If your doctor tells you to stop using this medicine or the expiry date has passed, ask your pharmacist what to do with any medicine that is left over.
Product description
What it looks like
Saxenda® is supplied as a clear and colourless solution for injection in a pre-filled pen. Each pen contains 3 mL of solution, and is able to deliver doses of 0.6 mg, 1.2 mg, 1.8 mg, 2.4 mg or 3.0 mg.
Saxenda® is available in packs containing 1, 3 or 5 pens. Not all pack sizes may be marketed.
Needles are not included.
Ingredients
Saxenda® contains liraglutide 6 mg/mL as the active ingredient.
Saxenda® also contains the following inactive ingredients: dibasic sodium phosphate dihydrate, propylene glycol, phenol, hydrochloric acid, sodium hydroxide and water for injections.
Supplier/Sponsor
Saxenda® is supplied in Australia by:
Novo Nordisk Pharmaceuticals Pty. Ltd.
Level 10
118 Mount Street
North Sydney NSW 2060
Australia
Saxenda® is supplied in New Zealand by:
Novo Nordisk Pharmaceuticals Ltd.
11-19 Customs Street West,
Commercial Bay Tower,
Level 18, Office 1834
Auckland 1010
New Zealand
Australian Registration Number:
AUST R 225804
Saxenda®, NovoFine® and NovoCare® are trademarks owned by Novo Nordisk A/S.
© 2022
Novo Nordisk A/S
Further information
For further information call the NovoCare® Customer Care Centre on 1800 668 626 (AU) or 0800 733 737 (NZ).
Always check the following websites to ensure you are reading the most recent version of the Saxenda® consumer medicine information:
www.novonordisk.com.au (AU)
https://www.ebs.tga.gov.au/ (AU)
www.novonordisk.co.nz (NZ)
https://medsafe.govt.nz/(NZ)
For further dietary advice, refer to the dietary guidelines on the NH&MRC website at www.nhmrc.gov.au (Australia), or the current food and nutrition guidelines on the Ministry of Health website at www.health.govt.nz (New Zealand).
This leaflet was prepared in August 2022.
Instructions on how to use Saxenda® 6 mg/mL solution for injection in pre-filled pen
Please read these instructions carefully before using your Saxenda® pre-filled pen.
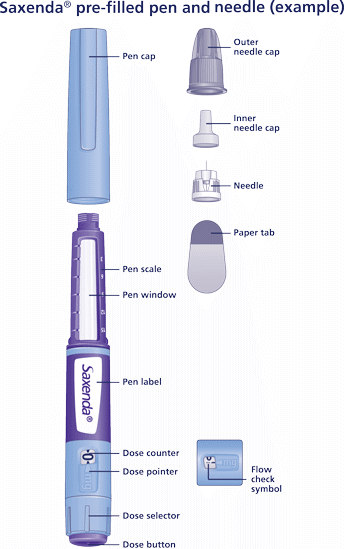
Do not use the pen without proper training from your doctor or nurse. Start by checking your pen to make sure that it contains Saxenda® 6 mg/mL, then look at the illustrations below to get to know the different parts of your pen and needle.
If you are blind or have poor eyesight and cannot read the dose counter on the pen, do not use this pen without help. Get help from a person with good eyesight who is trained to use the Saxenda® pre-filled pen.
Your pen is a pre-filled dial-a-dose pen. It contains 18 mg of liraglutide, and delivers doses of 0.6 mg, 1.2 mg, 1.8 mg, 2.4 mg and 3.0 mg. Your pen is designed to be used with NovoFine® disposable needles up to a length of 8 mm.
Needles are not included in the pack.
Important information
Pay special attention to these notes as they are important for safe use of the pen.
1 Prepare your pen with a new needle
- Check the name and coloured label of your pen, to make sure that it contains Saxenda®. This is especially important if you take more than one type of injectable medicine. Using the wrong medicine could be harmful to your health.
- Pull off the pen cap.
- Check that the solution in your pen is clear and colourless. Look through the pen window. If the solution looks cloudy, do not use the pen.
- Take a new NovoFine® needle and tear off the paper tab.
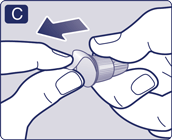
Make sure to attach the needle correctly.
- Push the needle straight onto the pen.
- Turn until it is on tight.
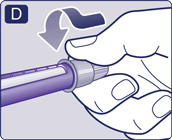
The needle is covered by two caps. You must remove both caps. If you forget to remove both caps, you will not inject any solution.
- Pull off the outer needle cap and keep it for later. You will need it after the injection, to safely remove the needle from the pen.
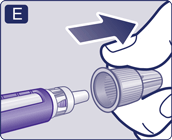
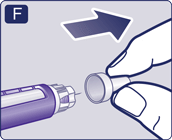
- Pull off the inner needle cap and throw it away. If you try to put it back on, you may accidentally stick yourself with the needle.
A drop of solution may appear at the needle tip. This is normal, but you must still check the flow, if you use a new pen for the first time.
Do not attach a new needle to your pen until you are ready to take your injection.
Always use a new needle for each injection.
This may prevent blocked needles, contamination, infection and inaccurate dosing.
Never use a bent or damaged needle.
2 Check the flow with each new pen
- If your pen is already in use, go to step 3 ‘Select your dose’. Only check the flow before your first injection with each new pen.
- Turn the dose to the flow check symbol (
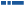 ) right past 0. Make sure the flow check symbol lines up with the pointer.
) right past 0. Make sure the flow check symbol lines up with the pointer.
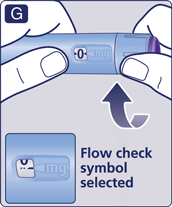
- Hold the pen with the needle pointing up.
Press and hold in the dose button until the dose counter returns to 0.
The 0 must line up with the dose pointer.
A drop of solution should appear at the needle tip.
A small drop may remain at the needle tip, but it will not be injected.
If no drop appears, repeat step 2 ‘Check the flow with each new pen’ up to 6 times. If there is still no drop, change the needle and repeat step 2 ‘Check the flow with each new pen’ once more. If a drop still does not appear, dispose of the pen and use a new one.
Always make sure that a drop appears at the needle tip before you use a new pen for the first time. This makes sure that the solution flows. If no drop appears, you will not inject any medicine, even though the dose counter may move. This may indicate a blocked or damaged needle.
If you do not check the flow before your first injection with each new pen, you may not get the prescribed dose and the intended effect of Saxenda®.
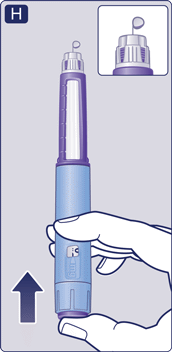
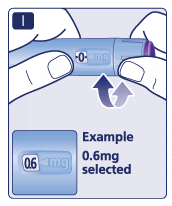
3 Select your dose
- Turn the dose selector until the dose counter shows your dose (0.6 mg, 1.2 mg, 1.8 mg, 2.4 mg or 3.0 mg).
If you select the wrong dose, you can turn the dose selector forward or backwards to the correct dose.
The pen can dial up to a maximum of 3.0 mg.
The dose selector changes the dose. Only the dose counter and dose pointer will show how many mg you select per dose.
You can select up to 3.0 mg per dose. When your pen contains less than 3.0 mg the dose counter stops before 3.0 is shown.
The dose selector clicks differently when turned forward, backwards or past the number of mg left. Do not count the pen clicks.
Always use the dose counter and the dose pointer to see how many mg you have selected before injecting this medicine.
Do not count the pen clicks.
Do not use the pen scale. It only shows approximately how much solution is left in your pen.
Only doses of 0.6 mg, 1.2 mg, 1.8 mg, 2.4 mg or 3.0 mg must be selected with the dose selector. The selected dose must line up precisely with the dose pointer to ensure that you get a correct dose.
How much solution is left?
- The pen scale shows you approximately how much solution is left in your pen.

- To see precisely how much solution is left, use the dose counter: Turn the dose selector until the dose counter stops. If it shows 3.0, at least 3.0 mg are left in your pen. If the dose counter stops before 3.0 mg, there is not enough solution left for a full dose of 3.0 mg.
If you need more medicine than what is left in your pen
Only if trained or advised by your doctor or nurse, you may split your dose between your current pen and a new pen. Use a calculator to plan the doses as instructed by your doctor or nurse.
Be very careful to calculate correctly.
If you are not sure how to split your dose using two pens, then select and inject the dose you need with a new pen.
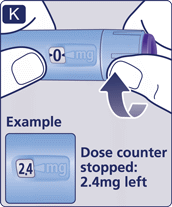
4 Inject your dose
- Insert the needle into your skin as your doctor or nurse has shown you.
- Make sure you can see the dose counter. Do not cover it with your fingers. This could interrupt the injection.
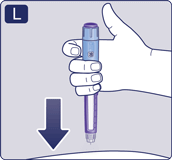
- Press and hold down the dose button. Watch as the dose counter returns to 0. The 0 must line up with the dose pointer. You may then hear or feel a click.
- Continue pressing the dose button while keeping the needle in your skin.
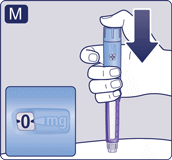
- Count slowly to 6 while keeping the dose button pressed.
- If the needle is removed earlier, you may see a stream of solution coming from the needle tip. If so, the full dose will not be delivered.

- Remove the needle from your skin. You can then release the dose button.
If blood appears at the injection site, press lightly.
You may see a drop of solution at the needle tip after injecting. This is normal and does not affect your dose.
Always watch the dose counter to know how many mg you inject.
Hold the dose button down until the dose counter shows 0.
How to identify a blocked or damaged needle?
- If 0 does not appear in the dose counter after continuously pressing the dose button, you may have used a blocked or damaged needle.
- In this case - you have not received any medicine - even though the dose counter has moved from the original dose that you have set.
How to handle a blocked needle?
Change the needle as described in step 5 ‘After your injection’, and repeat all steps starting with step 1‘Prepare your pen with a new needle’. Make sure you select the full dose you need.
Never touch the dose counter when you inject. This can interrupt the injection.
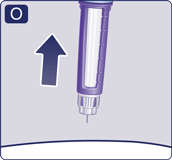
5 After your injection
- Always dispose of the needle after each injection to ensure convenient injections and prevent blocked needles. If the needle is blocked, you will not inject any medicine.
- Lead the needle tip into the outer needle cap on a flat surface without touching the needle or the outer needle cap.
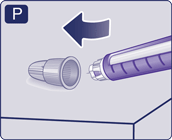
- Once the needle is covered, carefully push the outer needle cap completely on.
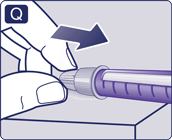
- Unscrew the needle and dispose of it carefully, as instructed by your doctor, nurse, pharmacist or local authorities.
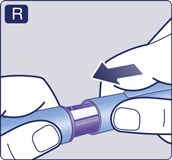
- Put the pen cap on your pen after each use to protect the solution from light.
When the pen is empty, throw it away without a needle on as instructed by your doctor, nurse, pharmacist or local authorities.
Never try to put the inner needle cap back on the needle. You may stick yourself with the needle.
Always remove the needle from your pen after each injection.
This may prevent blocked needles, contamination, infection, leakage of solution and inaccurate dosing.
Further important information
- Always keep your pen and needles out of sight and reach of others, especially children.
- Never share your pen or your needles with other people.
- Caregivers must be very careful when handling used needles - to prevent needle injury and cross-infection.
Caring for your pen
- Do not leave the pen in a car or other place where it can get too hot or too cold.
- Do not inject Saxenda® which has been frozen. If you do that, you may not get the intended effect of this medicine.
- Do not expose your pen to dust, dirt or liquid.
- Do not wash, soak or lubricate your pen. It may be cleaned with a mild detergent on a moistened cloth.
- Do not drop your pen or knock it against hard surfaces. If you drop it or suspect a problem, attach a new needle and check the flow before you inject.
- Do not try to refill your pen. Once empty, it must be disposed of.
- Do not try to repair your pen or pull it apart.
Published by MIMS October 2022

 The need for continued treatment should be re-evaluated whenever a new prescription is written and at least annually.
The need for continued treatment should be re-evaluated whenever a new prescription is written and at least annually.








