What is in this leaflet
Please read this leaflet carefully before you start using SHINGRIX. This leaflet answers some common questions about SHINGRIX. It does not contain all the available information. It does not take the place of talking to your doctor, nurse or pharmacist.
All medicines and vaccines have risks and benefits. Your doctor has weighed the risks of you receiving SHINGRIX against the benefits they expect it will have for you.
If you have any concerns about taking SHINGRIX, ask your doctor, nurse or pharmacist.
Keep this leaflet with the vaccine You may need to read it again.
What SHINGRIX is used for
SHINGRIX is a vaccine that helps to protect adults against herpes zoster (also called shingles) as well as the long-lasting nerve pain that can follow shingles called post-herpetic neuralgia or PHN.
SHINGRIX can be given to adults from the age of 50 years and above.
What is shingles and what are the complications related to shingles?
Shingles is caused by the same virus that causes chickenpox. After you have had chickenpox (primary varicella infection), the virus that caused it stays in your body in nerve cells. Sometimes, after many years, the virus can become active again and causes shingles (secondary varicella infection).
SHINGRIX cannot be used to prevent chickenpox (primary varicella infection).
Shingles is a painful, blistering rash. It usually occurs in one part of the body and can last for several weeks. It may lead to complications such as long-lasting pain (PHN) and scarring. Less commonly, bacterial skin infections, weakness, muscle paralysis, loss of hearing or vision can occur.
What is post-herpetic neuralgia (PHN)?
After the shingles blisters heal, pain can last for months or years and may be severe. This long-lasting nerve pain is called PHN. PHN is the most common complication of shingles.
How does the vaccine work?
SHINGRIX helps your body to build its own protection against shingles and its complications.
SHINGRIX cannot be used to treat shingles or the pain associated with shingles. It is for the protection against shingles only.
As with all vaccines, SHINGRIX may not fully protect all people who are vaccinated.
Ask your doctor if you have any questions about why this medicine has been prescribed for you.
This medicine is available only with a doctor's prescription.
Before you are given SHINGRIX
When you must not be given SHINGRIX
Do not have SHINGRIX if you are allergic (hypersensitive) to any of the ingredients contained in SHINGRIX. Signs of an allergic reaction may include itchy skin rash, shortness of breath and swelling of the face or tongue. Tell your doctor if you have experienced any of these symptoms with a previous dose of SHINGRIX or any other vaccine or medicine.
Do not use this vaccine after the expiry date printed on the pack or if the packaging is torn or shows signs of tampering. If it has expired or is damaged, return it to your pharmacist for disposal.
If you are not sure whether you should receive SHINGRIX, talk to your doctor.
Before being given SHINGRIX
Tell your doctor or nurse before you receive SHINGRIX:
- if you have an infection with a high temperature. In these cases, the vaccination may be postponed until recovery. A minor infection such as a cold should not be a problem, but talk to your doctor first.
- if you have a bleeding problem or bruise easily.
- if you fainted with a previous injection. Fainting can occur following, or even before, any needle injection.
- if you have been told by your doctor that you have a weakened immune system as a result of a disease, medications, (including high-dose corticosteroids or cancer medicines), or other treatment.
Tell your doctor if you have allergies to any other medicines, foods, preservatives or dyes.
Tell your doctor if you are pregnant or plan to become pregnant or are breast-feeding. Your doctor can discuss with you the risks and benefits involved.
If you have not told your doctor about any of the above, tell him/her before you receive SHINGRIX.
Taking other medicines
Tell your doctor or pharmacist if you are taking or have recently taken any other medicines, including any that you get without a prescription from your pharmacy, supermarket or health food shop. Some medicines and SHINGRIX may interfere with each other.
Having other vaccines
Tell your doctor or nurse if you have received another vaccine recently.
Some vaccines may be affected by other vaccines.
SHINGRIX can be given at the same time as the yearly influenza (flu) vaccine. If SHINGRIX is given at the same time as the influenza vaccine, a different injection site will be used for each vaccine.
How SHINGRIX is given
The doctor or nurse will give SHINGRIX as an injection.
If you have any concerns about how this vaccine is to be given, talk to your doctor, nurse or pharmacist.
How it is given
SHINGRIX is given as an injection of 0.5 ml into a muscle (usually in the upper arm).
When it is given
You will receive 2 injections with an interval of 2 to 6 months.
The first injection can be given from the age of 50 years. You will be informed when you should come back for the second dose of SHINGRIX.
Make sure you finish the complete vaccination course. This will maximise the protection offered by SHINGRIX.
If a dose is missed
If you miss a scheduled dose, talk to your doctor or nurse and arrange another visit as soon as possible.
If you are not sure what to do, ask your doctor, nurse or pharmacist.
While being given SHINGRIX
Things you must do
Keep the follow up visits with the doctor or clinic. Make sure you finish the complete vaccination course. This will maximise the protection offered by SHINGRIX.
Things to be careful of
There is no information on whether SHINGRIX affects the ability to drive or use machines.
However, do not drive or use machines if you are feeling unwell.
Side effects
Tell your doctor or pharmacist as soon as possible if you do not feel well during or after having had a dose of SHINGRIX.
This vaccine helps protect most people from shingles, but it may have unwanted side effects in some people. All medicines and vaccines can have side effects. Sometimes they are serious, most of the time they are not. You may need medical attention if you get some of the side effects.
Do not be alarmed by the following lists of side effects. You may not experience any of them.
Ask your doctor or pharmacist to answer any questions you may have.
The following side effects may occur after receiving SHINGRIX:
Very common (these may occur with more than 1 in 10 doses of the vaccine):
- headache
- stomach and digestive complaints (including nausea, vomiting, diarrhoea and/or stomach pain)
- muscle pain (myalgia)
- pain, redness and swelling at the injection site
- tiredness, chills, fever
Common (these may occur with up to 1 in 10 doses of the vaccine):
- injection site itching (pruritus)
- generally feeling unwell
If any of the side effects gets serious, or if you notice any side effects not listed in this leaflet, tell your doctor or pharmacist.
As with all vaccines given by injection there is a very small risk of serious allergic reaction. Contact your doctor immediately or go to the casualty department of your nearest hospital if any of the following happens:
- swelling of limbs, face, eyes, inside of nose, mouth or throat
- shortness of breath, breathing or swallowing difficulties
- hives, itching (especially of the hands or feet), reddening of skin (especially around the ears), or severe skin reactions
- unusual tiredness or weakness that is sudden and severe
Allergy to SHINGRIX is rare (may occur with up to 1 in 1,000 doses of the vaccine). Any such severe reactions will usually occur within the first few hours of vaccination. Tell your doctor or pharmacist if you notice anything that is making you feel unwell during or after a dose of vaccine.
Other side effects not listed above, can also occur during or soon after a dose of SHINGRIX.
Check with your doctor or nurse if you have any other side effects.
How to store SHINGRIX
Storage
- Keep this medicine out of the sight and reach of children.
- Store in a refrigerator (2°C – 8°C) Do not freeze.
- Store in the original package in order to protect from light.
- Do not use this medicine after the expiry date which is stated on the label and carton after EXP. The expiry date refers to the last day of that month.
Disposal
Ask your pharmacist what to do with any SHINGRIX that has expired or has not been used.
Product description
What it looks like
The powder is white.
The suspension is an opalescent, colourless to pale brownish liquid.
One pack of SHINGRIX consists of:
- Powder for 1 dose in a vial
- Suspension for 1 dose in a vial
SHINGRIX is available in pack sizes of:
- 1 vial of powder plus 1 vial of suspension
- 10 vials of powder plus 10 vials of suspension.
Ingredients
1 dose (0.5 ml) of SHINGRIX contains 50 micrograms of glycoprotein E (gE) powder mixed with AS01B adjuvant suspension.
gE is a protein present in the Varicella Zoster Virus. This protein is not infectious.
The adjuvant is composed of a plant extract (Quillaja saponaria saponin (QS-21) (50 micrograms) and a bacterial extract (3-O-desacyl-4’- monophosphoryl lipid A (MPL) (50 micrograms)) and is used to improve the body’s response to the vaccine.
The other ingredients are:
Powder
- Sucrose
- Polysorbate 80
- Monobasic sodium phosphate dihydrate
- Dibasic potassium phosphate
Suspension
- Dioleoylphosphatidylcholine
- Cholesterol
- Sodium chloride
- Dibasic sodium phosphate
- Monobasic potassium phosphate
- Water for injections
Supplier
GlaxoSmithKline Australia Pty Ltd
Level 4, 436 Johnston Street
Abbotsford, Victoria 3067 Australia.
Registration numbers: AUST R 289257
Date of preparation: 06 December 2018
Version 2.0
Trade marks are owned by or licensed to the GSK group of companies
©2018 GSK group of companies or its licensor
Published by MIMS June 2021

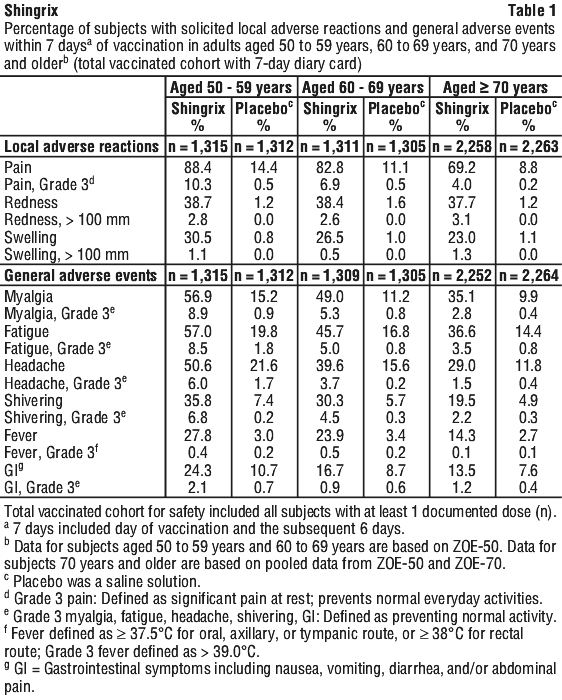 The incidence of solicited local and general symptoms was lower in subjects aged 70 years and older compared with those aged 50 to 69 years.
The incidence of solicited local and general symptoms was lower in subjects aged 70 years and older compared with those aged 50 to 69 years. In studies with immunocompromised adults aged 18 years and older, there was a higher incidence of pain at the injection site (90% and 82%), fatigue (65% and 56%), myalgia (61% and 50%), headache (49% and 32%), shivering (33% and 25%) and fever (30% and 19%) in subjects aged 18 to 49 years compared with those aged 50 years and older.
In studies with immunocompromised adults aged 18 years and older, there was a higher incidence of pain at the injection site (90% and 82%), fatigue (65% and 56%), myalgia (61% and 50%), headache (49% and 32%), shivering (33% and 25%) and fever (30% and 19%) in subjects aged 18 to 49 years compared with those aged 50 years and older.
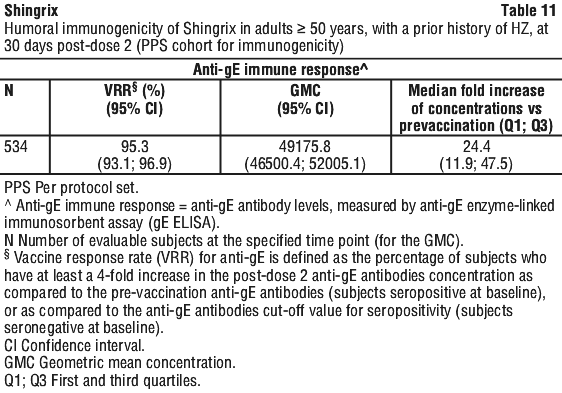
 In the fourth year after vaccination, the efficacy against HZ was 93.1% (95% CI: 81.2; 98.2) and 87.9% (95% CI: 73.3; 95.4) in subjects ≥ 50 years (Zoster-006) and subjects ≥ 70 years, (pooled Zoster-006 and Zoster-022), respectively.
In the fourth year after vaccination, the efficacy against HZ was 93.1% (95% CI: 81.2; 98.2) and 87.9% (95% CI: 73.3; 95.4) in subjects ≥ 50 years (Zoster-006) and subjects ≥ 70 years, (pooled Zoster-006 and Zoster-022), respectively. The benefit of Shingrix in the prevention of PHN can be attributed to the effect of the vaccine on the prevention of HZ. Due to the very low numbers of shingles occurring in the Shingrix group, the efficacy of Shingrix in the prevention of PHN in subjects with confirmed HZ could not be demonstrated.
The benefit of Shingrix in the prevention of PHN can be attributed to the effect of the vaccine on the prevention of HZ. Due to the very low numbers of shingles occurring in the Shingrix group, the efficacy of Shingrix in the prevention of PHN in subjects with confirmed HZ could not be demonstrated. In the eleventh year after vaccination, the efficacy against HZ was 82.0% (95% CI: 63.0; 92.2) and 72.0% (95% CI: 33.4; 89.8) in subjects ≥ 50 years (Shingrix group: N=5,849) and subjects ≥ 70 years (Shingrix group: N=2,891), respectively.
In the eleventh year after vaccination, the efficacy against HZ was 82.0% (95% CI: 63.0; 92.2) and 72.0% (95% CI: 33.4; 89.8) in subjects ≥ 50 years (Shingrix group: N=5,849) and subjects ≥ 70 years (Shingrix group: N=2,891), respectively. At 3 years post-dose 2, the median fold increase over baseline was 9.3 (Q1: 4.9; Q3: 19.5) in adults ≥ 50 years (Zoster-006) and 7.2 (Q1: 3.5; Q3: 14.5) in adults ≥ 70 years (pooled Zoster-006 and Zoster-022).
At 3 years post-dose 2, the median fold increase over baseline was 9.3 (Q1: 4.9; Q3: 19.5) in adults ≥ 50 years (Zoster-006) and 7.2 (Q1: 3.5; Q3: 14.5) in adults ≥ 70 years (pooled Zoster-006 and Zoster-022). At 3 years post-dose 2, in Zoster-006, the median fold increase over baseline was 7.9 (Q1: 2.7; Q3: 31.6) in adults ≥ 50 years and 7.3 (Q1: 1.7; Q3: 31.6) in adults ≥ 70 years.
At 3 years post-dose 2, in Zoster-006, the median fold increase over baseline was 7.9 (Q1: 2.7; Q3: 31.6) in adults ≥ 50 years and 7.3 (Q1: 1.7; Q3: 31.6) in adults ≥ 70 years.
 In Zoster-028, GMC 1-month post Dose 2 were 22,974.3 (19,080.0; 27,663.5) in the group that received the first dose of Shingrix at least 10 days prior to a chemotherapy cycle (PreChemo group) and 9,328.0 (4,492.5; 19,368.2) in the group that received the first dose of Shingrix simultaneously with chemotherapy cycle (OnChemo group). In Zoster-039, GMC 1-month post Dose 2 were 19,934.7 (14,674.1; 27,081.2) in the group that received the first dose of Shingrix after the full cancer therapy course and 5,777.4 (3,342.5; 9,985.9) in the group that received the first dose of Shingrix during a cancer therapy course. The clinical relevance in terms of impact on efficacy, on the short and long term, is unknown.
In Zoster-028, GMC 1-month post Dose 2 were 22,974.3 (19,080.0; 27,663.5) in the group that received the first dose of Shingrix at least 10 days prior to a chemotherapy cycle (PreChemo group) and 9,328.0 (4,492.5; 19,368.2) in the group that received the first dose of Shingrix simultaneously with chemotherapy cycle (OnChemo group). In Zoster-039, GMC 1-month post Dose 2 were 19,934.7 (14,674.1; 27,081.2) in the group that received the first dose of Shingrix after the full cancer therapy course and 5,777.4 (3,342.5; 9,985.9) in the group that received the first dose of Shingrix during a cancer therapy course. The clinical relevance in terms of impact on efficacy, on the short and long term, is unknown.