WHAT IS IN THIS LEAFLET
This leaflet answers some common questions about SINADOPA. It does not contain all the available information.
It does not take the place of talking to your doctor or pharmacist.
All medicines have risks and benefits. Your doctor has weighed the risks of you using SINADOPA against the benefits it is expected to have for you.
If you have any concerns about using this medicine, ask your doctor or pharmacist.
Keep this leaflet with the medicine. You may need to read it again.
WHAT SINADOPA IS USED FOR
SINADOPA is used to treat some of the symptoms of Parkinson's disease. This is a disease of the nervous system that mainly affects body movement. The three main symptoms are shaking (tremor), muscle stiffness and slow and unsteady movement. People with Parkinson's disease often walk with a shuffle as they have difficulty in initiating movement. If untreated, Parkinson's disease can cause difficulty in performing normal daily activities.
SINADOPA is most helpful in improving slow movement and muscle stiffness. It is also frequently helpful in treating shaking, difficulty in swallowing and drooling.
The symptoms of Parkinson's disease are caused by a lack of dopamine, a naturally occurring chemical produced by certain brain cells. Dopamine relays messages in the part of the brain that controls muscle movement.
When too little dopamine is produced slowness of movement results.
SINADOPA contains two active ingredients, levodopa and carbidopa. Levodopa is a chemical closely related to dopamine which allows the body to make its own dopamine. Carbidopa makes sure that enough levodopa gets to the brain where it is needed. In many patients, SINADOPA reduces some of the symptoms of Parkinson's disease.
Your doctor may have prescribed SINADOPA for another reason. Ask your doctor if you have any questions about why SINADOPA has been prescribed for you.
BEFORE YOU USE SINADOPA
When you must not use it
Do not take SINADOPA if:
- you have an allergy to SINADOPA or any of the ingredients listed at the end of this leaflet
- you have any unusual skin lumps or moles which have not been examined by your doctor, or if you have ever had skin cancer or melanoma
- you have a type of glaucoma called narrow-angle glaucoma
- are being treated for depression with certain medicines called monoamine oxidase (MAO) inhibitors
Ask your doctor or pharmacist if you are not sure whether you are taking one of these medicines. - you are breast-feeding or plan to breast-feed
It has been shown that one of the active ingredients of SINADOPA passes into breast milk. Therefore, because of the potential harm to the baby, SINADOPA should not be used during breast-feeding. - the packaging is torn or shows signs of tampering
- the expiry date on the pack has passed.
If you take this medicine after the expiry date has passed, it may not work.
If you are not sure whether you should start taking SINADOPA, talk to your doctor.
Do not give SINADOPA to a child or teenager below the age of 18 years, unless advised by the child’s doctor. The safety and effectiveness of SINADOPA in children and teenagers under 18 years of age has not been established.
Before you start to use it
Tell your doctor if:
- you are pregnant or intend to become pregnant
Your doctor will discuss the possible risks and benefits of using SINADOPA during pregnancy.
- you have or have had any medical conditions, especially the following:
- depression or mental disturbances
- heart disease, including irregular heartbeat, also known as arrhythmia
- lung disease, including asthma
- kidney, liver or hormonal problems
- convulsions or fits
- glaucoma
- peptic ulcer disease
- you or your family member/caregiver notices you are developing urges to gamble, excessive eating or spending, medicine use or repetitive purposeless activities with other medicines for Parkinson's Disease, and/or other intense urges that could harm yourself or others. These behaviours are called impulse control disorders. Your doctor may need to review your treatments.
- you have previously been or are currently being treated with levodopa.
- you have any allergies to any other medicines or any other substances, such as foods, preservatives or dyes.
If you have not told your doctor about any of the above, tell them before you take any SINADOPA.
Taking other medicines
Tell your doctor if you are taking any other medicines, including any that you buy without a prescription from your pharmacy, supermarket or health food shop. Some medicines and SINADOPA may interfere with each other. These include:
- some medicines used to treat high blood pressure
- some medicines used to treat depression
- some medicines used to treat psychiatric problems
- some medicines used to treat diseases related to involuntary movements
- phenytoin, a medicine used to treat convulsions
- isoniazid, a medicine used to treat tuberculosis
- selegiline, another medicine used to treat Parkinson's disease
- iron supplements and multivitamins containing iron
These medicines may be affected by SINADOPA, or may affect how well the tablets work. You may need different amounts of your medicine, or you may need to take different medicines.
Your doctor or pharmacist has more information on medicines to be careful with or avoid while taking SINADOPA.
HOW TO USE SINADOPA
How much to use
Take SINADOPA only when prescribed by your doctor.
Your doctor will tell you how many tablets to take each day. The dose varies considerably from patient to patient.
The usual starting dose is one 100/25 mg tablet taken three times a day. Your doctor will then adjust this dose depending on the severity of your condition, your response to treatment and whether you are taking other medicines.
Follow all directions given to you by your doctor carefully. They may differ from the information contained in this leaflet.
If you do not understand the instructions on the bottle, ask your doctor or pharmacist for help.
How to use it
Swallow SINADOPA, whole or as half tablets (only if told by your doctor), with a glass of water.
How long to use it for
SINADOPA helps control some of your symptoms of Parkinson's disease, but does not cure it. Therefore SINADOPA must be taken every day. Continue taking SINADOPA for as long as your doctor prescribes.
Do not stop taking SINADOPA, or lower the dosage, without checking with your doctor. Your doctor may want you to gradually reduce the amount of SINADOPA you are using before stopping completely. This may help reduce the possibility of withdrawal symptoms such as muscle stiffness, fever and mental changes.
If you forget to use it
If it is almost time for your next dose, skip the dose you missed and take your next dose when you are meant to. Otherwise, take it as soon as you remember, and then go back to taking your tablet(s) as you would normally.
If you are not sure whether to skip the dose, talk to your doctor or pharmacist.
Do not take a double dose to make up for the dose that you missed.
If you have trouble remembering to take your tablets, ask your pharmacist for some hints.
If you use too much (overdose)
Immediately telephone your doctor or Poisons Information Centre (telephone 13 11 26), or go to accident and emergency at your nearest hospital, if you think that you or anyone else may have taken too much SINADOPA. Do this even if there are no signs of discomfort or poisoning.
WHILE YOU ARE USING SINADOPA
Things you must do
If you feel light-headed, dizzy or faint get up slowly when getting out of bed or standing up. You may feel light-headed or dizzy while taking SINADOPA. This is because your blood pressure is falling suddenly. Standing up slowly, especially when you get up from bed or chairs, will help your body get used to the change in position and blood pressure. If this problem continues or gets worse, tell your doctor.
If you are about to be started on any new medicine, tell your doctor and pharmacist that you are taking SINADOPA.
If you experience times where SINADOPA does not appear to be working as well as it did previously, tell your doctor.
After taking this medicine for long periods of time, such as a year or more, some people suddenly lose the ability to move. This loss of movement may last from a few minutes to several hours. The person is then able to move as before. This condition may unexpectedly occur again and again. This problem is called the "on-off" effect. Your doctor may prescribe you a stronger dose of SINADOPA or may ask you to take it more frequently. Your doctor may need to prescribe you a different medicine.
Have blood tests taken when your doctor says to make sure SINADOPA is not causing any problems with your blood, liver, kidneys or heart.
If you plan to have surgery that needs a general anaesthetic, tell your doctor or dentist that you are taking SINADOPA.
If you become pregnant while taking SINADOPA, tell your doctor.
Things you must not do
Do not give SINADOPA to anyone else, even if they have the same condition as you.
Things to be careful of
Be careful when driving or operating machinery until you know how SINADOPA affects you. SINADOPA may cause dizziness or light-headedness in some people. Make sure you know how you react to SINADOPA before you drive a car, operate machinery, or do anything else that could be dangerous if you are dizzy or light-headed. If you drink alcohol, dizziness or lightheadedness may be worse.
In addition, in very rare cases, SINADOPA may cause excessive sleepiness and sudden onset of sleep. If you experience these effects, do not drive or operate machinery until these effects have resolved.
Be careful not to eat a diet high in protein. The amount of levodopa absorbed by the body may be impaired if you eat a diet high in protein. Ask your doctor, pharmacist or dietician to check your diet.
If you are diabetic, check with your doctor or pharmacist before using urine sugar tests. SINADOPA may cause false test results with some urine sugar tests.
AFTER USING SINADOPA
SIDE EFFECTS
Tell your doctor or pharmacist as soon as possible if you do not feel well while you are taking SINADOPA.
SINADOPA helps most people with Parkinson's disease, but it may have unwanted side effects in a few people. All medicines can have side effects. Sometimes they are serious, most of the time they are not. You may need medical treatment if you get some of the side effects.
Ask your doctor or pharmacist to answer any questions you may have.
Tell your doctor if you notice any of the following and they worry you:
- abnormal uncontrolled movements including muscle twitching or spasms, which may or may not resemble your Parkinson's symptoms
- dizziness, light-headedness when standing quickly
- feeling sick (nausea), vomiting, loss of appetite
- discolouration of urine, sweat and/or saliva
- dream abnormalities
- sleepiness or sudden onset of sleep
- slow movements
- twitching or spasm of the eyelids
- hair loss
- diarrhoea
- tingling, numbness, pain or burning sensation in the feet and/or hands
Tell your doctor if you experience any of these behaviours:
You may experience an inability to resist the impulse to perform an action that could be harmful, which may include:
- strong impulses to gamble
- increased sexual drive
- uncontrollable excessive shopping
- spending, binge/compulsive eating
- taking medicines and repetitive purposeless activities
- and/or other urges
These are possible side effects of SINADOPA. For the most part these have been mild.
Tell your doctor immediately if you notice any of the following:
- blood in the urine
- difficult or painful urination
- changes in mood such as depression
- forgetfulness
- signs of anaemia, such as tiredness, being short of breath, and looking pale
- signs of frequent or worrying infections such as fever, severe chills, sore throat or mouth ulcers
- bruising or bleeding more easily than normal, nose bleeds
- fainting
- skin rash, itchiness
- pinkish, itchy swellings on the skin, also called hives or nettle rash
- numbness or tingling in the hands or feet
- signs of melanoma, such as new skin spots or changes to the size, shape, colour or edges of an existing skin spot, freckle or mole.
These are serious side effects. You may need urgent medical attention.
Serious side effects are generally rare.
Tell your doctor immediately or go to accident and emergency at your nearest hospital if you notice any of the following:
- swelling of the face, lips, mouth, throat or tongue which may cause difficulty in swallowing or breathing
- bleeding from the back passage, black sticky bowel motions (stools) or bloody diarrhoea
- vomiting blood or material that looks like coffee grounds
- chest pain
- fast or irregular heartbeats, also called palpitations
- muscle stiffness accompanied by fever
- mental changes such as feeling very fearful or paranoid, hallucinations
- shortness of breath, difficulty breathing
These are all serious side effects that need urgent medical attention. Serious side effects are generally rare.
Other side effects not listed above may also occur in some patients. Tell your doctor if you notice any other effects.
Do not be alarmed by this list of possible side effects. You may not experience any of them.
AFTER USING SINADOPA
Storage
Keep your tablets in the bottle until it is time to take them. If you take the tablets out of the bottle they may not keep well.
Keep SINADOPA in a cool dry place where the temperature stays below 25°C. Do not store it or any other medicine in the bathroom or near a sink. Do not leave it in the car or on window sills. Heat and dampness can damage some medicines.
Keep it where children cannot reach it. A locked cupboard at least one-and-a- half metres above the ground is a good place to store medicines.
Disposal
If your doctor tells you to stop taking SINADOPA or the tablets have passed their expiry date, ask your pharmacist what to do with any that are left over.
PRODUCT DESCRIPTION
What it looks like
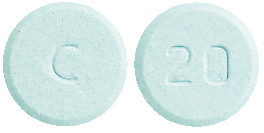
SINADOPA 250/25: Round, light blue uncoated tablets with ‘C’ on one side and ‘20’ on the other side.
SINADOPA 100/25: Round, light yellow uncoated tablets with ‘C’ on one side and ‘19’ on the other side.
The tablets are supplied in Alu/Alu blister packs of 100 tablets.
Ingredients
SINADOPA 250/25 contains 250 mg of levodopa and 25 mg of carbidopa (as monohydrate).
SINADOPA 100/25 contains 100 mg of levodopa and 25 mg of carbidopa (as monohydrate).
The tablets also contain the following inactive ingredients:
- crospovidone
- microcrystalline cellulose
- magnesium stearate
- pregelatinised maize starch
- indigo carmine aluminium lake (250/25)
- quinolone yellow aluminium lake (100/25)
SUPPLIER
Arrotex Pharmaceuticals Pty Ltd
15 – 17 Chapel Street
Cremorne VIC 3121
Australia
www.arrotex.com.au
250/25: AUST R 275983 (blister)
100/25: AUST R 275974 (blister)
This leaflet was prepared: November 2022
Published by MIMS January 2023