What is in this leaflet
This leaflet answers some common questions about SPRYCEL®. It does not take the place of talking to your doctor or pharmacist.
All medicines have risks and benefits. Your doctor has weighed the risks of you taking SPRYCEL against the benefits that are expected. This leaflet does not contain everything about SPRYCEL. Your doctor has been provided with full information and can answer any questions you may have. Follow your doctor's advice even if it differs from what is in this leaflet.
Please read this leaflet carefully and keep it in a safe place so you may refer to it later.
What SPRYCEL is used for
SPRYCEL is used to treat adults and children ages 12 months and older with chronic myeloid leukaemia (CML). It is also used to treat adults and children ages 12 months and older who have a particular form of acute lymphoblastic leukaemia (ALL) called Philadelphia chromosome positive or Ph+ ALL. Leukaemia is a cancer of immature white blood cells, which grow in the bone marrow. Under normal circumstances, as these white blood cells mature, they enter the blood stream where they fight infection and maintain the body's immune system. In leukaemia, these immature white blood cells multiply in an uncontrolled manner, occupying the bone marrow space and spilling out into the bloodstream. As a consequence, the production of normal red blood cells (oxygen carrying cells), white blood cells (cells which fight infection), and platelets (cells which help blood to clot) is compromised. Therefore patients with leukaemia are at risk of developing serious anaemia, infections and bleeding.
It is intended that SPRYCEL be used in adults with:
- Newly diagnosed Ph+ CML in the chronic phase who have not received any prior therapies, OR
- Ph+ CML across all phases who are no longer benefiting from other therapies for these diseases (resistance) or in patients who experience severe side effects to other therapies (intolerance), OR
- Newly diagnosed Ph+ ALL who have not received any prior therapies, OR
- Ph+ ALL who are no longer benefiting from other therapies for these diseases (resistance) or in patients who experience severe side effects to other therapies (intolerance).
It is intended that SPRYCEL be used in children with:
- Newly diagnosed Ph+ CML in the chronic phase who have not received any prior therapies, OR
- Ph+ CML in the chronic phase who are no longer benefiting from other therapies for these diseases (resistance) or in patients who experience severe side effects to other therapies (intolerance), OR.
- Newly diagnosed Ph+ ALL who have not received any prior therapies.
There is no experience with SPRYCEL treatment in children under 1 year of age,
Ask your doctor if you have any questions about why SPRYCEL was prescribed for you or your child.
This medicine is available only with a doctor's prescription.
How SPRYCEL works
The active ingredient in SPRYCEL is dasatinib. Dasatinib acts by inhibiting the activity of proteins within the leukaemia cells of patients with CML or Ph+ ALL. These proteins are responsible for the uncontrolled growth of the leukaemia cells. By inhibiting these proteins, SPRYCEL kills the leukaemia cells in the bone marrow and allows normal red cell, white cell and platelet production to resume.
Before you take SPRYCEL
When you must not take SPRYCEL:
- You or your child must not take SPRYCEL if you have a history of severe allergic reactions to SPRYCEL or to any of the ingredients listed at the end of this leaflet.
- Do not take SPRYCEL if you are a pregnant woman or intend to become pregnant.
SPRYCEL can cause harm to the unborn baby if it is given to a pregnant woman. - Do not take SPRYCEL after the expiry date printed on the pack.
If you or your child takes this medicine after the expiry date has passed, it may not work as well. - Do not take SPRYCEL if the packaging is torn or shows signs of tampering.
Before you start to take SPRYCEL
Tell your doctor if you or your child have allergies to:
- any other medicines
- any other substances, such as foods, preservatives or dyes.
Tell your doctor if you are breast-feeding or plan to breast-feed. Women who are taking SPRYCEL should not breast-feed.
Tell your doctor if you are a sexually active man. Men who take SPRYCEL are advised to use a condom to avoid pregnancy in their partner.
Tell your doctor if you or your child have or have had any medical conditions, especially the following:
- problems with your immune system
- liver problem
- heart problem
- lactose intolerant
- hepatitis B infection
If you have not told your doctor about any of the above, tell them before you or your child take SPRYCEL.
Taking other medicines
It is especially important to tell your doctor if you or your child are taking any other medicines, including any that you buy without a prescription from your pharmacy, supermarket or health food shop. These medicines may be affected by SPRYCEL or they may affect how well SPRYCEL works.
Tell your doctor if you or your child are taking:
- Nizoral® (ketoconazole), Sporanox® (itraconazole), Norvir® (ritonavir), Reyataz® (atazanavir), E-Mycin® (erythromycin), Klacid® (clarithromycin) and grapefruit juice may all increase the level of SPRYCEL in your bloodstream.
- Maxidex® (dexamethasone), Dilantin® (phenytoin), Tegretol® (carbamazepine), Rifadin® (rifampicin), and Phenobarbitone tablets may all decrease the levels of SPRYCEL in your bloodstream.
- SPRYCEL may alter the blood levels of Sandimmun® (cyclosporin).
- Blood thinning medicines such as Coumadin® (warfarin sodium), aspirin or non steroidal anti-inflammatory drugs (NSAIDs) taken with SPRYCEL may increase the risk of unwanted bleeding.
- Oral diabetes medicines from the glitazone family of drugs may interact with SPRYCEL.
You or your child should avoid taking Tagamet® (cimetidine), Pepcidine® (famotidine), Zantac® (ranitidine) or Probitor® (omeprazole) while taking SPRYCEL as they all reduce stomach acid, which is necessary for the absorption of SPRYCEL.
Medicines that neutralise stomach acid, such as Mylanta® (aluminium hydroxide/magnesium hydroxide), Andrews Tums Antacid® (calcium carbonate) or Mylanta® (calcium carbonate and magnesium hydroxide) may be taken up to 2 hours before or 2 hours after SPRYCEL.
Some medicines may be affected by SPRYCEL, or may affect how well it works. You or your child may need different amounts of your medicine, or you may need to take different medicines. Your doctor will advise you. Your doctor or pharmacist has more information on medicines to be careful with or avoid while taking SPRYCEL.
Know the medicines you take.
Keep a list of you or your child's medicines to show your doctor or pharmacist.
How to take SPRYCEL
How much to take:
You or your child should take your tablets consistently either in the morning or in the evening.
The usual starting dose for adults is 100 mg once daily (either as one 100 mg tablet or two 50 mg tablets). The entire dose of 100 mg is to be taken at one time either in the morning or the evening.
Some adult patients may require an alternative starting dose (140 mg daily taken as two 70 mg tablets). The entire dose of 140 mg is to be taken at one time either in the morning or the evening.
The starting dose for children is determined by body weight. The entire dose prescribed by your doctor should be taken at one time either in the morning of the evening.
Your doctor will decide the dose that is most appropriate for you. Please follow your doctor's instructions about how and when to take SPRYCEL.
How to take it:
Swallow your SPRYCEL tablet(s) whole with a glass of water. Do not break, cut, chew or crush the tablet.
Do not take SPRYCEL with grapefruit or grapefruit juice.
When to take it:
Try to take your dose of SPRYCEL at the same time each day, with or without a meal, as instructed by your doctor.
How long to take it:
You or your child should not stop taking SPRYCEL, or reduce the dose without first talking to your doctor. Depending on you or your child's response and any side effects that may be experienced, your doctor may adjust the dose of SPRYCEL, upward or downward, or may temporarily discontinue the medicine.
If you forget to take it:
If you miss a dose of SPRYCEL, take the next scheduled dose at its regular time. Don't make up for a missed dose by doubling up on tablets. Call your doctor or pharmacist if you are not sure what to do.
If you take too much (overdose):
Immediately telephone your doctor or Poisons Information Centre (telephone: 131126) or go to the Accident and Emergency Department at your nearest hospital, if you, your child or anyone else may have taken too much SPRYCEL. Do this even if there are no signs of discomfort or poisoning.
While you are taking SPRYCEL
Things you must do:
- You must tell your doctor immediately if you or your child become pregnant while taking SPRYCEL.
- Tell all of the doctors, dentists and pharmacists who are treating you or your child that you are taking SPRYCEL.
- If you or your child are about to be started on any new medicine, tell your doctor and pharmacist that you are taking SPRYCEL.
- Tell your doctor immediately if you or your child experience bleeding or easy bruising, no matter how mild.
- Have any tests recommended by your doctor done as soon as possible.
Your doctor may order routine laboratory tests to evaluate your blood counts to see how well SPRYCEL is working. - Notify your doctor immediately if you or your child develop a fever while taking SPRYCEL.
- Tell your doctor immediately if you or your child experience shortness of breath and fatigue while taking SPRYCEL
Things you must not do:
- Do not give SPRYCEL to anyone else, even if they have the same condition as you or your child.
- Do not use SPRYCEL to treat any other complaints unless your doctor tells you to.
- Do not stop taking SPRYCEL, or lower the dosage, without checking with your doctor.
- Do not stop taking SPRYCEL tablets because you or your child are feeling better unless advised to do so by your doctor.
Things to be careful of:
- Be careful driving or operating machinery until you know how SPRYCEL affects you.
SPRYCEL has been known to cause dizziness or light-headedness in some people. Make sure that you know how you or your child react to SPRYCEL before you drive a car, operate machinery, or do anything else that could be dangerous if you are dizzy.
Side effects
Tell your doctor or pharmacist as soon as possible if you or your child do not feel well while you are taking SPRYCEL.
Like all medicines, it is possible that SPRYCEL may have unwanted side effects in some people. There are many side effects caused by medicines used to treat leukaemia. During treatment with SPRYCEL you or your child will require close medical supervision.
The more common side effects of SPRYCEL are:
- headache; dizziness; numbness or tingling in arms, legs, fingers or toes; altered taste
- diarrhoea, nausea, vomiting
- shortness of breath, cough, accumulation of fluid in or around the lungs, pneumonia, chest pain
- infection, fever
- abdominal pain, bloating, flatulence, constipation, indigestion
- high blood pressure
- mouth ulcers
- skin rash, dermatitis (including eczema), dry skin, itching or peeling skin, flushing
- hair loss
- visual disturbances: blurry or diminished vision, dry eye
- fatigue
- swelling of the liver, abdomen, hands, lower legs, feet or face
- back pain, bone pain, joint pain, pain in fingers or toes, muscle aches or spasm, muscle weakness
- weight decrease, appetite disturbance
- gout
- conjunctivitis
- tinnitus (ringing in the ears)
- accumulation of fluid, foamy urine, weight gain (also known as nephrotic syndrome)
Tell your doctor immediately if you notice any of the following:
- swelling, weight gain, shortness of breath or chest pain
- swelling around your eyes
- skin infection - folliculitis, herpes
- shortness of breath and fatigue
- dark coloured urine or yellowing of your skin and eyes
Tell your doctor immediately, or go to accident and emergency at your nearest hospital if you notice any of the following:
- changes in the way your heart beats, for example - if you notice it beating faster
- bleeding - for example from: gastrointestinal tract; eye; nose; gums; serious bruising or excessive menstrual bleeding
- infections - urinary tract; eye infection; difficulty breathing; coughing
- fever
- painful rash with blisters
The use of SPRYCEL is also associated with a reduction in certain types of blood cells in the body:
- a reduction in red blood cells, which may result in fatigue or dizziness
- a reduction in white blood cells, which are used to fight infection
- a reduction in cells that help your blood to clot after injury
These are serious side effects. If you or your child have them you may need urgent medical attention.
Other side effects not listed above may also occur in some patients. Tell your doctor if you notice anything else that is making you or your child feel unwell.
Product Description:
What it looks like
SPRYCEL 20 mg tablet
White to off-white, biconvex, round tablet with "BMS" debossed on one side and "527" on the other
SPRYCEL 50 mg tablet
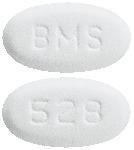
White to off-white, biconvex, oval tablet with "BMS" debossed on one side and "528" on the other
SPRYCEL 70 mg tablet
White to off-white, biconvex, round tablet with "BMS" debossed on one side and "524" on the other
SPRYCEL 100 mg tablet
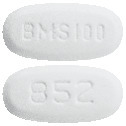
White to off-white, biconvex, oval film-coated tablet with "BMS 100" debossed on one side and "852" on the other
Ingredients
Active Ingredient:
dasatinib
Inactive Ingredients:
lactose monohydrate, microcrystalline cellulose, hyprolose, croscarmellose sodium, magnesium stearate and Opadry White YS-1-18177-A.
Storage
Keep SPRYCEL tablets in the pack until it is time to take them. If you or your child take the tablets out of the bottle they may not keep well.
Keep the pack in a cool dry place where the temperature stays below 30°C.
Do not store SPRYCEL or any other medicine in the bathroom or near a sink.
Do not leave it in the car on hot days. Heat and dampness can destroy some medicines.
Keep it where children cannot reach it. A locked cupboard at least one-and-a-half metres above the ground is a good place to store medicines.
Disposal
Return any unused medicine to your pharmacist.
Sponsored by
Bristol-Myers Squibb Australia Pty Ltd
4 Nexus Court, Mulgrave
Victoria 3170, Australia
Registration Numbers:
AUST R 125557 - 20 mg tablet - 60 tablets per bottle
AUST R 125559 - 50 mg tablet - 60 tablets per bottle
AUST R 125561 - 70 mg tablet - 60 tablets per bottle
AUST R 157352 - 100 mg tablet - 30 tablets per bottle
Where to get further information
Your doctor is the best person to answer any further questions you may have about SPRYCEL.
Anything your doctor tells you about SPRYCEL® should be followed even if it is different from what is in this leaflet.
Date of Preparation: June 2020
Published by MIMS August 2020




 Dose escalation is not recommended for paediatric patients with Ph+ ALL, as Sprycel is administered in combination with chemotherapy in these patients.
Dose escalation is not recommended for paediatric patients with Ph+ ALL, as Sprycel is administered in combination with chemotherapy in these patients.
 For paediatric patients with chronic phase CML, if grade ≥ 3 neutropenia or thrombocytopenia recurs during complete haematologic response (CHR), Sprycel should be interrupted, and may be subsequently resumed at a reduced dose. Temporary dose reductions for intermediate degrees of cytopenia and disease response should be implemented as needed.
For paediatric patients with chronic phase CML, if grade ≥ 3 neutropenia or thrombocytopenia recurs during complete haematologic response (CHR), Sprycel should be interrupted, and may be subsequently resumed at a reduced dose. Temporary dose reductions for intermediate degrees of cytopenia and disease response should be implemented as needed.
 Myelosuppression was commonly reported in all patient populations. In newly diagnosed chronic phase CML, myelosuppression was less frequently reported than in chronic phase CML patients with resistance or intolerance to prior imatinib therapy. The frequency of grade 3 or 4 neutropenia, thrombocytopenia, and anaemia was higher in patients with advanced CML or Ph+ ALL than in chronic phase CML.
Myelosuppression was commonly reported in all patient populations. In newly diagnosed chronic phase CML, myelosuppression was less frequently reported than in chronic phase CML patients with resistance or intolerance to prior imatinib therapy. The frequency of grade 3 or 4 neutropenia, thrombocytopenia, and anaemia was higher in patients with advanced CML or Ph+ ALL than in chronic phase CML.
 For time to cCCyR, a hazard ratio of 1.55 indicates that a patient treated with Sprycel is 55% more likely to achieve a cCCyR at any time compared to a patient treated with imatinib. Similarly, for time to MMR, a hazard ratio of 2.01 indicates a patient treated with Sprycel is more than two times more likely to achieve a MMR at any time compared to a patient treated with imatinib. For durability of cCCyR (time in response), a hazard ratio of 0.7 indicates a patient treated with Sprycel is 30% less likely to have disease progression after achieving a cCCyR (or never achieving a cCCyR) compared to a patient treated with imatinib.
For time to cCCyR, a hazard ratio of 1.55 indicates that a patient treated with Sprycel is 55% more likely to achieve a cCCyR at any time compared to a patient treated with imatinib. Similarly, for time to MMR, a hazard ratio of 2.01 indicates a patient treated with Sprycel is more than two times more likely to achieve a MMR at any time compared to a patient treated with imatinib. For durability of cCCyR (time in response), a hazard ratio of 0.7 indicates a patient treated with Sprycel is 30% less likely to have disease progression after achieving a cCCyR (or never achieving a cCCyR) compared to a patient treated with imatinib.
 The progression free survival rate by specific timepoint is displayed graphically in Figure 1. Rate of PFS was consistently higher in Sprycel treated patients who achieved BCR-ABL level ≤ 10 percent at 3 months than those who did not.
The progression free survival rate by specific timepoint is displayed graphically in Figure 1. Rate of PFS was consistently higher in Sprycel treated patients who achieved BCR-ABL level ≤ 10 percent at 3 months than those who did not. The overall survival rate by specific timepoint is displayed graphically in Figure 2. Rate of OS was consistently higher in Sprycel treated patients who achieved BCR-ABL level ≤ 10 percent at 3 months than those who did not.
The overall survival rate by specific timepoint is displayed graphically in Figure 2. Rate of OS was consistently higher in Sprycel treated patients who achieved BCR-ABL level ≤ 10 percent at 3 months than those who did not. The time to MMR is displayed graphically in Figure 3. The time to MMR was consistently shorter in Sprycel treated subjects compared with imatinib treated subjects.
The time to MMR is displayed graphically in Figure 3. The time to MMR was consistently shorter in Sprycel treated subjects compared with imatinib treated subjects. MMR rates by specific timepoint are displayed graphically in Figure 4. Rates of MMR were consistently higher in Sprycel treated subjects compared with imatinib treated subjects.
MMR rates by specific timepoint are displayed graphically in Figure 4. Rates of MMR were consistently higher in Sprycel treated subjects compared with imatinib treated subjects. MR4.5 rates over time are displayed graphically in Figure 5. Rate of MR4.5 over time was consistently higher in Sprycel treated subjects compared with imatinib treated subjects.
MR4.5 rates over time are displayed graphically in Figure 5. Rate of MR4.5 over time was consistently higher in Sprycel treated subjects compared with imatinib treated subjects. Disease progression was defined as increasing white blood cells despite appropriate therapeutic management, loss of CHR (complete haematologic response), partial CyR or CCyR, progression to accelerated phase or blast phase, or death. The estimated 60 month PFS rate was 88.9% (CI: 84.0%-92.4%) and 89.2% (CI: 84.3%-92.7%) for the Sprycel and imatinib treatment groups, respectively. Transformation to accelerated or blast phase occurred less frequently with Sprycel (n = 8; 3.1%) than with imatinib treated patients (n = 15; 5.8%). The estimated 60 month survival rates for Sprycel and imatinib treated patients were 90.9% (CI: 86.6%-93.8%) and 89.6% (CI: 85.2%-92.8%), respectively.
Disease progression was defined as increasing white blood cells despite appropriate therapeutic management, loss of CHR (complete haematologic response), partial CyR or CCyR, progression to accelerated phase or blast phase, or death. The estimated 60 month PFS rate was 88.9% (CI: 84.0%-92.4%) and 89.2% (CI: 84.3%-92.7%) for the Sprycel and imatinib treatment groups, respectively. Transformation to accelerated or blast phase occurred less frequently with Sprycel (n = 8; 3.1%) than with imatinib treated patients (n = 15; 5.8%). The estimated 60 month survival rates for Sprycel and imatinib treated patients were 90.9% (CI: 86.6%-93.8%) and 89.6% (CI: 85.2%-92.8%), respectively. Efficacy was also assessed in patients who were intolerant to imatinib. In this population of patients who received 100 mg once daily, MCyR was achieved in 77% and CCyR in 67% with a minimum of 2 years follow-up.
Efficacy was also assessed in patients who were intolerant to imatinib. In this population of patients who received 100 mg once daily, MCyR was achieved in 77% and CCyR in 67% with a minimum of 2 years follow-up. In the phase III, randomized, open label study in patients with advanced phase CML and Ph+ ALL, whose disease was resistant to or who were intolerant to imatinib, the primary endpoint was MaHR. A total of 611 patients were randomised to either the Sprycel 140 mg once daily or 70 mg twice daily group. Median duration of treatment was approximately 6 months (range < 1-31 months).
In the phase III, randomized, open label study in patients with advanced phase CML and Ph+ ALL, whose disease was resistant to or who were intolerant to imatinib, the primary endpoint was MaHR. A total of 611 patients were randomised to either the Sprycel 140 mg once daily or 70 mg twice daily group. Median duration of treatment was approximately 6 months (range < 1-31 months). In patients with accelerated phase CML treated with the 140 mg once daily regimen, the median duration of MaHR and the median overall survival in patients with accelerated phase CML was not reached for either group; the median PFS was 25 months and 26 months for the 140 mg once daily group and the 70 mg twice daily group, respectively; and the median overall survival was not reached for the 140 mg once daily group and 31 months for the 70 mg twice daily group.
In patients with accelerated phase CML treated with the 140 mg once daily regimen, the median duration of MaHR and the median overall survival in patients with accelerated phase CML was not reached for either group; the median PFS was 25 months and 26 months for the 140 mg once daily group and the 70 mg twice daily group, respectively; and the median overall survival was not reached for the 140 mg once daily group and 31 months for the 70 mg twice daily group. With a median follow-up of 4.5 years in newly diagnosed patients, the median durations of CCyR, MCyR, MMR could not be estimated as more than half of the responding patients had not progressed at the time of data cut-off. Range of duration of response was (2.5+ to 66.5+ months for CCyR), (1.4 to 66.5+ months for MCyR), and (5.4+ to 72.5+ months for subjects who achieved MMR by month 24 and 0.03+ to 72.5+ months for subjects who achieved MMR at any time), where '+' indicates a censored observation.
With a median follow-up of 4.5 years in newly diagnosed patients, the median durations of CCyR, MCyR, MMR could not be estimated as more than half of the responding patients had not progressed at the time of data cut-off. Range of duration of response was (2.5+ to 66.5+ months for CCyR), (1.4 to 66.5+ months for MCyR), and (5.4+ to 72.5+ months for subjects who achieved MMR by month 24 and 0.03+ to 72.5+ months for subjects who achieved MMR at any time), where '+' indicates a censored observation. The chemical name for dasatinib is N-(2-chloro-6-methylphenyl)- 2-[[6-[4-(2-hydroxyethyl)-1-piperazinyl]-2-methyl-4-pyrimidinyl] amino]-5-thiazolecarboxamide, monohydrate.
The chemical name for dasatinib is N-(2-chloro-6-methylphenyl)- 2-[[6-[4-(2-hydroxyethyl)-1-piperazinyl]-2-methyl-4-pyrimidinyl] amino]-5-thiazolecarboxamide, monohydrate.