What is in this leaflet
Please read this leaflet carefully before you start taking Sumatriptan generichealth.
This leaflet answers some common questions about Sumatriptan generichealth. It does not contain all of the available information.
It does not take the place of talking to your doctor or pharmacist.
All medicines have risks and benefits. Your doctor has weighed the expected benefits of you taking Sumatriptan generichealth against the risks this medicine could have for you.
If you have any concerns about taking this medicine, ask your doctor or pharmacist.
Keep this leaflet with the medicine. You may need to read it again.
What Sumatriptan generichealth is used for
Sumatriptan generichealth contains the active ingredient sumatriptan succinate. This medicine belongs to a group of drugs called serotonin agonists.
Sumatriptan generichealth are used to relieve a migraine attack. They should not be used to prevent migraine attacks from occurring. Sumatriptan generichealth may be used for migraine headaches with or without what is known as 'aura'.
It is thought that migraine headache is due to widening of certain blood vessels in the head. Sumatriptan generichealth work by making those vessels normal again and ease the symptoms of migraine.
Your Sumatriptan generichealth tablets do not work in other types of headache which are not a migraine.
Sumatriptan generichealth are not addictive.
Before you take Sumatriptan generichealth
When you must not take it
Do not take Sumatriptan generichealth if:
- you have ever had an allergic reaction to sumatriptan succinate (see "Side effects") or any of the ingredients listed toward the end of this leaflet (see "Ingredients").
- you have or have had:
- heart disease or heart attack;
- shortness of breath, pain or tightness in the chest, jaw or upper arm;
- peripheral vascular disease (pain in the back of the legs) or are prone to cold, tingling or numb hands and feet;
- Prinzmetal's angina (an uncommon form of angina where pain is experienced at rest rather than during activity);
- angina;
- high blood pressure;
- stroke;
- severe liver disease. - you have taken any of these medicines in the last 24 hours:
- Ergotamine (eg. Cafergot);
- Dihydroergotamine (eg. Dihydergot);
- Methysergide (eg. Deseril);
- Naratriptan (eg. Naramig);
- Zolmitriptan (eg. Zomig). - you have taken any of these medicines in the last two weeks:
- Monoamine oxidase inhibitors (MAOIs), a type of medicine used for depression.
- SSRIs (Selective Serotonin Reuptake Inhibitors) or SNRIs (Serotonin-noradrenaline reuptake inhibitors) used to treat depression. - the expiry date (EXP) printed on the pack has passed.
- the packaging is torn or shows signs of tampering.
Before you start to take it
You must tell your doctor if:
- you are allergic to foods, dyes, preservatives or any other medicines, including any that contain sulphur (eg. sulphonamide antibiotics).
- you are allergic to lactose.
- you are taking or have taken any other medicines in the last two weeks, including medicines you buy without a prescription, particularly herbal preparations containing St John's Wort and medicines prescribed for depression.
- you are breastfeeding, pregnant or trying to become pregnant.
- you have or have had medical conditions like:
- liver or kidney problems;
- heart problems. Risk factors including high blood pressure, even if it is under control, high blood cholesterol levels, a family history of heart problems, obesity, diabetes, you are male and over 40 years of age, you are female and have undergone menopause or you smoke;
- epilepsy, seizures, or fits or been told that you are prone to this problem;
- stroke.
How to take Sumatriptan generichealth
Take your medicine as your doctor has told you. The label on the pack will tell you how many tablets to take and how often you should take them. If you do not understand what you should do, ask your doctor or pharmacist.
How much to take
The recommended starting dose for adults aged 18 to 65 is 50 mg, however you may need to have your dose of Sumatriptan generichealth increased to 100 mg. Your doctor will tell you which dose is right for you.
If the first Sumatriptan generichealth tablet helps your migraine, but the migraine comes back later, you may take another Sumatriptan generichealth tablet. Do not take more than 300 mg of Sumatriptan generichealth in any 24 hours. Six pink (50 mg strength) or three white (100 mg strength) tablets contain 300 mg.
Do not take more Sumatriptan generichealth, or any other form of sumatriptan, if the first dose has not provided any relief from your symptoms. You may take your usual headache relief medication provided it does not contain ergotamine or methysergide. If you are not sure what to do, ask your doctor or pharmacist.
If your migraine is not relieved by Sumatriptan, you may use Sumatriptan generichealth on another occasion to treat another migraine attack. Provided there are no side effects, you can use Sumatriptan generichealth to treat at least three separate migraine attacks before you and your doctor decide this medicine is ineffective for you.
How to take it
Your Sumatriptan generichealth tablet should be swallowed with a drink of water. Do not crush or chew the tablet as it has a bitter taste.
When to take it
It is best to take your Sumatriptan generichealth tablet:
- when the migraine headache begins; or
- when other symptoms of the migraine begin, such as nausea (feeling sick), vomiting or your eyes becoming sensitive to light.
If you take your tablet later during the migraine attack it will still work for you. Do not take your Sumatriptan generichealth tablet before the above symptoms occur.
If you take too much (overdose)
Immediately telephone your doctor or Poisons Information Centre (telephone 13 11 26) for advice, if you think you or anyone else may have taken too many Sumatriptan generichealth tablets, even if there are no signs of discomfort or poisoning. You may need urgent medical attention. If you are not sure what to do, contact your doctor or pharmacist.
While you are taking Sumatriptan generichealth
Things you must do
Tell your doctor if, for any reason, you have not taken your medicine exactly as directed. Otherwise, your doctor may think that it is not working and change your treatment unnecessarily.
Things you must not do
Do not give this medicine to anyone else, even if their symptoms seem similar to yours.
Things to be careful of
As with many other medicines, Sumatriptan generichealth may cause drowsiness in some people.
Be careful driving or operating machinery until you know how Sumatriptan generichealth affect you.
If you use Sumatriptan generichealth too often, it may make your headache worse. If this happens, your doctor may tell you to stop taking Sumatriptan generichealth.
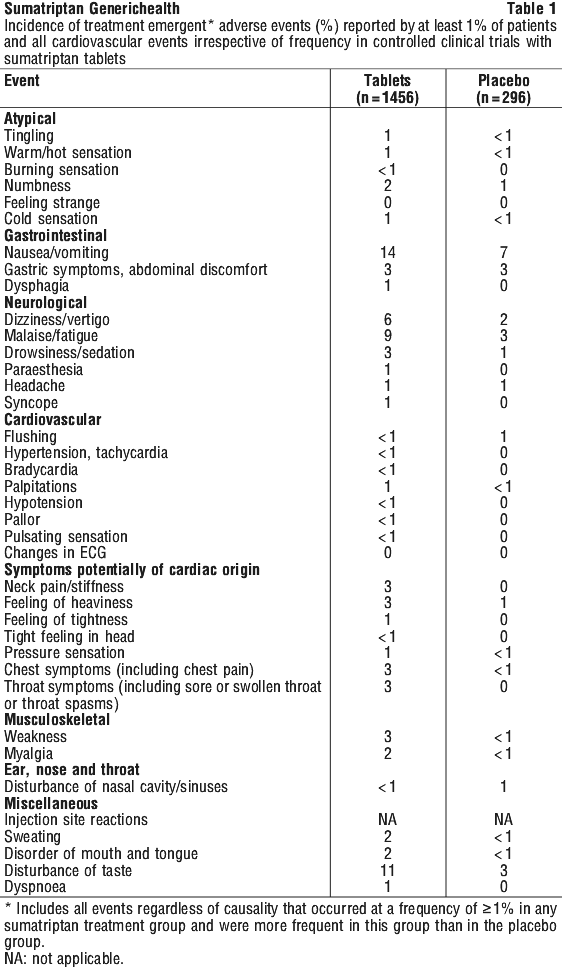
Side effects
Check with your doctor as soon as possible if you think you are experiencing any side effects or allergic reactions due to taking Sumatriptan generichealth, even if the problem is not listed below.
Like other medicines, Sumatriptan generichealth can cause some side effects. If they occur, they are most likely to be minor and temporary. However, some may be serious and need medical attention.
Tell your doctor if you experience any of the following after taking Sumatriptan generichealth:
- pain, tingling, heat or flushing in any part of the body;
- feeling of sleepiness, dizziness or tiredness;
- nausea (feeling sick) or vomiting;
- a change in blood pressure;
- feeling of faintness;
- problems with your eyesight;
- pain in the lower tummy and bloody diarrhoea (ischaemic colitis);
- shaking or tremors;
- uncontrolled movements;
- shortness of breath.
Tell your doctor immediately, or seek urgent medical attention, and do not take any more Sumatriptan generichealth if you:
- feel heaviness, pressure or tightness in any part of the body including the chest or throat;
- feel irregular heartbeats;
- have a fit or convulsion;
- have wheezing, swelling of the lips/mouth, difficulty in breathing, hay fever, lumpy rash ("hives") or fainting. These could be a symptom of an allergic reaction.
- have persistent purple discoloration and/or pain in the fingers, toes, ears, nose or jaw in response to cold.
These side effects are likely to be serious. Stop taking Sumatriptan generichealth and seek medical attention straight away.
This is not a complete list of all possible side effects. Others may occur in some people and there may be some side effects not yet known.
Do not be alarmed by this list of possible side effects. You may not experience any of them.
After taking Sumatriptan generichealth
Storage
Keep this medicine where children cannot reach it, such as in a locked cupboard. Keep Sumatriptan generichealth in the blister pack in a cool, dry place. Store below 30°C.
Do not leave them in a car, on a window sill or in a bathroom.
Disposal
Keep Sumatriptan generichealth in their pack until time to take. Return any unused or expired medicine to your pharmacist.
Product description
What it looks like
Sumatriptan generichealth 50 mg tablets are pink coloured, capsule shaped, biconvex film-coated tablets, debossed with “50” on one side and plain on the other side.
Sumatriptan generichealth 100 mg tablets are white coloured, capsule shaped, biconvex film-coated tablets, debossed with “100” on one side and plain on the other side.
Sumatriptan generichealth 50 mg & 100 mg are available in blister packs containing 4 tablets & 2 tablets respectively.
Ingredients
Active Ingredient
Each Sumatriptan generichealth tablet contains 50 mg or 100 mg sumatriptan (as succinate).
Other ingredients
- lactose;
- croscarmellose sodium;
- microcrystalline cellulose;
- crospovidone;
- colloidal anhydrous silica;
- magnesium stearate;
- OPADRY complete film coating system 03B84755 Pink (50 mg strength);
- OPADRY complete film coating system 03B28796 White (100 mg strength).
Australian Registration Numbers
Sumatriptan generichealth 50 mg: AUST R 160186
Sumatriptan generichealth 100 mg: AUST R 160194
Distributor
Generic Health Pty Ltd
Suite 2, Level 2
19-23 Prospect Street
Box Hill, VIC, 3128
Australia
Email: [email protected]
Telephone: +61 3 9809 7900
Website: www.generichealth.com.au
Where to go for further information
Pharmaceutical companies are not in a position to give people an individual diagnosis or medical advice. Your doctor or pharmacist is the best person to give you advice on the treatment of your condition.
This leaflet was prepared in December 2020.
Published by MIMS January 2021



 Chemical name: 3-[2-(dimethylamino)ethyl]-N-methyl-1H-indole-5-methane sulphonamide (sumatriptan);
Chemical name: 3-[2-(dimethylamino)ethyl]-N-methyl-1H-indole-5-methane sulphonamide (sumatriptan);