1 Name of Medicine
Lenacapavir sodium.
2 Qualitative and Quantitative Composition
Each single-dose vial contains lenacapavir sodium equivalent to 463.5 mg/1.5 mL of lenacapavir.
Each tablet contains lenacapavir sodium equivalent to 300 mg of lenacapavir.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Solution for injection.
Clear, yellow to brown solution with an apparent pH of 9.0 to 10.2 and a viscosity not more than 300 cP.
Film-coated tablet (tablet).
Beige, capsule-shaped, film-coated tablets, of dimensions 10 mm x 21 mm debossed with "GSI" on one side of the tablet and "62L" on the other side of the tablet.4.1 Therapeutic Indications
Sunlenca, in combination with other antiretrovirals, is indicated for the treatment of adults with multidrug resistant HIV-1 infection for whom it is otherwise not possible to construct a suppressive anti-viral regimen.
4.2 Dose and Method of Administration
Sunlenca should be prescribed by physicians experienced in the treatment of HIV. Prior to starting Sunlenca, the healthcare professional should carefully select patients who agree to the required injection schedule.
To help maintain viral suppression and reduce the risk of viral rebound and potential development of resistance associated with missed doses, the healthcare professional should also counsel patients about the importance of adherence to both the scheduled dosing visits and the optimised background regimen.
The recommended Sunlenca treatment regimen in adults consists of an initiation dosing period (oral tablets and subcutaneous injections) and once every 6-months maintenance dosing (subcutaneous injections). Sunlenca oral tablets may be taken with or without food.
If Sunlenca is discontinued, it is essential to adopt an alternative, fully suppressive antiretroviral regimen where possible, no later than 28 weeks after the final injection of Sunlenca (see Section 4.4 Special Warnings and Precautions for Use).
Initiation.
On treatment Day 1 and Day 2, the recommended dose of Sunlenca is 600 mg per day taken orally. On treatment Day 8, the recommended dose is 300 mg taken orally. Then, on treatment Day 15, the recommended dose is 927 mg administered by subcutaneous injection.
Oral tablets can be taken with or without food.
Maintenance.
The recommended dose is 927 mg of Sunlenca administered by subcutaneous injection once every 6 months (26 weeks) from the date of the last injection (+/- 2 weeks) (see Table 1).

Missed dose.
Missed tablets. If the Day 2 (600 mg) oral dose is missed by:
less than 6 days, the patient should take 600 mg as soon as possible, and 300 mg on Day 8;
6 days or more, the patient should take 600 mg as soon as possible, and 300 mg on Day 15.
If the Day 8 (300 mg) oral dose is missed by:
less than 6 days, the patient should take 300 mg as soon as possible;
6 days or more, the patient should take 300 mg on Day 15.
Regardless of when the Day 2 or Day 8 oral dose is being taken, subcutaneous injection should be administered on Day 15 as described in Table 1.
If the patient vomits within 3 hours of taking an oral dose of Sunlenca, another oral dose should be taken. If the patient vomits more than 3 hours after taking an oral dose of Sunlenca there is no need to take another oral dose of Sunlenca, and the scheduled dosing regimen should continue.
Missed injections. If the scheduled 6 month injection is delayed by less than 2 weeks (i.e. 26 to 28 weeks since the last injection) and if the patient is still able to receive subcutaneous injections, then administer the every 6 month maintenance dosage as soon as possible.
Oral bridging.
If the scheduled 6 month injection is anticipated to be delayed by more than 2 weeks, oral bridging with Sunlenca tablets may be initiated within 26 to 28 weeks since the last injection, on an interim basis. The recommended dosage is 300 mg taken orally once every 7 days for up to 26 weeks. Resume the maintenance injection dosage (see Table 1) within 7 days after the last oral dose.
Re-initiation.
If the scheduled 6 month injection is delayed by more than 2 weeks (i.e. > 28 weeks since the last injection) and Sunlenca tablets have not been taken for oral bridging, restart the initiation dosage regimen from Day 1, if clinically appropriate to continue treatment (see Table 1).
Elderly.
No dose adjustment of Sunlenca is required for elderly patients.
Renal impairment.
No dose adjustment of Sunlenca is required in patients with mild, moderate, or severe renal impairment (CrCl ≥ 15 mL/min). Sunlenca has not been studied in patients with end stage renal disease (ESRD).
Hepatic impairment.
No dose adjustment of Sunlenca is required in patients with mild or moderate hepatic impairment (Child-Pugh Class A or B). Sunlenca has not been studied in patients with severe hepatic impairment (Child-Pugh Class C).
Paediatric population.
The safety and efficacy of Sunlenca in children under the age of 18 years old has not been established. No data are available.
Method of administration.
Injection. For subcutaneous use.
Sunlenca injection is only for subcutaneous administration into the abdomen by a healthcare professional. Do not administer intradermally (see Section 4.4).
Use aseptic technique. Visually inspect the solution in the vials for particulate matter and discoloration prior to administration. Sunlenca injection is a yellow to brown solution.
Do not use Sunlenca injection if the solution contains particulate matter or discoloration. Once the solution is withdrawn from the vials, the subcutaneous injections should be administered as soon as possible (see Section 6.4 Special Precautions for Storage).
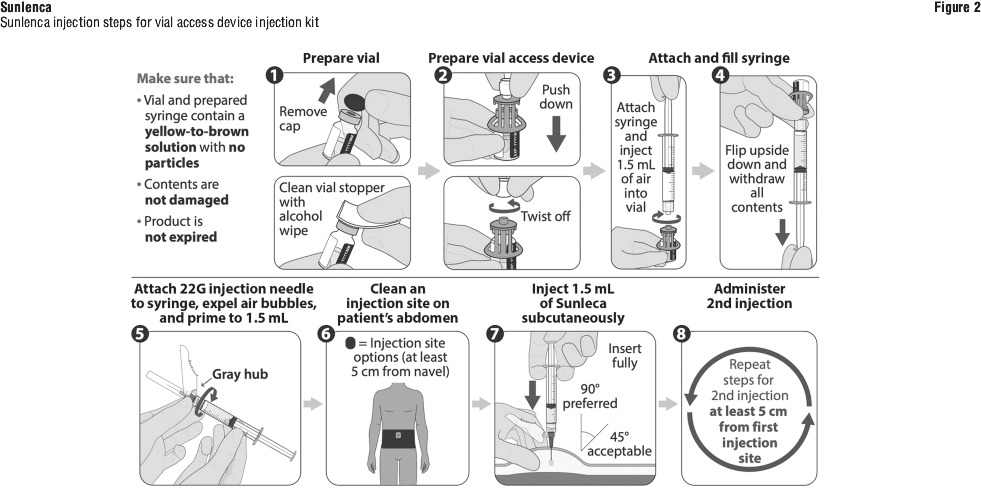
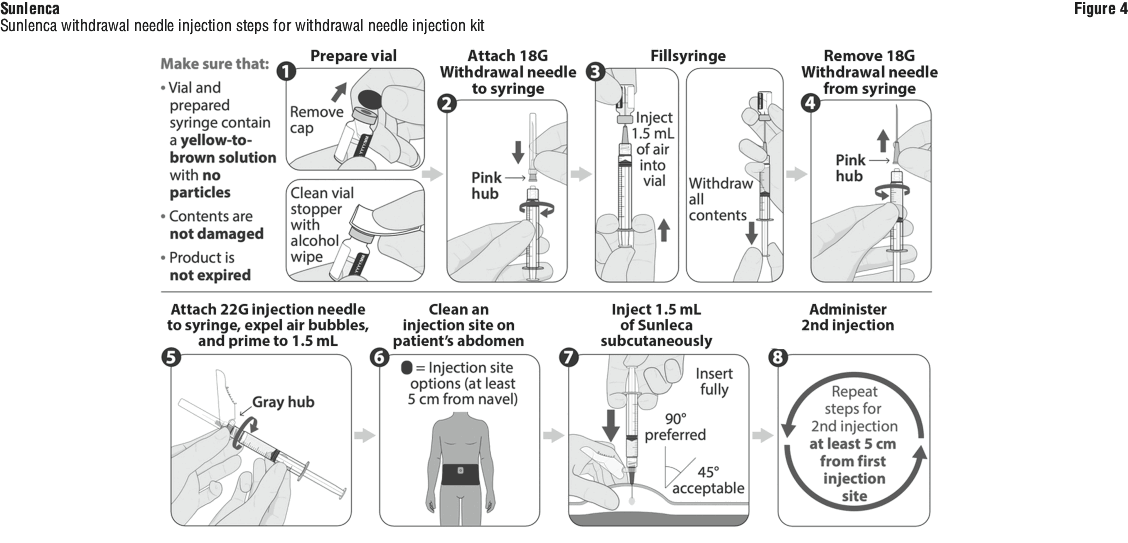
There are two available injection kits, which differ only in how Sunlenca injection is prepared (the components and associated method for withdrawal of the solution from the vials) (see Section 6.5). See Figures 1 and 2 for the relevant injection kit.
Sunlenca is for single use in one patient only. Discard any residue. Use of a vial access device is required. Two 1.5 mL injections are required for a complete dose.
Vial access device injection kit.
Figure 1 identifies the components for use in the administration steps for the vial access device injection kit, and the administration steps are provided in Figure 2. Use of a vial access device is required in this kit.

Withdrawal needle injection kit.
Figure 3 identifies the components for use in the administration steps for the withdrawal needle injection kit, and the administration steps are provided in Figure 4. The 18-gauge needle is for withdrawal only in this kit. See Figures 3 and 4.

 Tablets. For oral use.
Tablets. For oral use.
Sunlenca should be taken orally with or without food. The film-coated tablet should not be chewed, crushed, or split.4.3 Contraindications
Co-administration with strong inducers of CYP3A, P-gp, and UGT1A1, such as rifampicin, carbamazepine, phenytoin, or St. John's wort is contraindicated (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
4.4 Special Warnings and Precautions for Use
Risk of resistance following treatment discontinuation.
If Sunlenca is discontinued, to minimise the risk of developing viral resistance it is essential to adopt an alternative, fully suppressive antiretroviral regimen where possible, no later than 28 weeks after the final injection of Sunlenca.
If virologic failure is suspected, an alternative regimen should be adopted where possible.
Use of other medicinal products after discontinuation of lenacapavir.
If Sunlenca is discontinued, residual concentrations of lenacapavir may remain in the systemic circulation of patients for prolonged periods. These concentrations may affect the exposures of other medicinal products (i.e. sensitive CYP3A substrates) that are initiated within 9 months after the last subcutaneous dose of Sunlenca (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions). These concentrations are not expected to affect the exposures of other antiretroviral agents that are initiated after discontinuation of Sunlenca.
Immune reconstitution syndrome.
In patients with HIV treated with combination antiretroviral therapy, immune reconstitution syndrome has been reported. In patients with HIV with severe immune deficiency at the time of initiation of antiretroviral therapy, an inflammatory reaction to asymptomatic or residual opportunistic pathogens may arise and cause serious clinical conditions, or aggravation of symptoms. Typically, such reactions have been observed within the first few weeks or months of initiation of antiretroviral therapy. Relevant examples include cytomegalovirus retinitis, generalised and/or focal mycobacterial infections and Pneumocystis jirovecii pneumonia. Any inflammatory symptoms should be evaluated, and treatment instituted when necessary.
Autoimmune disorders (such as Graves' disease and autoimmune hepatitis) have also been reported to occur in the setting of immune reconstitution; however, the reported time to onset is more variable, and these events can occur many months after initiation of treatment.
Injection site reactions with improper administration.
Improper administration (intradermal injection) has been associated with serious injection site reactions, including necrosis and ulcer. Ensure Sunlenca is administered subcutaneously (see Section 4.2).
Opportunistic infections.
Patients should be advised that Sunlenca or any other antiretroviral therapy does not cure HIV infection and that they may still develop opportunistic infections and other complications of HIV infection. Therefore, patients should remain under close clinical observation by physicians experienced in the treatment of patients with HIV associated diseases.
Co-administration of other medicinal products.
Co-administration with medicinal products that are moderate inducers of CYP3A and P-gp (e.g. efavirenz) is not recommended (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
Co-administration with medicinal products that are strong inhibitors of CYP3A, P-gp, and UGT1A1 together (i.e. all 3 pathways), such as atazanavir/cobicistat is not recommended (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
Use in hepatic impairment.
See Section 4.2 Dose and Method of Administration; Section 5.2 Pharmacokinetic Properties, Pharmacokinetics in special populations.
Use in renal impairment.
See Section 4.2 Dose and Method of Administration; Section 5.2 Pharmacokinetic Properties, Pharmacokinetics in special populations.
Use in the elderly.
See Section 4.2 Dose and Method of Administration, Elderly.
Paediatric use.
The safety and efficacy of Sunlenca in children aged less than 18 years has not been established. No data are available.
Effects on laboratory tests.
See Section 4.8 Adverse Effects (Undesirable Effects), Laboratory abnormalities.4.5 Interactions with Other Medicines and Other Forms of Interactions
Effect of other medicinal products on the pharmacokinetics of Sunlenca.
Lenacapavir is a substrate of CYP3A, P-gp and UGT1A1. Drugs that are strong inducers of CYP3A, P-gp and UGT1A1 (e.g. rifampicin), or that are moderate inducers of CYP3A and P-gp (e.g. efavirenz), may significantly decrease plasma concentrations of lenacapavir, which may result in loss of therapeutic effect of Sunlenca and development of resistance.
Strong inhibitors of CYP3A, P-gp, and UGT1A1 together (i.e. all 3 pathways), such as atazanavir/cobicistat, may significantly increase plasma concentrations of Sunlenca, and therefore co-administration is not recommended (see Section 4.4 Special Warnings and Precautions for Use).
Effect of Sunlenca on the pharmacokinetics of other medicinal products.
Lenacapavir is a moderate inhibitor of CYP3A and a P-gp inhibitor. Caution is advised if Sunlenca is coadministered with a sensitive CYP3A and/or P-gp substrate with a narrow therapeutic index. Lenacapavir is not a clinically meaningful inhibitor of BCRP and does not inhibit OATP.
Use of other medicinal products drugs after discontinuation of Sunlenca.
If Sunlenca is discontinued, residual concentrations of lenacapavir may remain in the systemic circulation of patients for prolonged periods. These concentrations may affect the exposures of other drugs (i.e. sensitive CYP3A substrates) that are initiated within 9 months after the last subcutaneous dose of Sunlenca. These concentrations are not expected to affect the exposures of other antiretroviral agents that are initiated after discontinuation of Sunlenca.
Established and other potentially significant drug interactions.
Drug interaction information for Sunlenca with potential concomitant drugs is summarised in Table 2. The drug interactions described are based on the results of the studies conducted with Sunlenca, or are potential drug interactions that may occur with Sunlenca.


4.6 Fertility, Pregnancy and Lactation
Effects on fertility.
There are no data on the effects of lenacapavir on human male or female fertility. In rats, subcutaneous administration of lenacapavir (6 weeks prior to mating in males and 4 weeks prior to mating in females) had no adverse effects on fertility at doses up to 100 mg/kg (6 and 3.9 times the human clinical exposure based on AUC in males and females, respectively).
(Category B1)
There are no adequate and well-controlled studies of Sunlenca in pregnant women. Because animal reproductive studies are not always predictive of human response, Sunlenca should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
Studies in animals have shown no evidence of teratogenicity or an effect on reproductive function. No adverse effects on embryofetal development were seen in rats receiving ≤ 30 mg/kg/day PO from gestation day (GD) 6-17 (13 times the predicted human exposure at the recommended clinical dose). In rabbits, doses of up to 20 mg/kg/day IV from GD7-19 caused no adverse effects on embryofetal development (at 99 times the predicted human exposure).
It is unknown whether lenacapavir is excreted in human milk. After a single subcutaneous dose of 300 mg/kg on gestation day 6 to rats, lenacapavir was detected at low levels in the plasma of nursing rat pups, without effects on these nursing pups, at 3.9 times the predicted human exposure.4.7 Effects on Ability to Drive and Use Machines
The effects of this medicine on a person's ability to drive and use machines were not assessed as part of its registration.
4.8 Adverse Effects (Undesirable Effects)
Experience from clinical studies.
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The primary safety assessment of Sunlenca was based on data from heavily treatment experienced adult participants with HIV who received Sunlenca in a Phase 2/3 trial (CAPELLA; N=72) through week 156 (median duration on study of 165 weeks) [see Section 5.1, Clinical trials], as well as supportive data in treatment-naïve adult participants with HIV who received Sunlenca in a Phase 2 trial (CALIBRATE; N=157) at least through Week 80 (median duration of exposure of 152 weeks).
The most common adverse reactions (all Grades) reported in at least 3% of participants in CAPELLA were diarrhoea, nausea and injection site reactions.
Table 3 displays the frequency of adverse reactions (all Grades) greater than or equal to 3% in the Sunlenca group.
 The majority (89%) of all adverse reactions associated with Sunlenca were mild or moderate in severity.
The majority (89%) of all adverse reactions associated with Sunlenca were mild or moderate in severity.
During the functional monotherapy period of CAPELLA, the only adverse event reported in more than one subject receiving Sunlenca was nausea (see Table 4).
 Laboratory abnormalities. The frequency of laboratory abnormalities (Grades 3 to 4) occurring in at least 2% of participants in CAPELLA are presented in Table 5. A causal association between Sunlenca and these laboratory abnormalities has not been established.
Laboratory abnormalities. The frequency of laboratory abnormalities (Grades 3 to 4) occurring in at least 2% of participants in CAPELLA are presented in Table 5. A causal association between Sunlenca and these laboratory abnormalities has not been established.
 Description of selected adverse reactions.
Description of selected adverse reactions. Immune reconstitution inflammatory syndrome.
In patients with HIV with severe immune deficiency at the time of initiation of CART, an inflammatory reaction to asymptomatic or residual opportunistic infections may arise. Autoimmune disorders (such as Graves' disease and autoimmune hepatitis) have also been reported; however, the reported time to onset is more variable and these events can occur many months after initiation of treatment (see Section 4.4 Special Warnings and Precautions for Use).
Local injection site reactions.
Through at least Week 156, most participants had ISRs that were mild (Grade 1, 54%) or moderate (Grade 2, 17%). Six percent (4/72) of participants experienced a severe Grade 3 ISR with a median time to resolution of 15 (range: 1 to 71) days. No participants experienced a Grade 4 ISR. The median duration of all ISRs excluding nodules and indurations was 5 days. The median duration of nodules and indurations was 288 and 190 days, respectively. After a median follow-up of 554 days, nodules associated with the first injections of Sunlenca had not resolved in 10% (7/72) of the participants. All indurations associated with the first injections of Sunlenca had resolved.
Reporting suspected adverse effects.
Reporting suspected adverse reactions after registration of the medicinal product is important.
It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.4.9 Overdose
If overdose occurs, the patient must be monitored for evidence of toxicity. Treatment of overdose with Sunlenca consists of general supportive measures including monitoring of vital signs as well as observation of the clinical status of the patient. As lenacapavir is highly protein bound, it is unlikely to be significantly removed by dialysis.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action.
Lenacapavir is a multistage, selective inhibitor of HIV-1 capsid function that directly binds to the interface between capsid protein (CA) subunits. Lenacapavir inhibits HIV-1 replication by interfering with multiple, essential steps of the viral lifecycle, including capsid-mediated nuclear uptake of HIV-1 proviral DNA (by blocking nuclear import proteins binding to capsid), virus assembly (by interfering with Gag/Gag-Pol functioning, reducing production of CA subunits), and capsid core formation (by disrupting the rate of capsid subunit association, leading to virions with malformed capsids and reduced infectivity).
Lenacapavir has activity that is specific to human immunodeficiency virus (HIV-1 and HIV-2).
Antiviral activity.
The antiviral activity of lenacapavir against laboratory and clinical isolates of HIV-1 was assessed in lymphoblastoid cell lines, PBMCs, primary monocyte/macrophage cells, and CD4+ T-lymphocytes. The EC50 and selectivity (CC50/EC50) values ranged from 30 to 190 picoM and 140,000 to > 1,670,000, respectively, for wild-type HIV-1 virus. The protein binding adjusted EC95 for lenacapavir was 4 nanoM (3.87 nanogram per mL) in the MT-4 T-cell line for wild-type HIV-1 virus.
In a study of lenacapavir in combination with representatives from the main classes of antiretroviral agents (NRTIs, NNRTIs, INSTIs, and PIs), synergistic antiviral effects were observed. No antagonism was observed for these combinations.
Lenacapavir displayed antiviral activity in cell culture against all HIV-1 groups (M, N, O), including subtypes A, A1, AE, AG, B, BF, C, D, E, F, G, H.
Lenacapavir was 15- to 25-fold less active against HIV-2 isolates relative to HIV-1.
Resistance.
In cell culture.
HIV-1 variants with reduced susceptibility to lenacapavir have been selected in cell culture.
In vitro resistance selections with lenacapavir identified 7 mutations in CA: L56I, M66I, Q67H, K70N, N74D/S, and T107N singly or in dual combination. Another mutation conferring resistance to lenacapavir was N57H. Phenotypic susceptibility to lenacapavir was reduced 4- to > 5000-fold, relative to wild-type virus.
In heavily treatment experienced patients.
In CAPELLA, 39% (28/72) of participants met the criteria for resistance analyses through Week 156 (HIV-1 RNA ≥ 50 copies/mL at confirmed virologic failure [suboptimal virologic response at Week 4, virologic rebound, or viremia at last visit]) and were analysed for lenacapavir-associated mutation emergence. Lenacapavir-associated capsid mutations were found in 19% (n=14) participants. The M66I CA mutation was observed in 8% (n=6) of participants, alone or in combination with other lenacapavir-associated capsid mutations including, Q67Q/H//K/N, K70K/N/R/S, N74D/H, A105T, and T107T/A/C. Four participants had emergence of Q67H + K70R in CA with or without A105T and/or T107N. One participant had emergence of K70N + N74K +T107T/N, one participant had emergence of N74D alone, one participant had emergence of Q67Q/H alone and one participant had emergence of Q67K + K70H. Eight participants with virologic failure had emergent resistance substitutions to components of the optimised background regimen (OBR).
Phenotypic analyses indicated that the M66I and Q67K + K70H mutation patterns were associated with decrease in lenacapavir susceptibility of 234-fold (median) and 167-fold, respectively, in comparison to wild-type. The Q67H + K70R + A105T or T107N resistance pattern was associated with an average 195-fold decrease in Sunlenca susceptibility compared to wild-type and Q67H + K70R alone was associated with a 15-fold decrease in Sunlenca susceptibility compared to wild-type. The presence of mutations K70N + N74K was associated with a 289-fold decrease in Sunlenca susceptibility compared to wildtype, and the Q67Q/H mutation was associated with a 5.9-fold decrease in Sunlenca susceptibility compared to wild-type.
Cross resistance.
The in vitro antiviral activity of lenacapavir was determined against a broad spectrum of HIV-1 site-directed mutants and patient-derived HIV-1 isolates with resistance to the 4 main classes of antiretroviral agents (NRTIs, NNRTIs, INSTIs and PIs; n=58), as well as to viruses resistant to maturation inhibitors (n=32), and to viruses resistant to the entry inhibitors (EI) class (fostemsavir, ibalizumab, maraviroc, and enfuvirtide; n=42). These data indicated that lenacapavir remained fully active against all variants tested, thereby demonstrating a nonoverlapping resistance profile. In addition, the antiviral activity of lenacapavir in patient isolates was unaffected by the presence of naturally occurring Gag polymorphisms.
Effects on electrocardiogram.
In a parallel-design thorough QT/QTc study, lenacapavir had no clinically relevant effect on the QTcF interval. At supratherapeutic exposures of lenacapavir (9-fold higher than the therapeutic exposures of lenacapavir), the predicted mean (upper 90% confidence interval) increase in QTcF interval was 2.6 (4.8) msec, and there was no association (p=0.36) between observed lenacapavir plasma concentrations and change in QTcF.
Clinical trials.
The efficacy and safety of Sunlenca in heavily treatment experienced participants with multidrug resistant HIV-1 is based on 156-week data from a partially randomised, placebo-controlled, double-blind, multicentre study, GS-US-200-4625 ("CAPELLA").
CAPELLA was conducted in 72 heavily treatment-experienced participants with multiclass resistant HIV-1. Participants were required to have a viral load ≥ 400 copies/mL, documented resistance to at least two antiretroviral medications from each of at least 3 of the 4 classes of antiretroviral medications (nucleoside reverse transcriptase inhibitors [NRTI], non-nucleoside reverse transcriptase inhibitors [NNRTI], protease inhibitors [PI] and integrase strand-transfer inhibitors [INSTI]), and ≤ 2 fully active antiretroviral medications from the 4 classes of antiretroviral medications remaining at baseline due to resistance, intolerability, drug access, contraindication, or other safety concerns.
The trial was composed of two cohorts. Participants were enrolled into the randomised cohort (Cohort 1) if they had a < 0.5 log10 HIV-1 RNA decline compared to the screening visit.
Participants were enrolled into the non-randomised cohort (Cohort 2) if they had a ≥ 0.5 log10 HIV-1 RNA decline compared to the screening visit or after Cohort 1 reached its planned sample size. Participants were administered 600 mg, 600 mg, and 300 mg lenacapavir orally on Days 1, 2, and 8, respectively, followed by 927 mg subcutaneously on Day 15 and 927 mg subcutaneously every 6 months thereafter (see Section 5.1 Pharmacodynamic Properties).
Cohort 1 (N=36, randomised).
In the 14-day functional monotherapy period, participants in cohort 1 were randomised in a 2:1 ratio in a blinded fashion, to receive either Sunlenca or placebo, while continuing their failing regimen. This period was to establish the virologic activity of Sunlenca. After the functional monotherapy period, participants who had received Sunlenca continued on Sunlenca along with an OBR; participants who had received placebo during this period initiated Sunlenca along with an OBR.
Participants in cohort 1 had a mean age of 52 years (range: 24 to 71), 72% were male, 46% were White, 46% were Black, and 9% were Asian. 29% percent of participants identified as Hispanic/Latino. The mean baseline plasma HIV-1 RNA was 4.3 log10 copies/mL (range: 2.3 to 5.4). 19% of participants had baseline viral loads greater than 100,000 copies/mL. The mean baseline CD4+ cell count was 161 cells/mm3 (range: 6 to 827). 75% of participants had CD4+ cell counts below 200 cells/mm3. The mean number of years since participants first started HIV treatment was 24 years (range: 7 to 33); the mean number of antiretroviral agents in failing regimens at baseline was 4 (range: 1 to 7). The percentage of participants in the randomised cohort with known resistance to at least 2 agents from the NRTI, NNRTI, PI and INSTI classes was 97%, 94%, 78% and 75%, respectively. In cohort 1, 53% of participants had no fully active agents, 31% had 1 fully active agent, and 17% had 2 or more fully active agents within their initial failing regimen, including 6% of participants who were receiving fostemsavir, which was an investigational agent at the start of the CAPELLA trial.
Cohort 2 (N=36, non-randomised).
Participants in cohort 2 initiated Sunlenca and an OBR on Day 1.
Participants in cohort 2 had a mean age of 48 years (range: 23 to 78), 78% were male, 36% were White, 31% were Black, 33% were Asian, and 14% of patients identified as Hispanic/Latino.
The mean baseline plasma HIV-1 RNA was 4.1 log10 copies/mL (range: 1.3 to 5.7). 19% of patients had baseline viral loads greater than 100,000 copies/mL. The mean baseline CD4+ cell count was 258 cells/mm3 (range: 3 to 1296). 53% of participants had CD4+ cell counts below 200 cells/mm3. The mean number of years since patients first started HIV treatment was 19 years (range: 3 to 35); the mean number of antiretroviral agents in failing regimens at baseline was 4 (range: 2 to 7). The percentage of participants in the non-randomised cohort with known resistance to at least 2 agents from the NRTI, NNRTI, PI and INSTI classes was 100%, 100%, 83% and 64%, respectively. In cohort 2, 31% of participants had no fully active agents, 42% had 1 fully active agent, and 28% had 2 or more fully active agents within their initial failing regimen, including 6% of participants who were receiving fostemsavir, which was an investigational agent at the start of the CAPELLA trial.
The primary efficacy endpoint was the proportion of participants in cohort 1 achieving ≥ 0.5 log10 copies/mL reduction from baseline in HIV-1 RNA at the end of the functional monotherapy period. The results of the primary endpoint analysis demonstrated the superiority of Sunlenca compared with placebo, as shown in Table 6.
 The results at Weeks 26, 52 and 156 are provided in Table 7 and Table 8.
The results at Weeks 26, 52 and 156 are provided in Table 7 and Table 8.

 In cohort 1, at Weeks 26, 52 and 156, the mean change from baseline in CD4+ cell count was 81 cells/mm3 (range: -101 to 522) and 82 cells/mm3 (range: -194 to 467), and 157 cells/mm3 (range: -93 to 659), respectively.
In cohort 1, at Weeks 26, 52 and 156, the mean change from baseline in CD4+ cell count was 81 cells/mm3 (range: -101 to 522) and 82 cells/mm3 (range: -194 to 467), and 157 cells/mm3 (range: -93 to 659), respectively.
In cohort 2, at Weeks 26, 52 and 156, 81% (29/36) and 72% (26/36) and 58% (21/36) of participants achieved HIV-1 RNA < 50 copies/mL, respectively, and the mean change from baseline in CD4+ cell count was 98 cells/mm3 (range: -103 to 459), 113 cells/ mm3 (range: -124 to 405), and 173 cells/mm3 (range: -168 to 455), respectively.
Oral bridging.
In CAPELLA across Cohorts 1 and 2, 79% of patients (57/72) received Sunlenca 300 mg once every 7 days as oral bridging. A total of 13, 29, and 15 patients started oral bridging following Weeks 26, 52, and 78 injections, respectively. The median (Q1, Q3) duration of oral bridging was 19 weeks (11, 22), and 12% (7/57) received oral bridging for at least 28 weeks.
In a post-hoc analysis, rates of virologic suppression and change from baseline in CD4+ cell counts in the subset of patients who received oral bridging were consistent before and during the oral bridging period.
5.2 Pharmacokinetic Properties
Absorption.
Oral administration.
Lenacapavir is absorbed following oral administration with peak plasma concentrations occurring 4 hours after administration of Sunlenca. Absolute bioavailability following oral administration of lenacapavir is low (approximately 6-10%). Lenacapavir is a substrate of P-gp.
Lenacapavir AUC, Cmax and Tmax were comparable following administration of a low fat (~400 kcal, 25% fat) or high fat (~1000 kcal, 50% fat) meal relative to fasted conditions. Oral lenacapavir can be administered without regard to food.
Subcutaneous administration.
Lenacapavir is completely absorbed following subcutaneous administration. Due to slow release from the site of subcutaneous administration, the absorption profile of subcutaneously administered lenacapavir is complex with peak plasma concentrations occurring 77 to 84 days postdose.
Pharmacokinetic parameters.
Simulated steady state exposures of lenacapavir following recommended dosing regimen in heavily treatment experienced participants with HIV are provided in Table 9.
 Lenacapavir exposures after subcutaneous administration were similar between heavily treatment experienced people with HIV-1 and people without HIV-1 infection based on population pharmacokinetics analysis. Lenacapavir exposures (AUCtau, Cmax and Ctrough) after oral administration were 28% to 43% higher in subjects with HIV-1 infection who were heavily treatment experienced compared to subjects without HIV-1 infection based on population PK analysis. These differences were not considered clinically relevant.
Lenacapavir exposures after subcutaneous administration were similar between heavily treatment experienced people with HIV-1 and people without HIV-1 infection based on population pharmacokinetics analysis. Lenacapavir exposures (AUCtau, Cmax and Ctrough) after oral administration were 28% to 43% higher in subjects with HIV-1 infection who were heavily treatment experienced compared to subjects without HIV-1 infection based on population PK analysis. These differences were not considered clinically relevant.
Distribution.
Lenacapavir steady state volume of distribution was 976 litres in heavily treatment experienced participants with HIV 1 infection based on population pharmacokinetic analysis.
Lenacapavir is highly bound to plasma proteins (> 98.5%).
Biotransformation.
Following a single intravenous dose of radiolabelled-lenacapavir to healthy participants, 76% of the total radioactivity was recovered from faeces and < 1% from urine. Unchanged lenacapavir was the predominant moiety in plasma (69%) and faeces (33%). Metabolism played a lesser role in lenacapavir elimination. Lenacapavir was metabolised via oxidation, N-dealkylation, hydrogenation, amide hydrolysis, glucuronidation, hexose conjugation, pentose conjugation, and glutathione conjugation; primarily via CYP3A and UGT1A1. No single circulating metabolite accounted for > 10% of plasma drug-related exposure.
Elimination.
The median half-life following oral and subcutaneous administration ranged from 10 to 12 days, and 8 to 12 weeks, respectively. Lenacapavir clearance was 3.62 L/h in heavily treatment experienced participants with HIV 1 infection based on population pharmacokinetic analysis.
Linearity/non-linearity.
The single dose pharmacokinetics of lenacapavir after oral administration are non-linear and less than dose proportional over the dose range of 50 to 1800 mg.
The single dose pharmacokinetics of lenacapavir after subcutaneous injection (309 mg/mL) are dose proportional over the dose range of 309 to 927 mg.
Pharmacokinetics in special populations.
Age, gender and race.
Population PK analyses using data from adult trials did not identify any clinically relevant differences in the exposure of lenacapavir due to age, gender, race/ethnicity or weight.
Hepatic impairment.
The pharmacokinetics of a single 300 mg oral dose of lenacapavir were evaluated in a dedicated study in participants with moderate hepatic impairment (Child-Pugh Class B). Lenacapavir mean exposures were increased (47% and 161% for AUCinf and Cmax, respectively) in participants with moderate hepatic impairment (Child-Pugh B) compared to participants with normal hepatic function; however, the increase was not considered clinically relevant. The pharmacokinetics of lenacapavir have not been studied in patients with severe hepatic impairment (Child-Pugh C).
Renal impairment.
The pharmacokinetics of a single 300 mg oral dose of lenacapavir were evaluated in a dedicated study in participants with severe renal impairment (estimated creatinine clearance ≥ 15 and < 30 mL/minute). Lenacapavir exposures were increased (84% and 162% for AUCinf and Cmax, respectively) in participants with severe renal impairment compared with participants with normal renal function; however, the increase was not considered clinically relevant. The pharmacokinetics of lenacapavir have not been studied in participants with end-stage renal disease, including those on dialysis. As lenacapavir is > 98.5% protein bound, dialysis is not expected to alter exposures of lenacapavir.
5.3 Preclinical Safety Data
Genotoxicity.
Lenacapavir was not mutagenic in bacteria or clastogenic in cultured mammalian cells in vitro or an in vivo rat micronucleus assay.
Carcinogenicity.
Lenacapavir was not carcinogenic in a 6-month rasH2 transgenic mouse study at doses of up to 300 mg/kg/dose once every 13 weeks, which resulted in exposures of approximately 71 times the exposure in humans at the recommended human dose. A 104-week carcinogenicity study was conducted in male and female rats at lenacapavir doses of 0, 102, 309, or 927 mg/kg by subcutaneous injection once every 13-weeks. A treatment-related increase in the incidence of malignant sarcoma at the injection site was observed at the highest dose (927 mg/kg or 464 mg/kg per subcutaneous injection site). 11/110 animals manifested sarcomas where each animal had up to 16 injection sites - corresponding to an incidence of < 1% total injection sites across animals at the high dose. The no-observed-adverse-effect level (NOAEL) at the injection site was 17 times the recommended human dose (RHD) on a mg/kg per injection site basis. These tumors are considered to be a secondary response to chronic tissue irritation and granulomatous inflammation in rats, which are prone to sarcoma formation at the subcutaneous injection site. The clinical relevance of these findings is unknown. There were no neoplasms associated with systemic exposure to lenacapavir at any dose up to 927 mg/kg/dose once every 13 weeks, which resulted in exposures of approximately 26 times the exposure in humans at the RHD, based on AUC.6 Pharmaceutical Particulars
6.1 List of Excipients
Sunlenca injection.
Macrogol 300, water for injection.
Sunlenca tablets.
Tablet core.
Mannitol, microcrystalline cellulose, croscarmellose sodium, copovidone, magnesium stearate, poloxamer 407.
Film coat.
Polyvinyl alcohol, titanium dioxide, macrogol 3350, purified talc, iron oxide yellow, iron oxide black, iron oxide red.
See Section 2 Qualitative and Quantitative Composition.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Sunlenca injection.
Store in the original package. Store below 30°C. Keep the vials in the original carton until just prior to preparation of the injections in order to protect them from light. Once the solution has been drawn into the syringes, the injections should be administered as soon as possible.
Sunlenca tablets.
Store in the original package. Store below 30°C.
6.5 Nature and Contents of Container
Sunlenca injection is packaged in one of two different injection kits.
Vial access device injection kit.
Sunlenca injection is packaged in a dosing kit containing:
2 single-use clear glass vials of Sunlenca, each containing sufficient volume to allow withdrawal of 1.5 mL/463.5 mg (309 mg/mL) of lenacapavir. Vials are sealed with an elastomeric closure and aluminium overseal with flip-off cap;
2 vial access devices, 2 disposable syringes, and 2 injection safety needles for subcutaneous injection (22-gauge, ½ inch).
Withdrawal needle injection kit.
Sunlenca injection is packaged in a withdrawal needle injection kit containing:
2 single-use clear glass vials of Sunlenca, each containing sufficient volume to allow
withdrawal of 1.5 mL/463.5 mg (309 mg/mL) of lenacapavir. Vials are sealed with an
elastomeric closure and aluminum overseal with flip-off cap;
2 disposable syringes, 2 withdrawal needles (18-gauge, 1½ inch), and 2
injection safety needles for subcutaneous injection (22-gauge, ½ inch).
The Sunlenca tablets blister pack presentation is intended for the initiation regimen. The Sunlenca tablets bottle presentation is intended for the oral bridging regimen (see Section 4.2 Dose and Method of Administration).
Sunlenca tablets are packaged in a blister pack containing: five (5) tablets of Sunlenca, each containing 300 mg of lenacapavir, are packaged in a clear blister film sealed to a foil lidding material. The blister card, which is fitted between child-resistant sealed paperboard cards, is packaged with silica gel desiccant in a sealed flexible laminated pouch.
Sunlenca tablets are packaged in a bottle containing: four (4) tablets are packaged in a 45 mL, white, high-density polyethylene (HDPE) bottle containing polyester coil and silica gel desiccant. Each bottle is capped using a white, continuous thread, child-resistant polypropylene screw cap with an induction sealed, aluminium-faced liner.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of in accordance with local requirements.
6.7 Physicochemical Properties
Chemical structure.
The chemical name of lenacapavir is (4-chloro-7-(2-((S)-1-(2-((3bS,4aR)-5,5-difluoro-3-(trifluoromethyl)-3b,4,4a,5-tetrahydro-1H -cyclopropa[3,4] cyclopenta[1,2-c]pyrazol-1-yl)acetamido)-2-(3,5-difluorophenyl)ethyl)-6-(3-methyl-3-(methylsulfonyl)but-1-yn-1-yl)pyridin-3-yl)-1-(2,2,2-trifluoroethyl)-1H-indazol-3-yl)(methylsulfonyl)amide.

CAS number.
2283356-12-5.7 Medicine Schedule (Poisons Standard)
Schedule 4 - Prescription Only Medicine.
Summary Table of Changes










 The majority (89%) of all adverse reactions associated with Sunlenca were mild or moderate in severity.
The majority (89%) of all adverse reactions associated with Sunlenca were mild or moderate in severity.

 The results at Weeks 26, 52 and 156 are provided in Table 7 and Table 8.
The results at Weeks 26, 52 and 156 are provided in Table 7 and Table 8.
 In cohort 1, at Weeks 26, 52 and 156, the mean change from baseline in CD4+ cell count was 81 cells/mm3 (range: -101 to 522) and 82 cells/mm3 (range: -194 to 467), and 157 cells/mm3 (range: -93 to 659), respectively.
In cohort 1, at Weeks 26, 52 and 156, the mean change from baseline in CD4+ cell count was 81 cells/mm3 (range: -101 to 522) and 82 cells/mm3 (range: -194 to 467), and 157 cells/mm3 (range: -93 to 659), respectively. Lenacapavir exposures after subcutaneous administration were similar between heavily treatment experienced people with HIV-1 and people without HIV-1 infection based on population pharmacokinetics analysis. Lenacapavir exposures (AUCtau, Cmax and Ctrough) after oral administration were 28% to 43% higher in subjects with HIV-1 infection who were heavily treatment experienced compared to subjects without HIV-1 infection based on population PK analysis. These differences were not considered clinically relevant.
Lenacapavir exposures after subcutaneous administration were similar between heavily treatment experienced people with HIV-1 and people without HIV-1 infection based on population pharmacokinetics analysis. Lenacapavir exposures (AUCtau, Cmax and Ctrough) after oral administration were 28% to 43% higher in subjects with HIV-1 infection who were heavily treatment experienced compared to subjects without HIV-1 infection based on population PK analysis. These differences were not considered clinically relevant.
