1 Name of Medicine
Talquetamab.
2 Qualitative and Quantitative Composition
Talvey (talquetamab) is a humanised immunoglobulin G4-proline, alanine, alanine (IgG4-PAA)-based bispecific antibody directed against G Protein-coupled receptor family C group 5 member D (GPRC5D) and the cluster of differentiation 3 (CD3) receptors, produced by cultivation of recombinant Chinese hamster ovary cells, followed by isolation, chromatographic purification, and formulation.
Talvey is available in the following presentations:
Each 1.5 mL vial contains 3 mg of talquetamab (2 mg of talquetamab per mL).
Each 1.0 mL vial contains 40 mg of talquetamab (40 mg of talquetamab per mL).
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Talvey is a colourless to light yellow preservative-free solution for injection.
4.1 Therapeutic Indications
Talvey as monotherapy has provisional approval in Australia and is indicated for the treatment of adult patients with relapsed or refractory multiple myeloma, who have previously received at least four prior therapies, including a proteasome inhibitor, an immunomodulatory agent, and an anti-CD38 antibody.
The decision to approve this indication has been made on the basis of the overall response rate in a single arm study. Continued approval of this indication depends on verification and description of benefit in confirmatory trials.
4.2 Dose and Method of Administration
Dosage - adults (18 years of age and older).
Talvey is administered via subcutaneous injection.
Administer pretreatment medications prior to each dose of the Talvey during the step-up phase (see Section 4.2 Dose and Method of Administration, Pretreatment medications).
Administer Talvey subcutaneously on a weekly or biweekly (every 2 weeks) dosing schedule to Table 1.
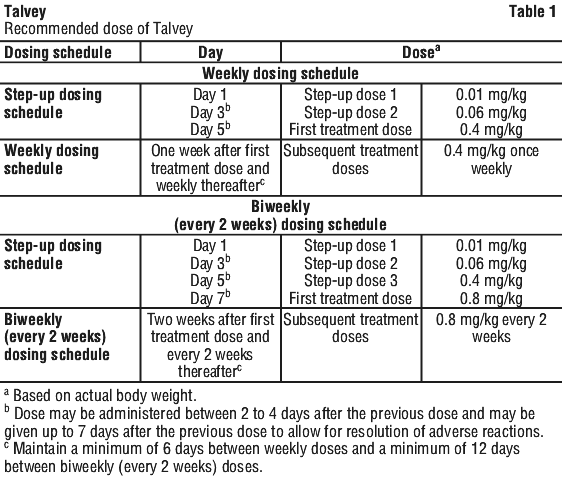 Instruct patients to remain within proximity of a healthcare facility and monitor patients for 48 hours after administration of all doses within the Talvey step-up phase for signs and symptoms of cytokine release syndrome (CRS) and immune effector cell-associated neurotoxicity syndrome (ICANS). (See Section 4.4 Special Warnings and Precautions for Use, Cytokine release syndrome (CRS), Neurologic toxicities, including immune effector cell-associated neurotoxicity syndrome (ICANS)).
Instruct patients to remain within proximity of a healthcare facility and monitor patients for 48 hours after administration of all doses within the Talvey step-up phase for signs and symptoms of cytokine release syndrome (CRS) and immune effector cell-associated neurotoxicity syndrome (ICANS). (See Section 4.4 Special Warnings and Precautions for Use, Cytokine release syndrome (CRS), Neurologic toxicities, including immune effector cell-associated neurotoxicity syndrome (ICANS)).
Continue treatment until disease progression or unacceptable toxicity.
Pretreatment medications.
Administer the following pretreatment medications 1 to 3 hours before each dose of the Talvey step-up phase to reduce the risk of CRS (see Section 4.4 Special Warnings and Precautions for Use, Cytokine release syndrome (CRS)).
Corticosteroid (oral or intravenous dexamethasone, 16 mg or equivalent).
Antihistamine (oral or intravenous diphenhydramine, 50 mg or equivalent).
Antipyretics (oral paracetamol 500 mg to 1000 mg).
Administration of pretreatment medications may be required for subsequent doses of Talvey for patients who repeat doses within the Talvey step-up phase due to dose delays (Table 2) or for patients who experience CRS (Table 3).
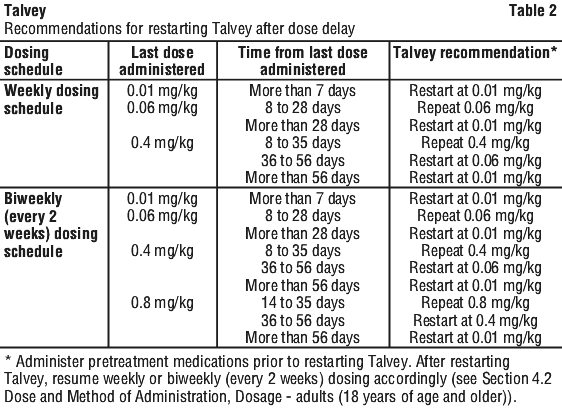
Restarting Talvey after dose delays.
If a dose of Talvey is delayed, restart therapy based on the recommendations listed in Table 2 and resume weekly or biweekly (every 2 weeks) dosing accordingly (see Section 4.2 Dose and Method of Administration, Dosage - adults (18 years of age and older)). Administer pretreatment medications prior to restarting Talvey and monitor patients following administration of Talvey accordingly (see Section 4.2 Dose and Method of Administration, Pretreatment medications). See Table 2.
Dose modifications for adverse reactions.
Dose delays may be required to manage toxicities related to Talvey (see Section 4.4 Special Warnings and Precautions for Use).
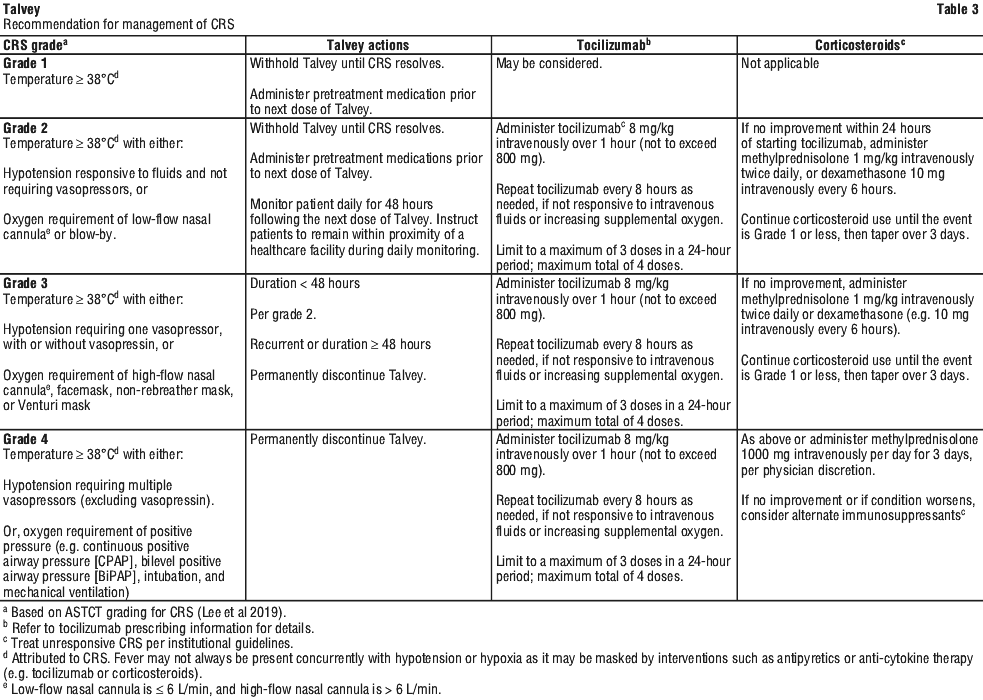
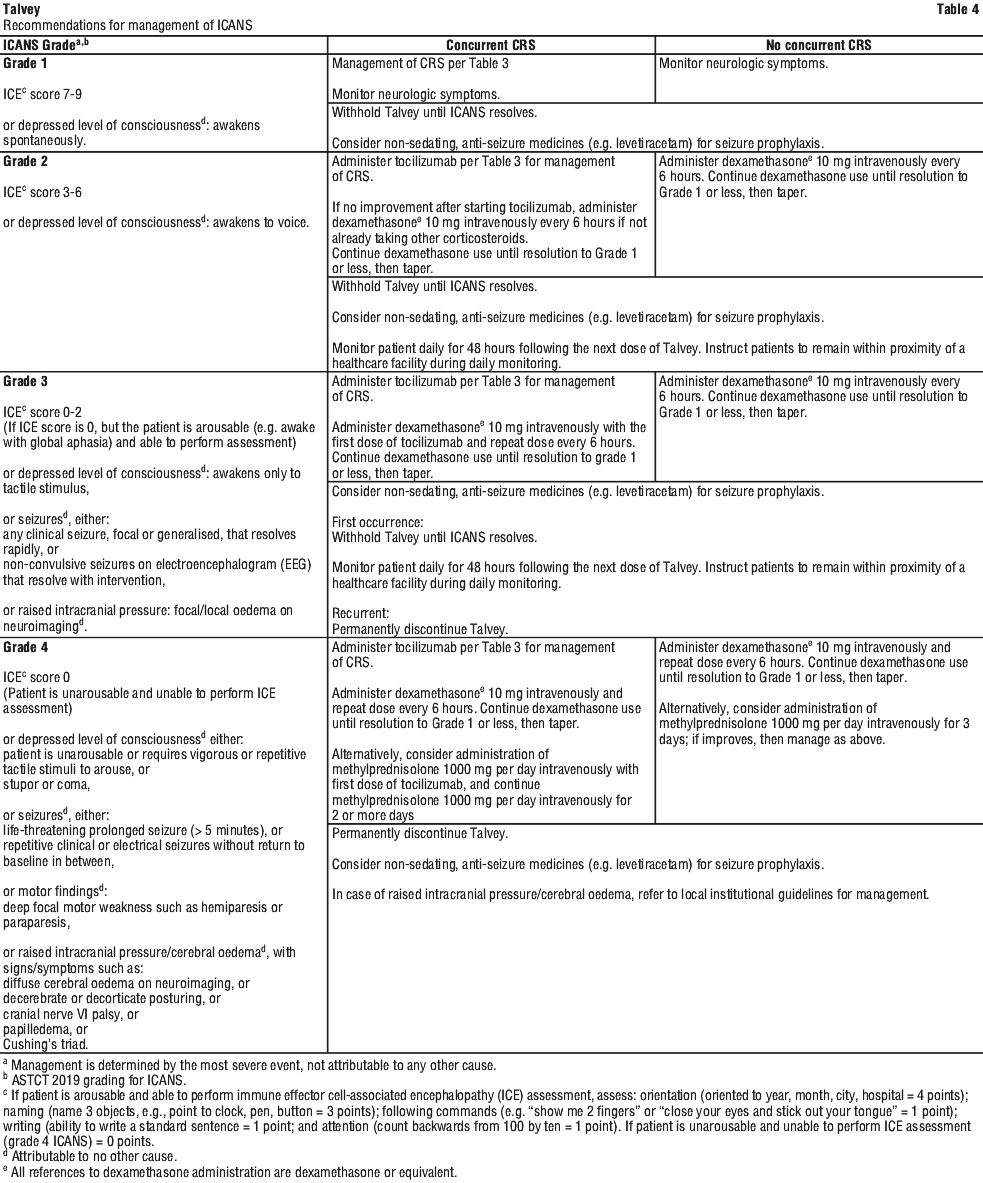
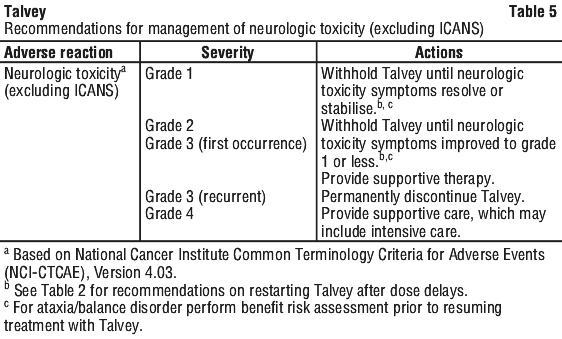
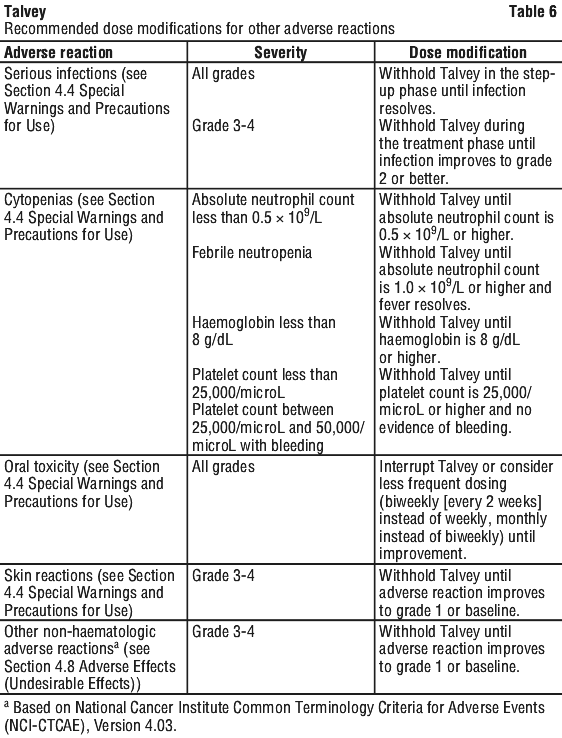
See Table 3, Table 4 and Table 5 for recommended actions for the management of CRS, ICANS and neurologic toxicities. See Table 6 for recommended dose modifications for other adverse reactions.
Management of severe adverse reactions.
Cytokine release syndrome (CRS).
Identify CRS based on clinical presentation (see Section 4.4 Special Warnings and Precautions for Use, Cytokine release syndrome (CRS)). Evaluate and treat other causes of fever, hypoxia, and hypotension. If CRS is suspected, withhold Talvey until the adverse reaction resolves and manage according to the recommendations in Table 3. Administer supportive care for CRS which may include intensive care for severe or life-threatening CRS. Consider laboratory testing to monitor for disseminated intravascular coagulation (DIC), hematology parameters, as well as pulmonary, cardiac, renal, and hepatic function.
Neurologic toxicities, including immune effector cell-associated neurotoxicity syndrome (ICANS).
At the first sign of neurologic toxicity, including ICANS, withhold Talvey and consider neurology evaluation. Rule out other causes of neurologic symptoms. Provide supportive therapy, which may include intensive care, for severe or life-threatening ICANS (see Section 4.4 Special Warnings and Precautions for Use, Neurologic toxicities, including immune effector cell-associated neurotoxicity syndrome (ICANS). Management recommendations for ICANS and neurologic toxicity are summarised in Table 4 and Table 5.
Other adverse reactions.
The recommended dose modifications for other adverse reactions are provided in Table 6.
Dosage in special populations.
Use in renal impairment.
No formal studies of Talvey in patients with renal impairment have been conducted.
Based on population pharmacokinetic analyses, no dose adjustment is recommended for patients with mild or moderate renal impairment (see Section 5.2 Pharmacokinetic Properties).
Use in hepatic impairment.
No formal studies of talquetamab in patients with hepatic impairment have been conducted.
Based on population pharmacokinetic analyses, no dose adjustment is recommended for patients with mild or moderate hepatic impairment (see Section 5.2 Pharmacokinetic Properties).
Administration.
Administer Talvey via subcutaneous injection.
Talvey should be administered by a healthcare professional with adequate medical equipment and personnel to manage severe reactions, including cytokine release syndrome (see Section 4.4 Special Warnings and Precautions for Use, Cytokine release syndrome (CRS)).
Talvey 2 mg/mL vial and 40 mg/mL vial are supplied as ready-to-use solution for injection that do not need dilution prior to administration.
Do not combine Talvey vials of different concentrations to achieve treatment dose. Use aseptic technique to prepare and administer Talvey.
Preparation of Talvey.
Refer to the following reference tables for the preparation of Talvey.
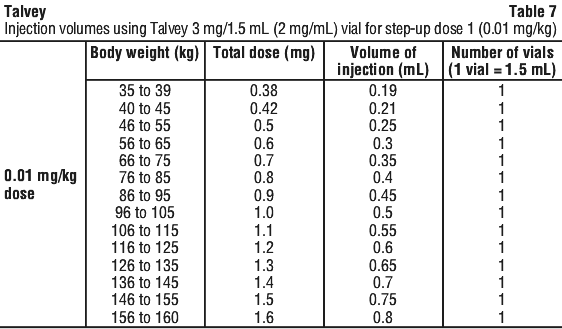
See Table 7 to determine total dose, injection volume, and number of vials required based on patient's actual body weight for the 0.01 mg/kg dose using Talvey 2 mg/mL vial.
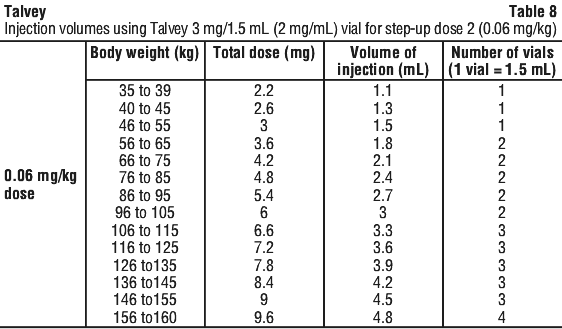
 See Table 8 to determine total dose, injection volume, and number of vials required based on patient's actual body weight for the 0.06 mg/kg dose using Talvey 2 mg/mL vial.
See Table 8 to determine total dose, injection volume, and number of vials required based on patient's actual body weight for the 0.06 mg/kg dose using Talvey 2 mg/mL vial.
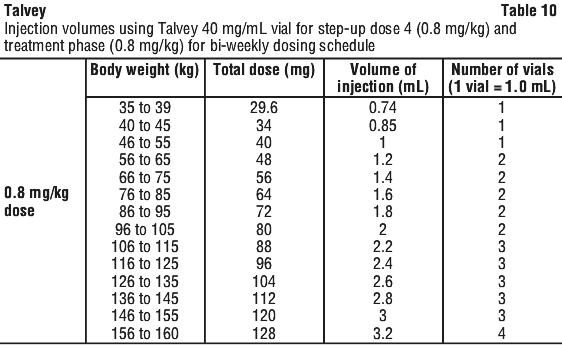
 See Table 9 to determine total dose, injection volume, and number of vials required based on patient's actual body weight for the 0.4 mg/kg dose using Talvey 40 mg/mL vial.
See Table 9 to determine total dose, injection volume, and number of vials required based on patient's actual body weight for the 0.4 mg/kg dose using Talvey 40 mg/mL vial.
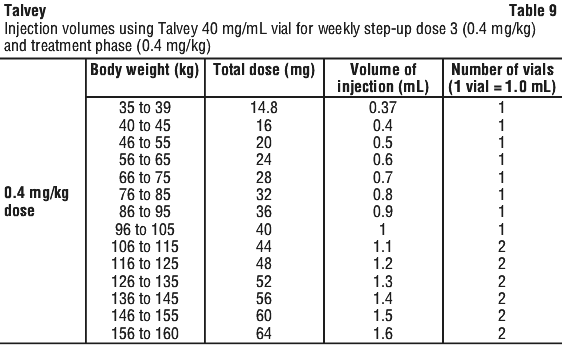 See Table 10 to determine total dose, injection volume, and number of vials required based on patient's actual body weight for the 0.8 mg/kg dose using Talvey 40 mg/mL vial.
See Table 10 to determine total dose, injection volume, and number of vials required based on patient's actual body weight for the 0.8 mg/kg dose using Talvey 40 mg/mL vial.
 Visually inspect the Talvey solution for injection is colourless to light yellow. Do not use if the solution is discoloured, cloudy, or if foreign particles are present.
Visually inspect the Talvey solution for injection is colourless to light yellow. Do not use if the solution is discoloured, cloudy, or if foreign particles are present.
Remove the appropriate strength Talvey vial(s) from refrigerated storage 2°C to 8°C and equilibrate to ambient temperature 15°C to 30°C for at least 15 minutes in the original carton protected from light. Do not warm Talvey in any other way.
Once equilibrated, gently swirl the vial for approximately 10 seconds to mix. Do not shake.
Withdraw the required injection volume of Talvey from the vial(s) into an appropriately sized syringe using a transfer needle. Each injection volume should not exceed 2.0 mL. Divide doses requiring greater than 2.0 mL equally into multiple syringes.
Talvey is compatible with stainless steel injection needles and polypropylene or polycarbonate syringe material.
Replace the transfer needle with an appropriately sized needle for injection.
Administration of Talvey.
Inject the required volume of Talvey into the subcutaneous tissue of the abdomen (preferred injection site). Alternatively, Talvey may be injected into the subcutaneous tissue at other sites (e.g. thigh). If multiple injections are required, Talvey injections should be at least 2 cm apart.
Do not inject into tattoos or scars or areas where the skin is red, bruised, tender, hard or not intact.
Any unused medicinal product or waste material should be disposed in accordance with local requirements.
Product is for single use in one patient only. Discard any residue.4.3 Contraindications
Hypersensitivity to the active substance or to any of the excipients, see Section 6.1 List of Excipients.
4.4 Special Warnings and Precautions for Use
Cytokine release syndrome (CRS).
Cytokine release syndrome, including life-threatening or fatal reactions, may occur in patients receiving Talvey (see Section 4.8 Adverse Effects (Undesirable Effects)). Clinical signs and symptoms of CRS may include but are not limited to pyrexia, hypotension, chills, hypoxia, headache, and tachycardia. Potentially life-threatening complications of CRS may include cardiac dysfunction, acute respiratory distress syndrome, neurologic toxicity, renal and/or hepatic failure, and disseminated intravascular coagulation (DIC).
Initiate Talvey therapy with step-up phase dosing and administer pre-treatment medications (corticosteroids, antihistamine, and antipyretics) prior to each dose of Talvey during the step-up phase to reduce the risk of CRS. Monitor patients following administration of Talvey accordingly. In patients who experience CRS, administer pre-treatment medications prior to the next Talvey dose (see Section 4.2 Dose and Method of Administration, Dosage - adults (18 years and older), Pretreatment medications, Dose modifications for adverse reactions).
Counsel patients to seek medical attention should signs or symptoms of CRS occur. A Patient Card to inform patients of CRS associated with Talvey is available. The Patient Card should be kept with the patient at all times whilst on treatment.
At the first sign of CRS, immediately evaluate patient for hospitalisation and institute treatment with supportive care, tocilizumab and/or corticosteroids, based on severity. The use of myeloid growth factors, particularly granulocyte macrophage-colony stimulating factor (GM-CSF), should be avoided during CRS. Withhold Talvey until CRS resolves (see Section 4.2 Dose and Method of Administration, Dose modifications for adverse reactions).
Neurologic toxicities, including immune effector cell-associated neurotoxicity syndrome (ICANS).
Serious or life-threatening neurologic toxicities, including ICANS, have occurred following treatment with Talvey.
ICANS, including fatal reactions, have occurred following treatment with Talvey (see Section 4.8 Adverse Effects (Undesirable Effects)). The onset of ICANS can be concurrent with CRS, following resolution of CRS, or in the absence of CRS. Clinical signs and symptoms of ICANS may include but are not limited to confusional state, depressed level of consciousness, disorientation, somnolence, lethargy and bradyphrenia.
Monitor patients for signs and symptoms of neurologic toxicities, including ICANS and treat promptly.
Counsel patients to seek medical attention should signs or symptoms of neurologic toxicities, including ICANS occur. A Patient Card to inform patients of neurologic toxicities including ICANS associated with Talvey is available. The Patient Card should be kept with the patient at all times whilst on treatment.
At the first sign of neurologic toxicities, including ICANS, immediately evaluate the patient. Provide supportive care based on severity and withhold or discontinue Talvey based on severity and follow management recommendations (see Section 4.2 Dose and Method of Administration, Dose modifications for adverse reactions).
Ataxia/balance disorder has been reported in subjects receiving Talvey. Monitor for any new onset of or changes in pre-existing neurological signs or symptoms including ataxia, balance disorder and cerebellar symptoms that may include but are not limited to dyskinesia, dysmetria, gait disturbance, intentional tremor, nystagmus and dysarthria.
At the first sign of ataxia/balance disorder, withhold Talvey, immediately evaluate the patient, and consider neurology evaluation. Rule out other causes of neurologic symptoms and provide supportive care based on severity; withhold or permanently discontinue Talvey based on severity and consider further management per current practice guidelines (see Section 4.2 Dose and Method of Administration, Dose modifications for adverse reactions).
Oral toxicity.
Oral toxicities, including dysgeusia, dry mouth, dysphagia, and stomatitis, may occur following treatment with Talvey (see Section 4.8 Adverse Effects (Undesirable Effects)). Seventy-eight percent (78%) of patients had Grade 1 or 2 events, with Grade 3 events occurring in 2% of patients.
Monitor patients for signs and symptoms of oral toxicity. Counsel patients to seek medical attention should signs or symptoms of oral toxicity occur and provide supportive care. Supportive care may include saliva stimulating agents, steroid mouth wash, or consultation with a nutritionist. Interrupt Talvey or consider less frequent dosing (see Section 4.2 Dose and Method of Administration, Dose modifications for adverse reactions).
Over time, notable weight loss may occur (see Section 4.8 Adverse Effects (Undesirable Effects)). Weight change should be monitored regularly during therapy. Clinically significant weight loss should be further evaluated.
Serious infections.
Serious infections, including life-threatening or fatal infections, have been reported in patients receiving Talvey (see Section 4.8 Adverse Effects (Undesirable Effects)). Monitor patients for signs and symptoms of infection prior to and during treatment with Talvey and treat appropriately. Administer prophylactic antimicrobials according to local guidelines. Withhold Talvey as indicated (see Section 4.2 Dose and Method of Administration, Dose modifications for adverse reactions).
Cytopenia.
Treatment-emergent Grade 3 or 4 neutropenia and thrombocytopenia have been observed in patients who received Talvey. A majority of events occurred during the first 8 to 10 weeks. Monitor complete blood counts during treatment and withhold Talvey as warranted (see Section 4.2 Dose and Method of Administration, Dose modifications for adverse reactions).
Skin reactions.
Rash, including maculo-papular rash, erythema, and erythematous rash, occurred in patients who received Talvey (see Section 4.8 Adverse Effects (Undesirable Effects)). Monitor rash progression for early intervention and treatment with corticosteroids. Rashes should be managed aggressively with topical steroids and early consideration of a short course of oral steroids to reduce the risk of rash progression.
Vaccines.
Immune response to vaccines may be reduced when taking Talvey. The safety of immunisation with live viral vaccines during or following Talvey treatment has not been studied. Vaccination with live virus vaccines is not recommended for at least 4 weeks prior to the start of treatment, during treatment, and at least 4 weeks after treatment.
Use in the elderly (65 years of age and older).
Of the 339 patients treated with Talvey in MonumenTAL-1, 36% were 65 to less than 75 years of age, and 17% were 75 years of age or older. No clinically important differences in safety or effectiveness were observed in patients 65 to 75 years of age compared to younger patients. There are limited clinical data with talquetamab in patients 75 years of age or over. No dose adjustment is required (see Section 5.2 Pharmacokinetic Properties).
Paediatric use (17 years of age and younger).
The safety and efficacy of Talvey have not been established in paediatric patients.
Effects of laboratory tests.
No data available.4.5 Interactions with Other Medicines and Other Forms of Interactions
No drug interaction studies have been performed with Talvey.
Talquetamab causes release of cytokines (see Section 5.1 Pharmacodynamic Properties) that may suppress activity of cytochrome P450 (CYP) enzymes, potentially resulting in increased exposure of CYP substrates. The highest risk of drug-drug interaction is expected to occur from initiation of talquetamab step-up phase up to 9 days after the first treatment dose and during and after CRS (see Section 4.4 Special Warnings and Precautions for Use, Cytokine release syndrome (CRS)). Monitor for toxicity or concentrations of drugs that are CYP (e.g. CYP2C9, CYP2C19, CYP3A4/5) substrates where minimal concentration changes may lead to serious adverse reactions. Adjust the dose of the concomitant CYP (e.g. CYP2C9, CYP2C19, CYP3A4/5) substrate drugs as needed.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility.
There are no data on the effect of Talvey on fertility. Effects of Talvey on male and female fertility have not been evaluated in animal studies.
(Category C)
There are no available data on the use of Talvey in pregnant women or animal data to assess the risk of Talvey in pregnancy. Human IgG is known to cross the placenta after the first trimester of pregnancy. Therefore, talquetamab has the potential to be transmitted from the mother to the developing fetus. The effects of Talvey on the developing fetus are unknown. Based on its mechanism of action, T-cell activation with cytokine release and pro-inflammatory effects in a mother may potentially adversely affect a developing fetus. Pregnant women should be advised there may be risks to the fetus. Talvey is not recommended for women who are pregnant or for women of childbearing potential not using contraception.
Pregnancy testing.
Verify pregnancy status of females of child-bearing potential prior to initiating Talvey.
Contraception.
Advise females of reproductive potential to use effective contraception during treatment and for three months after the last dose of Talvey.
Advise male patients with a female partner of reproductive potential to use effective contraception during treatment and for three months after the last dose of Talvey.
It is not known whether talquetamab is excreted in human or animal milk, affects breastfed infants, or affects milk production. Because the potential for serious adverse reactions in breastfed infants is unknown for Talvey, advise patients not to breastfeed during treatment with Talvey and for at least 3 months after the last dose.4.7 Effects on Ability to Drive and Use Machines
Due to the potential for ICANS, patients receiving Talvey are at risk of depressed level of consciousness (see Section 4.4 Special Warnings and Precautions for Use). Advise patients to refrain from driving or operating heavy or potentially dangerous machinery during the step-up phase and for 48 hours after completion of the step-up phase (see Section 4.2 Dose and Method of Administration, Dosage - adults (18 years of age and older)) and in the event of new onset of any neurological symptoms, until symptoms resolve.
4.8 Adverse Effects (Undesirable Effects)
Throughout this section, adverse reactions are presented. Adverse reactions are adverse events that were considered to be reasonably associated with the use of talquetamab based on the comprehensive assessment of the available adverse event information. A causal relationship with talquetamab cannot be reliably established in individual cases.
Clinical studies experience.
The safety data of Talvey was evaluated in 339 adult patients with relapsed or refractory multiple myeloma, including patients exposed to prior T cell redirection therapy, treated with Talvey at the recommended dosing regimen. The median duration of treatment was 7.4 (range: 0.0 to 32.9) months.
The most frequent adverse reactions (≥ 20%) were CRS, dysgeusia, hypogammaglobulinemia, nail disorder, musculoskeletal pain, anaemia, fatigue, skin disorder, weight decreased, rash, dry mouth, neutropenia, pyrexia, xerosis, thrombocytopenia, upper respiratory tract infection, lymphopenia, diarrhoea, dysphagia, pruritus, cough, decreased appetite, pain, and headache.
Serious adverse reactions reported in ≥ 2% of patients included CRS, pyrexia, ICANS, sepsis, COVID-19, bacterial infection, pneumonia, viral infection, neutropenia, and pain.
The most frequent adverse reactions leading to treatment discontinuation were ICANS (1.1%) and weight decreased (0.9%).
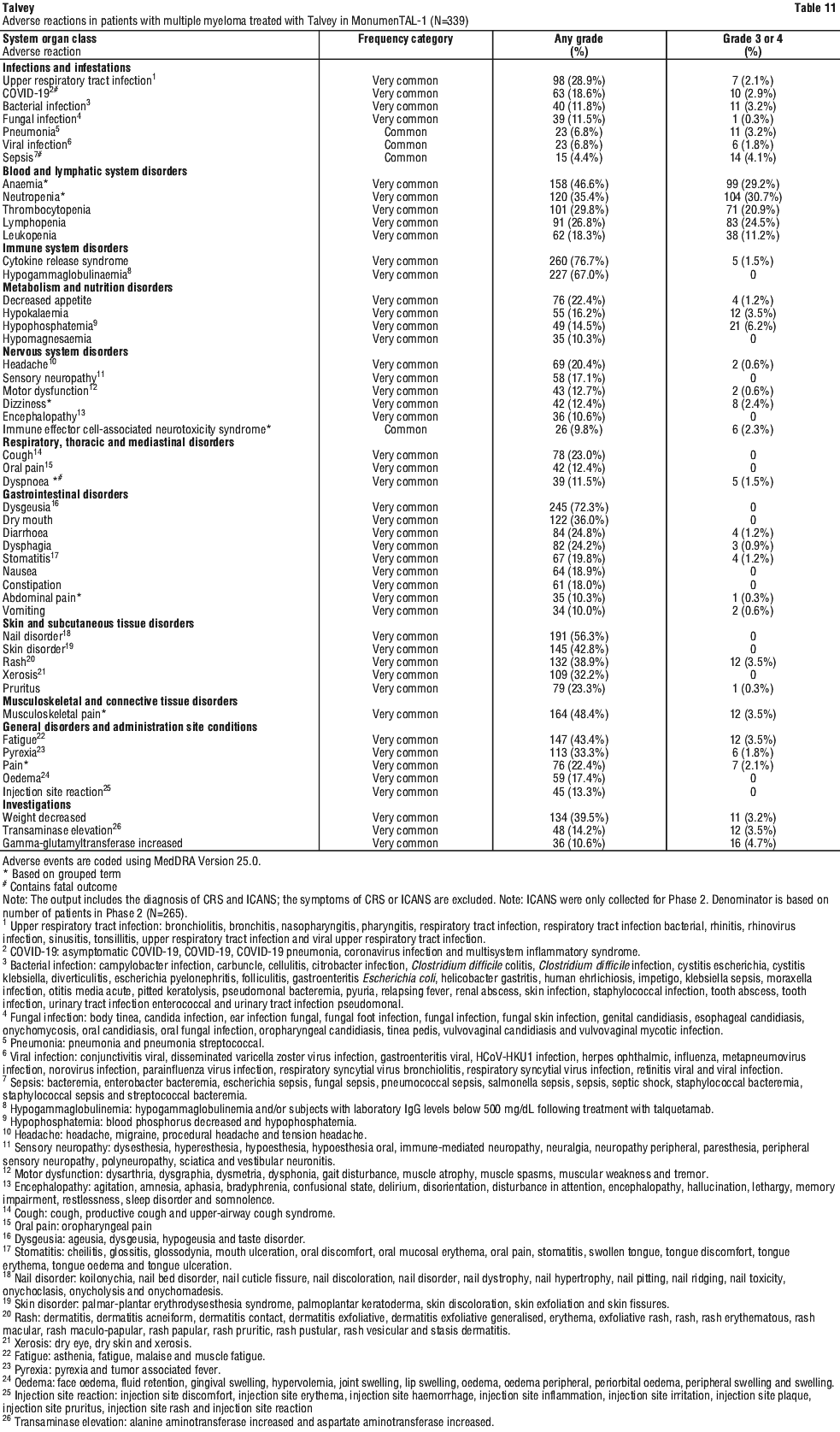
Adverse reactions observed during clinical studies are listed in Table 11 by frequency category. Frequency categories are defined as follows: very common (≥ 1/10); common (≥ 1/100 to < 1/10); uncommon (≥ 1/1,000 to < 1/100); rare (≥ 1/10,000 to < 1/1,000); very rare (< 1/10,000) and not known (frequency cannot be estimated from the available data). Within each frequency grouping, adverse reactions are presented in order of decreasing frequency.
Table 11 summarises adverse reactions reported in patients who received Talvey. See Table 11.
Description of selected adverse reactions.
Cytokine release syndrome.
In MonumenTAL-1 (N=339), CRS occurred in 77% of patients. Most events were Grade 1 or 2, with Grade 3 events occurring in 1.5% of patients. Thirty one percent (31%) of patients experienced more than one CRS event. Most events occurred during the step-up phase following the 0.01 mg/kg dose (29%), the 0.06 mg/kg dose (44%), the 0.3 mg/kg dose (for patients who received biweekly [every 2 weeks] dosing; 33%), or the initial treatment dose (0.4 mg/kg [30%] or 0.8 mg/kg [12%]). Less than 4% of CRS events occurred from Week 5 onward; all events were Grade 1. The median time to onset of CRS was 27 hours from the last dose, 91% of events occurred within 48 hours from the last dose, and the median duration was 17 hours. Tocilizumab and corticosteroids were used to treat 39% and 5% of CRS events, respectively.
Neurologic toxicities, including immune effector cell-associated neurotoxicity syndrome (ICANS).
In MonumenTAL-1 (N=339), neurologic toxicities were reported in 29% of patients receiving Talvey. Neurologic toxicity events were Grade 1 (17%), Grade 2 (10%), Grade 3 (2.4%) or Grade 4 (0.3%). The most frequently reported neurologic toxicity event was headache (9%).
In MonumenTAL-1 (N=265), ICANS occurred in 10% (n=26) of patients. Most events were Grade 1 or 2, with Grade 3 and 4 events occurring in 2.3% of patients. The most frequent clinical manifestation of ICANS reported were confusional state (4.2%), disorientation (1.9%), and somnolence (1.9%). Sixty-eight percent (68%) were concurrent with CRS (during or within 7 days of CRS resolution). Three percent (3%) of patients experienced more than one ICANS event. Most patients experienced ICANS during the step-up phase following the 0.01 mg/kg dose, the 0.06 mg/kg dose, or the initial treatment dose (0.4 mg/kg and 0.8 mg/kg) (3% each). The median time to onset was 28 hours from the last dose, 68% of events started within 48 hours from the last dose, and the median duration was 9 hours.
In addition, one fatal ICANS event was reported in MonumenTAL-1.
In MonumenTAL-1 (N=339), ataxia/balance disorder occurred in 4.1% of patients (N=14). Ataxia/balance disorder events were Grade 1 (1.5%), Grade 2 (2.4%), and Grade 3 (0.3%). No Grade 4 or 5 events occurred. The most frequent clinical manifestation of ataxia/balance disorder events reported were dysarthria (1.5%), gait disturbance (1.5%), and balance disorder (0.9%), and 0.9% of patients experienced more than one ataxia/balance disorder event. The median time to onset was 77 days (range 2; 463 days) from the first dose and 4 days (range 1; 13 days) from the last dose, and 7 (38.9%) of 18 events did not resolve.
Serious infections.
In MonumenTAL-1 (N=339), Grade 3 or Grade 4 infections occurred in 19% of patients, and fatal infections occurred in 1.5% of patients.
Skin reactions.
In MonumenTAL-1 (N=339), the majority of rash cases were Grade 1 or 2, with Grade 3 events occurring in 3.5% of patients. The median time to onset for rash was 22 days.
Reporting suspected adverse effects.
Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.4.9 Overdose
Symptoms and signs.
The maximum tolerated dose of talquetamab has not been determined. In clinical trials, doses of up to 1.2 mg/kg once every 2 weeks and 1.6 mg/kg monthly have been administered.
Treatment.
In the event of an overdose, the patient should be monitored for any signs or symptoms of adverse reactions and appropriate symptomatic treatment should be instituted immediately.
For information on the management of overdose, contact the Poisons Information Centre on 131126 (Australia).5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Pharmacotherapeutic group: Other monoclonal antibodies and antibody drug conjugates, ATC code: L01FX29.
Within the first month of treatment with talquetamab, activation and redistribution of T cells and induction of serum cytokines were observed.
Mechanism of action.
Talquetamab (also known as JNJ-64407564) is a humanised immunoglobulin G4 proline, alanine, alanine (IgG4 PAA) bispecific antibody directed against GPRC5D on multiple myeloma cells and the CD3 receptor on T Cells.
Talquetamab promotes enhanced T cell-mediated cytotoxicity through recruitment of CD3-expressing T cells to GPRC5D-expressing cells. This leads to the activation of T cells and induces subsequent lysis of GPRC5D-expressing cells mediated by secreted perforin and various granzymes stored in the secretory vesicles of cytotoxic T cells. Based on the expression of GPRC5D on plasma cells with minimal to no expression detected on B cells and B cell precursors, talquetamab targets multiple myeloma cells particularly, reducing potential off-target effects toward other cell lineages. In addition to non-malignant plasma cells, GPRC5D is expressed on healthy tissues such as epithelial cells in keratinised tissues of the skin and tongue.
Immunogenicity.
In MonumenTAL-1, 260 patients treated with subcutaneous talquetamab monotherapy at 0.4 mg/kg weekly or 0.8 mg/kg biweekly (every 2 weeks) were evaluated for antibodies to talquetamab. Following treatment of 0.4 mg/kg weekly or 0.8 mg/kg biweekly (every 2 weeks), 64 of 260 patients (24.6%) developed anti-talquetamab antibodies. None of these participants were positive for neutralizing antibodies to talquetamab. There was no identified clinically significant effect of anti-talquetamab antibodies on the pharmacokinetics, efficacy, or safety (e.g. CRS, systemic administration-related reaction, and injection site reaction).
Clinical trials.
The efficacy of Talvey monotherapy was evaluated in patients with relapsed or refractory multiple myeloma in a single-arm, open-label, multicentre study, MonumenTAL-1 (MMY1001). The study included patients who had previously received at least three prior therapies, including a proteasome inhibitor, an immunomodulatory agent, and an anti-CD38 monoclonal antibody. The study included patients who received prior T cell redirection therapy (N=51). Patients received Talvey 0.4 mg/kg subcutaneously weekly, following two step-up doses (0.01 and 0.06 mg/kg) in the first week of therapy, or Talvey 0.8 mg/kg subcutaneously biweekly (every 2 weeks), following three step-up doses (0.01, 0.06 and 0.3 mg/kg), until disease progression or unacceptable toxicity. Patients were hospitalised for monitoring for at least 48 hours after each Talvey dose during the step-up phase.
Of 117 patients treated with Talvey 0.4 mg/kg weekly who were not exposed to prior T cell redirection therapy, the median age was 67 (range: 46 to 86) years, 54% were male, 92% were White, and 7% were Black or African American. Patients had received a median of 5 (range: 4 to 13) prior therapies, and 79% of patients had received prior autologous stem cell transplantation (ASCT). Ninety-three percent (93%) of patients were refractory to their last therapy and 76% were refractory to a proteasome inhibitor, immunomodulatory agent, and anti-CD38 antibody. Of the 108 patients for whom baseline cytogenetic data were available, high-risk cytogenetic factors (presence of t(4:14), t(14:16), and/or del(17p)) were present in 31% of patients.
Of 113 patients treated with Talvey 0.8 mg/kg biweekly (every 2 weeks) who were not exposed to prior T cell redirection therapy, the median age was 67 (range: 38 to 84) years, 59% were male, 85% were White, and 7% were Black or African American. Patients had received a median of 6 (range: 4 to 17) prior therapies, and 80% of patients had received prior autologous stem cell transplantation (ASCT). Ninety-five percent (95%) of patients were refractory to their last therapy and 73% were refractory to a proteasome inhibitor, immunomodulatory agent, and anti-CD38 antibody. Of the 99 patients for whom baseline cytogenetic data were available, high-risk cytogenetic factors (presence of t(4:14), t(14:16), and/or del(17p)) were present in 26% of patients.
Efficacy results were based on overall response rate assessed by an Independent Review Committee using IMWG criteria.
The efficacy results from 117 patients treated with Talvey 0.4 mg/kg weekly who were not exposed to prior T cell redirection therapy and who had received at least 4 prior therapies are presented in Table 12.
 The efficacy results from 113 patients treated with Talvey 0.8 mg/kg weekly who were not exposed to prior T cell redirection therapy and who had received at least 4 prior therapies are presented in Table 13.
The efficacy results from 113 patients treated with Talvey 0.8 mg/kg weekly who were not exposed to prior T cell redirection therapy and who had received at least 4 prior therapies are presented in Table 13.
 Overall response rate (ORR) results were consistent across pre-specified subgroups, including number of prior lines of therapy, refractoriness to prior therapy, and cytogenetic risk at baseline.
Overall response rate (ORR) results were consistent across pre-specified subgroups, including number of prior lines of therapy, refractoriness to prior therapy, and cytogenetic risk at baseline.
At approximately week 29, there were 54 patients who completed the EORTC QLQ-C30 (European Organization for Research and Treatment of Cancer Quality of Life Questionnaire Core-30 Item) for the 0.4 mg/kg weekly group and 60 patients in the 0.8 mg/kg biweekly (every 2 weeks) group. Patients reported meaningful improvement from baseline in global health status, increased physical functioning and ability to participate in social roles and activities, decreased fatigue, and meaningful reductions in pain with 0.4 mg/kg weekly of Talvey. With 0.8 mg/kg biweekly of Talvey patients reported improvements in global health status, physical functioning, fatigue and pain and preserved ability to participate in social roles and activities.
MonumenTAL-1 also included 51 patients who were exposed to prior T cell redirection therapy and who had previously received at least three prior therapies, including a proteasome inhibitor, an immunomodulatory agent, and an anti-CD38 monoclonal antibody. Patients received Talvey 0.4 mg/kg subcutaneously once a week, following 2 step-up doses (0.01 and 0.06 mg/kg), or 0.8 mg/kg Q2W, following 3 step-up doses (0.01, 0.06, and 0.3 mg/kg), until disease progression or unacceptable toxicity. The median age was 61 (range: 38 to 78) years, 61% were male, 92% were White, and 6% were Black or African-American. Patients had received a median of 6 (range: 3 to 15) prior therapies. Prior T cell redirection therapy was CAR-T cell therapy for 75% of patients and bispecific antibody treatment for 31%. With a median duration of follow-up of 15.3 months, ORR per IRC assessment was 65%.
LocoMMotion and MoMMent analysis.
A patient-level, pooled analysis of outcomes of patients from the LocoMMotion and MoMMent studies (N=130) with relapsed or refractory multiple myeloma was conducted to provide contemporaneous context for interpreting the efficacy results reported in MonumenTAL-1. The analysis included patients who had received at least 4 prior therapies, including a proteasome inhibitor, an immunomodulatory agent, and an anti-CD38 monoclonal antibody. Patients received available treatments, and response and survival were evaluated. In a weighted analysis against patients with at least 4 prior therapies receiving talquetamab 0.4 mg/kg weekly, the ORR in LocoMMotion and MoMMent patients receiving available therapy was 28.2%, the median duration of response (DOR) was 7.7 months, the median progression-free survival (PFS) was 4.1 months, and the median overall survival (OS) was 9.2 months. In a similar weighted analysis against patients with at least 4 prior therapies receiving talquetamab 0.8 mg/kg, the ORR in LocoMMotion and MoMMent patients receiving available therapy was 29.8%, the median DOR was 11.1 months, the median PFS was 4.4 months, and the median OS was 10.2 months.
The adjusted comparative analysis of outcomes from LocoMMotion and MoMMent indicated that patients receiving talquetamab 0.4 mg/kg weekly or 0.8 mg/kg biweekly (every 2 weeks) in MonumenTAL-1 had better outcomes than patients receiving available treatments, as measured by ORR, DOR, PFS, and OS.
5.2 Pharmacokinetic Properties
0.4 mg/kg weekly.
Talquetamab exhibited approximately dose-proportional pharmacokinetics following subcutaneous administration across a dose ranging from 0.005 to 0.8 mg/kg weekly (0.0125 to 2 times the recommended 0.4 mg/kg weekly dose). The mean accumulation ratio between the 1st and 7th weekly dose of talquetamab 0.4 mg/kg was 3.9 and 4.5-fold for Cmax and AUCtau, respectively.
Pharmacokinetic parameters of talquetamab following the 1st and 7th recommended weekly dose of 0.4 mg/kg are shown in Table 14.

0.8 mg/kg biweekly (every two weeks).
Talquetamab exhibited approximately dose-proportional pharmacokinetics following subcutaneous administration across a dose ranging from 0.8 mg/kg to 1.2 mg/kg biweekly (1.0 to 1.5 times the recommended 0.8 mg/kg biweekly dose). The mean accumulation ratio between the 1st and 5th biweekly dose of talquetamab 0.8 mg/kg was 2.3- and 2.2-fold for Cmax and AUCtau, respectively.
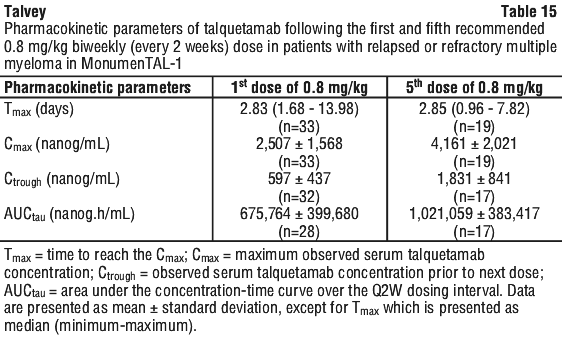
Pharmacokinetic parameters of talquetamab following the 1st and 5th recommended biweekly (every 2 weeks) dose of 0.8 mg/kg are shown in Table 15.
Absorption.
Based on the population pharmacokinetic model, the typical value of the bioavailability of talquetamab was 62% when administered subcutaneously relative to intravenous dosing.
At 0.4 mg/kg weekly dose regimen, the median (range) Tmax of talquetamab after the 1st and 7th treatment doses were 3 (1 to 8) days and 2 (1 to 6) days, respectively.
At 0.8 mg/kg biweekly (every 2 weeks) dose regimen, the median (range) Tmax of talquetamab after the 1st and 5th treatment doses were 3 (2 to 14) days and 3 (1 to 8) days, respectively.
Distribution.
Based on the population pharmacokinetic model, the typical value of the volume of distribution was 4.3 L (22% CV [coefficient of variation]) for the central compartment, and 5.8 L (83% CV) for the peripheral compartment.
Excretion.
Talquetamab exhibited both linear time-independent and time-dependent clearance. Based on the population pharmacokinetic model, the typical total clearance is 2.08 L/day at initial treatment and 1.06 L/day at steady state for participants with IgG subtype of myeloma and ISS stage I. The time-dependent clearance accounted for 48.8% of total clearance at initial treatment and then decreased exponentially to < 5% at around Week 16. The concentration-time profile at Week 16 would reach 90% of steady-state concentration for both 0.4 mg/kg weekly and 0.8 mg/kg biweekly regimens. The median terminal phase half-life based on the post hoc parameters of all SC population (N=392) was 7.56 days at initial treatment, and 12.2 days at steady state.
Special populations.
Renal impairment.
No formal studies of talquetamab in patients with renal impairment have been conducted.
Results of population pharmacokinetic analyses indicate that mild (60 mL/min/1.73 m2 ≤ estimated glomerular filtration rate (eGFR) < 90 mL/min/1.73 m2) or moderate (30 mL/min/1.73 m2 ≤ eGFR < 60 mL/min/1.73 m2) renal impairment did not significantly influence the pharmacokinetics of talquetamab. No data is available in patients with severe renal impairment.
Hepatic impairment.
No formal studies of talquetamab in patients with hepatic impairment have been conducted. Results of population pharmacokinetic analyses indicate that mild hepatic impairment (total bilirubin > 1 to 1.5 times upper limit of normal [ULN] and any aspartate aminotransferase [AST], or total bilirubin ≤ ULN and AST > ULN) and moderate hepatic impairment (total bilirubin 1.5 to 3 times ULN and any AST > ULN) did not significantly influence the pharmacokinetics of talquetamab. No data is available in patients with severe hepatic impairment.
Paediatrics (17 years of age and younger).
The pharmacokinetics of Talvey in paediatric patients aged 17 years and younger have not been investigated.
Elderly (65 years of age and older).
Results of population pharmacokinetic analyses indicate that age (33 to 86 years) did not influence the pharmacokinetics of talquetamab.
5.3 Preclinical Safety Data
Genotoxicity.
No genotoxicity studies have been performed to assess the genotoxic potential of talquetamab.
Carcinogenicity.
No carcinogenicity studies have been performed to assess the carcinogenic potential of talquetamab.6 Pharmaceutical Particulars
6.1 List of Excipients
Disodium edetate (EDTA), glacial acetic acid, polysorbate 20, sodium acetate trihydrate, sucrose, water for injections.
6.2 Incompatibilities
In the absence of compatibility studies, this medicinal product must not be mixed with other medicinal products.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
Prepared syringe.
The prepared syringes should be administered immediately. If immediate administration is not possible, store the Talvey solution for up to 24 hours refrigerated at 2°C to 8°C followed by up to 24 hours at ambient temperature of 15°C to 30°C. Discard if stored for more than 24 hours refrigerated or more than 24 hours of being at ambient temperature. If stored in the refrigerator, allow the solution to come to ambient temperature before administration.
6.4 Special Precautions for Storage
Store at 2°C to 8°C. Refrigerate, do not freeze.
Store in the original carton in order to protect from light.
6.5 Nature and Contents of Container
1.5 mL solution for injection in a Type 1 glass vial with an elastomeric stopper and a flip-off seal containing 3 mg of sterile talquetamab (2 mg/mL). Pack size of 1 vial.
1.0 mL solution for injection in a Type 1 glass vial with an elastomeric stopper and a flip-off seal containing 40 mg of sterile talquetamab (40 mg/mL). Pack size of 1 vial.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of in accordance with local requirements.
6.7 Physicochemical Properties
CAS number.
2226212-40-2.7 Medicine Schedule (Poisons Standard)
S4 - Prescription Only Medicine.
Summary Table of Changes


 Instruct patients to remain within proximity of a healthcare facility and monitor patients for 48 hours after administration of all doses within the Talvey step-up phase for signs and symptoms of cytokine release syndrome (CRS) and immune effector cell-associated neurotoxicity syndrome (ICANS). (See Section 4.4 Special Warnings and Precautions for Use, Cytokine release syndrome (CRS), Neurologic toxicities, including immune effector cell-associated neurotoxicity syndrome (ICANS)).
Instruct patients to remain within proximity of a healthcare facility and monitor patients for 48 hours after administration of all doses within the Talvey step-up phase for signs and symptoms of cytokine release syndrome (CRS) and immune effector cell-associated neurotoxicity syndrome (ICANS). (See Section 4.4 Special Warnings and Precautions for Use, Cytokine release syndrome (CRS), Neurologic toxicities, including immune effector cell-associated neurotoxicity syndrome (ICANS)).




 See Table 8 to determine total dose, injection volume, and number of vials required based on patient's actual body weight for the 0.06 mg/kg dose using Talvey 2 mg/mL vial.
See Table 8 to determine total dose, injection volume, and number of vials required based on patient's actual body weight for the 0.06 mg/kg dose using Talvey 2 mg/mL vial. See Table 9 to determine total dose, injection volume, and number of vials required based on patient's actual body weight for the 0.4 mg/kg dose using Talvey 40 mg/mL vial.
See Table 9 to determine total dose, injection volume, and number of vials required based on patient's actual body weight for the 0.4 mg/kg dose using Talvey 40 mg/mL vial. See Table 10 to determine total dose, injection volume, and number of vials required based on patient's actual body weight for the 0.8 mg/kg dose using Talvey 40 mg/mL vial.
See Table 10 to determine total dose, injection volume, and number of vials required based on patient's actual body weight for the 0.8 mg/kg dose using Talvey 40 mg/mL vial. Visually inspect the Talvey solution for injection is colourless to light yellow. Do not use if the solution is discoloured, cloudy, or if foreign particles are present.
Visually inspect the Talvey solution for injection is colourless to light yellow. Do not use if the solution is discoloured, cloudy, or if foreign particles are present.
 The efficacy results from 113 patients treated with Talvey 0.8 mg/kg weekly who were not exposed to prior T cell redirection therapy and who had received at least 4 prior therapies are presented in Table 13.
The efficacy results from 113 patients treated with Talvey 0.8 mg/kg weekly who were not exposed to prior T cell redirection therapy and who had received at least 4 prior therapies are presented in Table 13. Overall response rate (ORR) results were consistent across pre-specified subgroups, including number of prior lines of therapy, refractoriness to prior therapy, and cytogenetic risk at baseline.
Overall response rate (ORR) results were consistent across pre-specified subgroups, including number of prior lines of therapy, refractoriness to prior therapy, and cytogenetic risk at baseline.

