1 Name of Medicine
Talazoparib (as tosilate).
2 Qualitative and Quantitative Composition
Talzenna 0.1 mg strength.
Each capsule contains 0.145 mg talazoparib tosilate equivalent to 0.1 mg talazoparib free base.
Talzenna 0.25 mg strength.
Each capsule contains 0.363 mg talazoparib tosilate equivalent to 0.25 mg talazoparib free base.
Talzenna 0.35 mg strength.
Each capsule contains 0.509 mg talazoparib tosilate equivalent to 0.35 mg talazoparib free base.
Talzenna 0.5 mg strength.
Each capsule contains 0.727 mg talazoparib tosilate equivalent to 0.5 mg talazoparib free base.
Talzenna 1 mg strength.
Each capsule contains 1.453 mg talazoparib tosilate equivalent to 1 mg talazoparib free base.
For the full list of excipients, see Section 6.1 List of Excipients.3 Pharmaceutical Form
Hard capsule.
Talzenna 0.1 mg strength.
Opaque, size #4 hard hypromellose capsule with a white cap (printed with "Pfizer" in black) and a white body (printed with "TLZ 0.1" in black).
Talzenna 0.25 mg strength.
Opaque, size #4 hard hypromellose capsule with an ivory cap (printed with "Pfizer" in black) and a white body (printed with "TLZ 0.25" in black).
Talzenna 0.35 mg strength.
Opaque, size #4 hard hypromellose capsule with an ivory cap (printed with "Pfizer" in black) and an ivory body (printed with "TLZ 0.35" in black).
Talzenna 0.5 mg strength.
Opaque, size #4 hard hypromellose capsule with a light pink cap (printed with "Pfizer" in black) and a white body (printed with "TLZ 0.5" in black).
Talzenna 1 mg strength.
Opaque, size #4 hard hypromellose capsule with a light red cap (printed with "Pfizer" in black) and a white body (printed with "TLZ 1" in black).4.1 Therapeutic Indications
Breast cancer.
Talzenna is indicated for the treatment of patients with a deleterious or suspected deleterious germline breast cancer susceptibility gene (BRCA) mutation according to a validated diagnostic test, who have human epidermal growth factor receptor 2 (HER2)-negative, locally advanced or metastatic breast cancer.
Prostate cancer.
Talzenna is indicated in combination with enzalutamide for the treatment of adult patients with homologous recombination repair (HRR) gene-mutated metastatic castration-resistant prostate cancer (mCRPC).4.2 Dose and Method of Administration
Treatment with Talzenna should be initiated and supervised by a physician experienced in the use of anticancer medicinal products.
Patient selection.
Breast cancer - detection of BRCA mutation.
Detection of mutations in hereditary breast cancer-related BRCA1 and BRCA2 genes should be determined by an experienced laboratory using a validated test method (see Section 4.4 Special Warnings and Precautions for Use, Diagnostic test selection).
Prostate cancer.
Detection of mutations in genes involved in HRR should be determined by an experienced laboratory using a validated test method (see Section 4.4 Special Warnings and Precautions for Use, Diagnostic test selection).
Recommended dosing for breast cancer.
The recommended dose of Talzenna is 1 mg taken orally once daily.
The 0.25 mg and 0.5 mg strength capsules are available for dose reduction.
Patients should be treated until disease progression or unacceptable toxicity occurs.
Recommended dosing for prostate cancer.
The recommended dose of Talzenna is 0.5 mg administered orally once daily in combination with enzalutamide 160 mg orally once daily.
Patients receiving Talzenna and enzalutamide should also receive a luteinising hormone releasing hormone (LHRH) analogue concurrently or should have had bilateral orchiectomy.
The 0.1 mg, 0.25 mg and 0.35 mg strength capsules of Talzenna are available for dose reduction.
Refer to the enzalutamide Product Information for recommended enzalutamide dosing information.
Patients should be treated until disease progression or unacceptable toxicity occurs.
Administration.
Talzenna may be taken with or without food. The capsules should be swallowed whole and must not be opened or dissolved.
Missed dose.
If the patient vomits or misses a dose, an additional dose should not be taken. The next prescribed dose should be taken at the usual time.
Dosage adjustment.
To manage adverse reactions, consider interruption of treatment or dose reduction based on severity and clinical presentation. Recommended dose reductions are indicated in Table 1, Table 2 and Table 3. Talzenna should be discontinued if more than three dose reductions are required.
Full blood counts should be obtained prior to starting Talzenna therapy and monitored monthly and as clinically indicated (see Table 1; see Section 4.4 Special Warnings and Precautions for Use).


 Refer to the enzalutamide Product Information for dose modifications for adverse reactions associated with enzalutamide.
Refer to the enzalutamide Product Information for dose modifications for adverse reactions associated with enzalutamide.
Concomitant treatment with inhibitors of P-glycoprotein (P-gp). Talzenna monotherapy (breast cancer).
Strong inhibitors of P-gp may lead to increased talazoparib exposure. Concomitant use of strong P-gp inhibitors during treatment with talazoparib should be avoided. If coadministration with a strong P-gp inhibitor is unavoidable, the Talzenna dose should be reduced to the next lower dose level (see Table 2). When the strong P-gp inhibitor is discontinued, the Talzenna dose should be increased (after 3 to 5 half-lives of the P-gp inhibitor) to the dose used prior to the initiation of the strong P-gp inhibitor (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions, P-gp inhibitors).
Talzenna when used in combination with enzalutamide (prostate cancer).
The effect of co-administration of P-gp inhibitors on talazoparib exposure when talazoparib is given in combination with enzalutamide has not been studied. Therefore, concomitant use of P-gp inhibitors during treatment with talazoparib should be avoided when possible (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions, P-gp inhibitors).
Hepatic impairment. No dose adjustment is required for patients with mild hepatic impairment (total bilirubin ≤ 1 x upper limit of normal [ULN] and aspartate aminotransferase (AST) > ULN, or total bilirubin > 1.0 to 1.5 x ULN and any AST), moderate hepatic impairment (total bilirubin > 1.5 to 3.0 x ULN and any AST) or severe hepatic impairment (total bilirubin > 3.0 x ULN and any AST) (see Section 5.2 Pharmacokinetic Properties, Hepatic impairment).
Renal impairment. Breast cancer.
No dose adjustment is required for patients with mild renal impairment (60 mL/min ≤ creatinine clearance [CrCl] < 90 mL/min). For patients with moderate renal impairment (30 mL/min ≤ CrCl < 60 mL/min), the recommended dose of Talzenna is 0.75 mg once daily. For patients with severe renal impairment (15 mL/min ≤ CrCl < 30 mL/min), the recommended dose of Talzenna is 0.5 mg once daily. Talzenna has not been studied in patients requiring haemodialysis (see Section 5.2 Pharmacokinetic Properties, Renal impairment).
Prostate cancer.
No dose adjustment is required for patients with mild renal impairment (60 mL/min ≤ CrCl < 90 mL/min). For patients with moderate renal impairment (30 mL/min ≤ CrCl < 60 mL/min), the recommended dose of Talzenna is 0.35 mg once daily in combination with enzalutamide orally once daily. For patients with severe renal impairment (15 mL/min ≤ CrCl < 30 mL/min), the recommended dose of Talzenna is 0.25 mg once daily in combination with enzalutamide orally once daily. Talzenna has not been studied in patients with CrCl < 15 mL/min or patients requiring haemodialysis (see Section 5.2 Pharmacokinetic Properties, Renal impairment).
Elderly population. No dose adjustment is necessary in elderly (≥ 65 years of age) patients (see Section 5.2 Pharmacokinetic Properties, Elderly population).4.3 Contraindications
Use of Talzenna is contraindicated in patients with hypersensitivity to talazoparib tosilate or any of the excipients listed in Section 6.1 List of Excipients.
4.4 Special Warnings and Precautions for Use
Myelodysplastic syndrome/acute myeloid leukaemia.
Myelodysplastic syndrome/acute myeloid leukaemia (MDS/AML) have been reported in patients who received talazoparib. Overall, MDS/AML has been reported in < 1% of solid tumour patients treated with talazoparib in clinical studies. In TALAPRO-2, MDS/AML occurred in 2 out of 511 (0.4%) patients treated with Talzenna and enzalutamide and in 0 out of 517 (0%) patients treated with placebo and enzalutamide. Potential contributing factors for the development of MDS/AML include previous platinum-containing chemotherapy, other DNA damaging agents or radiotherapy.
Do not start Talzenna until patients have adequately recovered from haematological toxicity caused by previous chemotherapy. Monitor full blood counts for cytopenia at baseline and monthly thereafter. For prolonged haematological toxicities, interrupt Talzenna and monitor blood counts weekly until recovery. If the levels have not recovered after 4 weeks, refer the patient to a haematologist for further investigations, including bone marrow analysis and blood sample for cytogenetics. If MDS/AML is confirmed, discontinue talazoparib.
Myelosuppression.
Myelosuppression, consisting of anaemia, leucopenia/neutropenia and/or thrombocytopenia, is very common in patients treated with talazoparib (see Section 4.8 Adverse Effects (Undesirable Effects)).
In TALAPRO-2, Grade ≥ 3 anaemia, neutropenia, and thrombocytopenia were reported, respectively, in 45%, 18%, and 8% of patients receiving Talzenna and enzalutamide. Overall, 39% of patients (199/511) required a red blood cell transfusion, including 22% (111/511) who required multiple transfusions. Discontinuation due to anaemia, neutropenia, and thrombocytopenia occurred, respectively, in 7%, 3%, and 0.4% of patients.
Do not start talazoparib until patients have recovered from haematological toxicity caused by previous therapy (≤ Grade 1). Monitor clinically and check full blood counts for cytopenia at baseline and monthly thereafter. If haematological toxicity occurs, dose modification (interruption with or without reduction) is recommended (see Section 4.2 Dose and Method of Administration, Dosage adjustment). Provide supportive care, transfusion of blood/platelets and treatment with colony stimulating factors as appropriate.
Embryo-fetal toxicity.
Based on its mechanism of action and findings from animal data, Talzenna can cause fetal harm when administered to a pregnant woman. In an animal reproduction study, administration of talazoparib to pregnant rats during the period of organogenesis caused fetal malformations and structural skeletal variations, and embryo-fetal death at exposures that were 0.24 times the total area under the concentration-time curve (AUC) in patients receiving the recommended human dose of 1 mg daily (see Section 5.3 Preclinical Safety Data, Reproductive toxicology).
Conduct a blood test for pregnancy prior to initiating Talzenna treatment in any female of reproductive potential. Advise pregnant women and females of reproductive potential of the potential risk to a fetus. Advise females of reproductive potential to use effective contraception during treatment and for at least 7 months following the last dose of Talzenna (see Section 4.6 Fertility, Pregnancy and Lactation, Use in pregnancy).
Based on findings from genetic toxicity and animal reproduction studies, advise male patients with female partners of reproductive potential and pregnant partners to use effective contraception (even after vasectomy), during treatment with Talzenna and for at least 4 months after the final dose (see Section 4.6 Fertility, Pregnancy and Lactation, Use in pregnancy; Section 5.3 Preclinical Safety Data).
Use in the elderly.
No overall differences in safety or effectiveness of Talzenna were observed between patients ≥ 65 years of age and younger patients, but greater sensitivity of some older individuals cannot be ruled out (see Section 5.2 Pharmacokinetic Properties, Elderly population). No dose adjustment is required in elderly patients (see Section 4.2 Dose and Method of Administration, Dosage adjustment, Elderly population).
Paediatric use.
The safety and efficacy of Talzenna in children and adolescents < 18 years of age have not been established.
Effects on laboratory tests.
See Section 4.8 Adverse Effects (Undesirable Effects).
Diagnostic test selection.
When assessing a patient for mutations in hereditary breast cancer-related BRCA1 and BRCA2 genes or other genes involved in HRR, it is important that a well-validated and robust test is chosen to minimise false negative or false positive test results.4.5 Interactions with Other Medicines and Other Forms of Interactions
Talazoparib is a substrate for drug transporters P-gp and breast cancer resistance protein (BCRP) and is mainly eliminated by renal clearance as unchanged compound. Information on agents that may affect talazoparib plasma concentrations is provided below.
Effect of enzalutamide.
Coadministration with enzalutamide increases talazoparib exposure approximately 2-fold. Administration of talazoparib 0.5 mg daily in combination with enzalutamide achieves approximately the same steady-state trough (Ctrough) concentration reported for talazoparib 1 mg daily (see Section 5.2 Pharmacokinetic Properties). When Talzenna is coadministered with enzalutamide, the Talzenna starting dose is 0.5 mg (see Section 4.2 Dose and Method of Administration, Recommended dosing for prostate cancer).
P-gp inhibitors.
Coadministration with P-gp inhibitors may increase talazoparib exposure.
Data from a drug-drug interaction study in patients with advanced solid tumours indicated that coadministration of multiple daily doses of a P-gp inhibitor, itraconazole 100 mg twice daily, with a single 0.5 mg talazoparib dose increased talazoparib total exposure (AUCinf) and peak concentration (Cmax) by approximately 56% and 40%, respectively, relative to a single 0.5 mg talazoparib dose administered alone. A population pharmacokinetic (PK) analysis has also shown that concomitant use of strong P-gp inhibitors with Talzenna increased talazoparib exposure by 44.7% relative to Talzenna given alone.
If coadministration with a strong P-gp inhibitor, those that result in ≥ 2-fold increase in the exposure of an in vivo probe P-gp substrate, (including but not limited to amiodarone, carvedilol, clarithromycin, cobicistat, darunavir, dronedarone, erythromycin, indinavir, itraconazole, ketoconazole, lapatinib, lopinavir, propafenone, quinidine, ranolazine, ritonavir, saquinavir, telaprevir, tipranavir, valspodar and verapamil) is unavoidable, the Talzenna dose should be reduced (see Section 4.2 Dose and Method of Administration, Dosage adjustment, Concomitant treatment with inhibitors of P-glycoprotein (P-gp)).
A population PK analysis has shown that coadministration with relatively weak P-gp inhibitors (including azithromycin, atorvastatin, diltiazem, felodipine, fluvoxamine and quercetin) in clinical studies had no significant effect on talazoparib exposure.
The effect of co-administration of P-gp inhibitors on talazoparib exposure when talazoparib is given in combination with enzalutamide has not been studied. If co-administration of P-gp inhibitors cannot be avoided, when Talzenna is given with enzalutamide, the patient should be monitored for potential increased adverse reactions.
P-gp inducers.
Data from a drug-drug interaction study in patients with advanced solid tumours indicated that coadministration of multiple daily doses of a P-gp inducer, rifampicin 600 mg, with a single 1 mg talazoparib dose increased talazoparib Cmax by approximately 37% with no effect on talazoparib total exposure. No talazoparib dose adjustments are required with P-gp inducers.
Breast cancer resistance protein (BCRP) inhibitors.
The effect of BCRP inhibitors on the PK of talazoparib has not been studied. Coadministration with BCRP inhibitors may increase talazoparib exposure. Concomitant use of strong BCRP inhibitors (including but not limited to curcumin, ciclosporin and elacridar [GF120918]) should be avoided. If coadministration cannot be avoided, monitor patients for potential adverse reactions when coadministering.
Acid-reducing agents.
A population PK analysis indicates that coadministration of acid-reducing agents including proton pump inhibitors (PPI), histamine receptor 2 antagonists (H2RA) or other acid-reducing agents had no significant impact on the absorption of talazoparib.
Administration with CYP substrates.
In vitro, talazoparib was not an inhibitor of cytochrome (CYP)1A2, CYP2B6, CYP2C8, CYP2C9, CYP2C19, CYP2D6 or CYP3A4/5 or an inducer of CYP1A2, CYP2B6 or CYP3A4 at clinically relevant concentrations.
Administration with substrates of transporters.
In vitro, talazoparib did not inhibit any of the major intestinal, hepatic or renal membrane transporters (P-gp, BCRP, organic anion transporting polypeptide [OATP]1B1, OATP1B3, organic cationic transporter [OCT]1, OCT2, organic anion transporter [OAT]1, OAT3, bile salt export pump [BSEP], multidrug and toxin extrusion [MATE]1 and MATE2-K) at clinically relevant concentrations.
Administration with UGT substrates.
In vitro, talazoparib did not inhibit any of the major uridine-diphosphate glucuronosyltransferase (UGT) isoforms (1A1, 1A4, 1A6, 1A9, 2B7 and 2B15) at clinically relevant concentrations.4.6 Fertility, Pregnancy and Lactation
Effects on fertility.
There is no information on fertility in humans. In repeat-dose toxicity studies up to 3 months duration, talazoparib-related findings in the testis and epididymis at doses ≥ 0.04 mg/kg/day in rats and ≥ 0.01 mg/kg/day in dogs included decreased organ weights, luminal cellular debris, reduced sperm and degeneration/atrophy. These doses in rats and dogs resulted in exposures approximately 0.4 times and 0.3 times, respectively, the exposure (AUC) in humans at the recommended dose. Follicular atresia of the ovary was observed in rats at doses ≥ 1 mg/kg/day talazoparib, approximately 5 times the AUC in patients at the recommended dose. Based on non-clinical findings in testes and ovary, male and female fertility may be compromised by treatment with Talzenna.
(Category D)
There are no data from the use of Talzenna in pregnant women. Based on findings from animal studies and its mechanism of action, Talzenna can cause embryo-fetal harm when administered to a pregnant woman.
In an embryo-fetal development toxicity study, pregnant rats received oral doses of 0.015, 0.05 and 0.15 mg/kg/day talazoparib during the period of organogenesis. Talazoparib caused embryo-fetal death at doses ≥ 0.015 mg/kg/day (approximately 0.24 times the total AUC in patients at the recommended dose). A dose of 0.015 mg/kg/day caused decreased fetal body weights and an increased incidence of fetal malformations (depressed eye bulge, small eye, split sternebra and fused cervical vertebral arch) and structural variations including misshapen or incomplete ossification of the sternebra, skull, rib and vertebra.
Talzenna is not recommended during pregnancy or for women of childbearing potential not using contraception (see Section 4.4 Special Warnings and Precautions for Use, Embyro-fetal toxicity).
There are no data on the presence of talazoparib in human milk, the effects of the drug on milk production or the effects of the drug on the breastfed child. Because of the potential for serious adverse reactions in a breastfed child from talazoparib, advise lactating women not to breastfeed during treatment with Talzenna and for at least 1 month after the final dose.4.7 Effects on Ability to Drive and Use Machines
No studies have been conducted on the effects of talazoparib on the ability to drive or operate machinery. However, patients experiencing fatigue/asthenia or dizziness while taking Talzenna should exercise caution when driving or operating machinery.
4.8 Adverse Effects (Undesirable Effects)
Adverse drug reactions are listed by System organ class (SOC). Frequency categories are defined using the following conventions: Very common (≥ 1/10), common (≥ 1/100 to < 1/10), uncommon (≥ 1/1000 to < 1/100), rare (≥ 1/10,000 to < 1/1,000) and very rare (< 1/10,000).
Breast cancer randomised phase 3 study - EMBRACA.
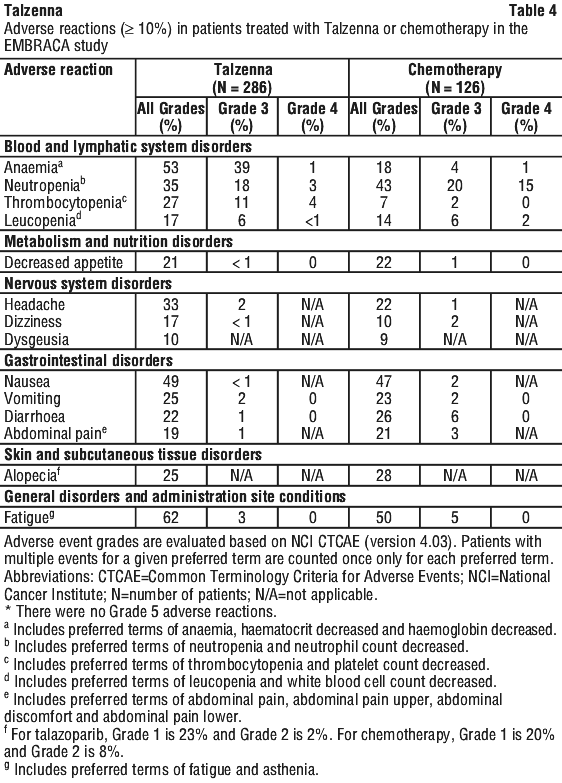
The safety of Talzenna as monotherapy was evaluated in patients with a germline BRCA mutation and HER2-negative locally advanced or metastatic breast cancer who had previously received no more than 3 lines of chemotherapy for the treatment of locally advanced/metastatic disease. EMBRACA (see Section 5.1 Pharmacodynamic Properties, Clinical trials) was a randomised, open-label, multi-centre study in which 412 patients received either Talzenna 1 mg once daily (n=286) or a chemotherapy agent (capecitabine [n=55], eribulin [n=50], gemcitabine [n=12] or vinorelbine [n=9]) of the healthcare provider's choice (n=126) until disease progression or unacceptable toxicity. The median duration of study treatment was 6.1 months in patients who received Talzenna and 3.9 months in patients who received chemotherapy. Dosing interruptions due to an adverse reaction of any grade occurred in 65% of patients receiving Talzenna and 50% of those receiving chemotherapy; dose reductions due to any cause occurred in 53% of Talzenna patients and 40% of chemotherapy patients. Permanent discontinuation due to adverse reactions occurred in 5% of Talzenna patients and 6% of chemotherapy patients.
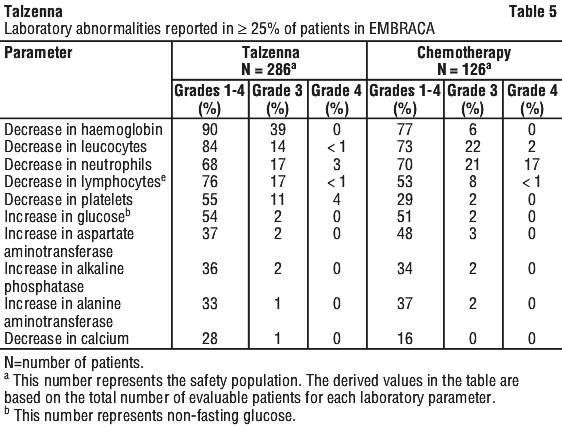
Table 4 and Table 5 summarise the most common adverse reactions and laboratory abnormalities, respectively, in patients treated with Talzenna (n=286) or chemotherapy (n=126) in the EMBRACA study.
 The following adverse reactions have been identified in < 10% of the 286 patients receiving Talzenna and are not included in Table 4:
The following adverse reactions have been identified in < 10% of the 286 patients receiving Talzenna and are not included in Table 4:
Blood and lymphatic system disorders.
Common: lymphopenia (7.3%).
Gastrointestinal disorders.
Common: dyspepsia (9.8%), stomatitis (8.4%).
Prostate cancer randomised phase 3 study - TALAPRO-2.
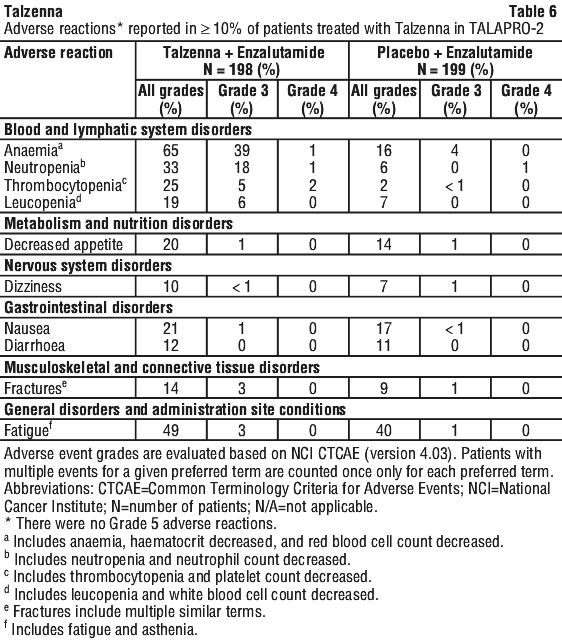
The safety of Talzenna in combination with enzalutamide was evaluated in patients with HRR gene-mutated mCRPC enrolled in TALAPRO-2. TALAPRO-2 (see Section 5.1 Pharmacodynamic Properties, Clinical trials) was a randomised, double-blind, placebo controlled trial in which patients received either Talzenna 0.5 mg in combination with enzalutamide 160 mg once daily (N=198) or placebo in combination with enzalutamide 160 mg once daily (N=199). The median duration of study treatment was 63.3 weeks in patients who received Talzenna and 52.1 weeks in patients who received placebo, in combination with enzalutamide.
Serious adverse reactions of Talzenna in combination with enzalutamide occurred in 30% of patients. Serious adverse reactions reported in > 2% of patients included anemia (9%) and fracture (3%). Fatal adverse reactions occurred in 1.5% of patients, including pneumonia, COVID infection, and sepsis (1 patient each).
The most common Grade 3 or higher adverse reaction was anaemia (41%). Median time to onset of Grade 3 or higher anaemia was 99 days in the Talzenna and enzalutamide arm, and 111 days in placebo and enzalutamide arm. Dosing interruptions due to adverse reactions occurred in 57.6% of patients receiving Talzenna in combination with enzalutamide; the most common was anaemia (41.4%). Dose reductions due to adverse reactions occurred in 52.0% of Talzenna patients; the most common was anaemia (42.9%). Permanent discontinuation of Talzenna due to adverse reactions occurred in 10.1% of patients; the most common was anaemia (4%).
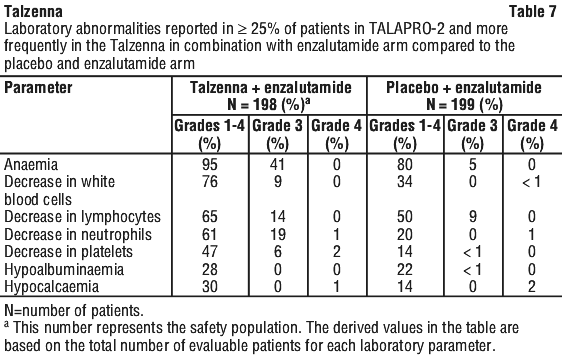
Table 6 and Table 7 summarise the most common adverse reactions and laboratory abnormalities, respectively, in the TALAPRO-2 study.
 The following adverse reactions have been identified in < 10% of the 198 patients receiving Talzenna in combination with enzalutamide, and thus were not included in Table 6:
The following adverse reactions have been identified in < 10% of the 198 patients receiving Talzenna in combination with enzalutamide, and thus were not included in Table 6:
Blood and lymphatic system disorders.
Common: lymphopenia (8%) (includes lymphopenia and lymphocyte count decreased).
Neoplasms benign, malignant and unspecified (including cysts and polyps).
Rare: MDS/AML < 1% (see Section 4.4 Special Warnings and Precautions for Use, Myelodysplastic syndrome/acute myeloid leukaemia).
Nervous system disorders.
Common: headache (6%), dysgeusia (9%).
Vascular disorders.
Common: venous thromboembolism (3%).
Gastrointestinal disorders.
Common: abdominal pain (9%), vomiting (8%), dyspepsia (3%), stomatitis (1%).
Skin and subcutaneous tissue disorders.
Common: alopecia (7%).
Reporting suspected adverse effects.
Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.4.9 Overdose
There is no specific treatment in the event of talazoparib overdose, and symptoms of overdose are not established. In the event of overdose, discontinue treatment with talazoparib, consider gastric decontamination, follow general supportive measures and treat symptomatically.
For information on the management of overdose, contact the Poison Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action.
Talazoparib is an inhibitor of poly (ADP-ribose) polymerase (PARP) enzymes, including PARP1 (IC50 = 0.7 nanoM) and PARP2 (IC50 = 0.3 nanoM), which play a role in DNA repair. In vitro studies with cancer cell lines that harboured defects in DNA repair genes, including BRCA 1 and 2, have shown that talazoparib-induced cytotoxicity may involve inhibition of PARP enzymatic activity and increased formation of PARP-DNA complexes resulting in DNA damage, decreased cell proliferation and apoptosis. Talazoparib anti-tumour activity was observed in human patient-derived xenograft breast cancer tumour models that expressed mutated or wild-type BRCA 1 and 2, as well as in an androgen receptor (AR) positive prostate cancer cell line xenograft model.
The combination of a PARP inhibitor and androgen receptor signalling inhibitor (ARSi) has been identified as a mechanism-based interaction that expands the functional state of sensitivity to broader inhibition of homologous recombination DNA repair mechanisms. AR signalling inhibition suppresses the expression of homologous recombination repair (HRR) genes including BRCA1, resulting in sensitivity to PARP inhibition. PARP1 activity has been shown to be required for maximal AR function and thus inhibiting PARP may reduce AR signalling and increase sensitivity to AR signalling inhibitors. Clinical resistance to AR blockade is sometimes associated with co-deletion of retinoblastoma RB1 and BRCA2, which is in turn associated with sensitivity to PARP inhibition.
Clinical trials.
Breast cancer randomised phase 3 study - EMBRACA.
EMBRACA was an open-label, randomised, multicentre study in which patients with a germline BRCA mutation who had HER2-negative locally advanced or metastatic breast cancer (n=431) were randomised 2:1 to receive Talzenna 1 mg or healthcare provider's choice of chemotherapy (capecitabine, eribulin, gemcitabine, vinorelbine) until disease progression or unacceptable toxicity. Randomisation was stratified by prior lines of chemotherapy for metastatic disease (0 versus 1, 2 or 3), triple-negative disease status (triple-negative breast cancer [TNBC] versus non-TNBC) and history of central nervous system (CNS) metastasis (yes versus no).
Patients had received no more than 3 prior cytotoxic chemotherapy regimens for their metastatic or locally advanced disease. Patients were required to have received treatment with an anthracycline and/or a taxane (unless contraindicated) in the neoadjuvant, adjuvant and/or metastatic setting. First-line treatment for advanced or metastatic disease with no prior adjuvant chemotherapy was allowed if the investigator determined that one of the chemotherapy choices in the control arm would be an appropriate treatment option for the patient. Patients with prior platinum therapy for advanced disease were required to have no evidence of disease progression during platinum therapy. No prior treatment with a PARP inhibitor was permitted.
Of the 431 patients randomised in the EMBRACA study, 408 (95%) were centrally confirmed to have a deleterious or suspected deleterious germline BRCA mutation using a clinical trial assay, out of which 354 (82%) were confirmed using the BRACAnalysis companion diagnostic test. A similar percentage of patients in both treatment arms had a BRCA1 versus BRCA2 mutation.
The median age of patients treated with Talzenna was 45 years (range 27 to 84) and 50 years (range 24 to 88) among patients treated with chemotherapy. Of note, 63% versus 47% of patients were < 50 years of age in the talazoparib and chemotherapy arms, respectively, 27% versus 47% were 50 to < 65 years of age, and 9% versus 7% were ≥ 65 years of age. Among all randomised patients, 1% versus 2% were males, 67% versus 75% were White, 11% versus 11% were Asian, and 4% versus 1% were Black or African American in the talazoparib and chemotherapy arms, respectively. Almost all patients (98%) in both arms had an Eastern Cooperative Oncology Group (ECOG) performance status of 0 or 1. Approximately 56% of patients had hormone receptor (HR)-positive (either estrogen receptor [ER]-positive- or progesterone receptor [PR]-positive) disease; 44% of patients had triple-negative breast cancer (TNBC) and the proportions were balanced across treatment arms. Fifteen percent (15%) of patients in the Talzenna arm and 14% of patients in the chemotherapy arm had a history of CNS metastases. Ninety-one percent (91%) of patients in the Talzenna arm had received prior taxane therapy and 85% had received prior anthracycline therapy in any setting. Sixteen percent of patients in the talazoparib arm and 21% of patients in the chemotherapy arm had received prior platinum treatment in any setting. The median number of prior cytotoxic regimens for patients with advanced breast cancer was one: 38% had received no prior cytotoxic regimens for advanced or metastatic disease, 37% had received one, 20% had received two and 5% had received three or more prior cytotoxic regimens.
The primary efficacy endpoint was progression-free survival (PFS) evaluated according to Response Evaluation Criteria in Solid Tumours (RECIST) version 1.1, as assessed by blinded independent central review (BICR). The secondary objectives were objective response rate (ORR), overall survival (OS), safety and PK. Exploratory objectives included duration of response (DOR).
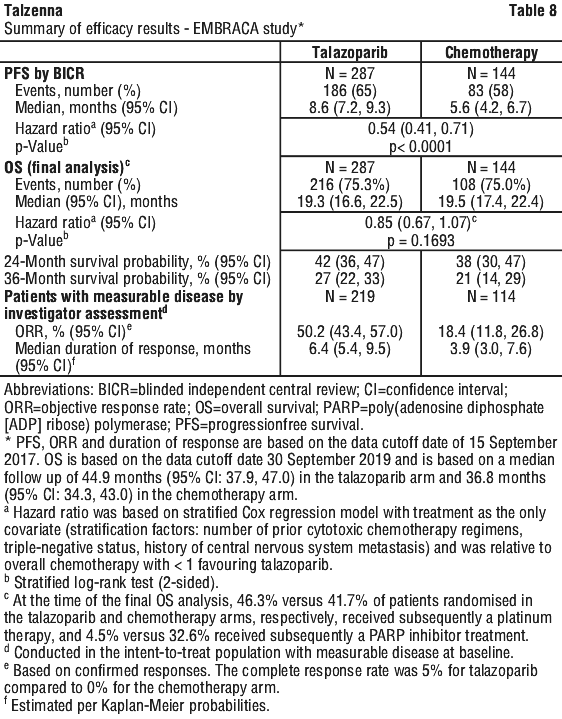
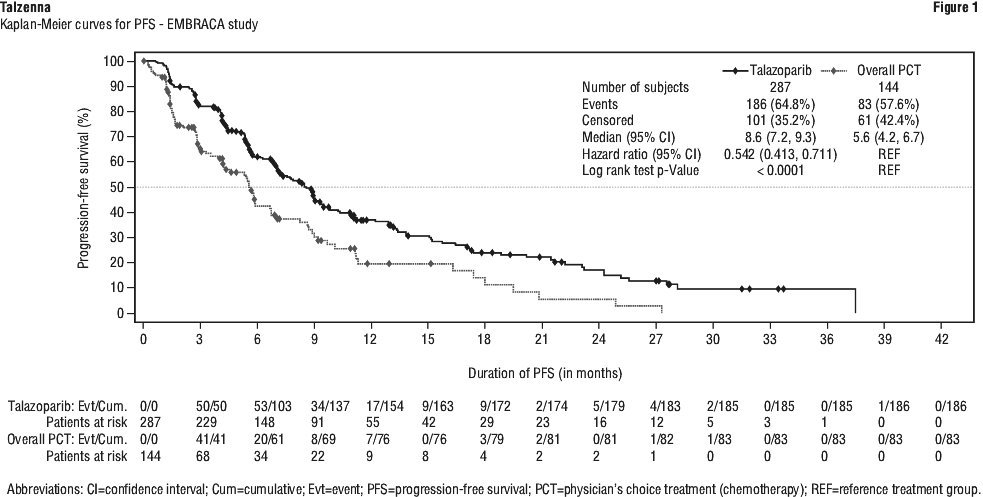
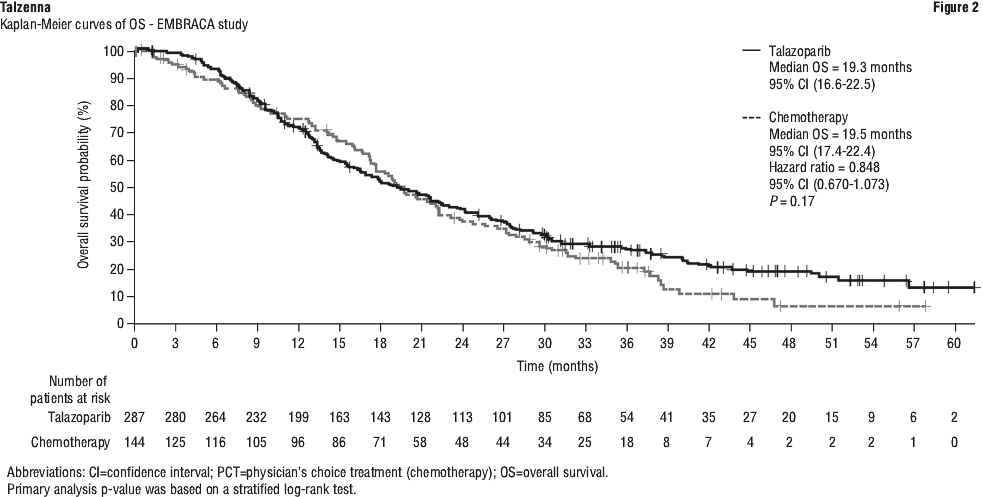
A statistically significant improvement in PFS was demonstrated for Talzenna compared with chemotherapy. A sensitivity analysis of investigator-assessed PFS was consistent with the BICR-assessed PFS results. Consistent PFS results were observed across patient subgroups defined by study stratification factors (line of therapy, TNBC status and history of CNS metastases). Efficacy data for EMBRACA are summarised in Table 8 and the Kaplan-Meier curves for PFS and OS are shown in Figures 1 and 2.
Prostate cancer randomised phase 3 study - TALAPRO-2.
The efficacy of Talzenna in combination with enzalutamide was evaluated in TALAPRO2, a randomised, double-blind, placebo controlled, multi-cohort study in which patients with mCRPC were randomised 1:1 to receive enzalutamide 160 mg daily plus either Talzenna 0.5 mg or placebo daily until unacceptable toxicity or progression. The study included an all-comers cohort (N=805) and a HRR gene-mutated (HRRm) cohort (N=399, of which 169 HRRm patients were from the all-comers cohort). All patients received a gonadotropin-releasing hormone (GnRH) analogue or had prior bilateral orchiectomy and needed to have progressed on prior androgen deprivation therapy. Prior treatment with abiraterone or taxane based chemotherapy for metastatic castration-sensitive prostate cancer (mCSPC) was permitted. Mutation status of HRR genes was determined prospectively using solid tumour tissue or circulating tumour DNA (ctDNA)-based next generation sequencing tests. Patients were required to have a mutation in at least one of 12 genes involved directly or indirectly in the HRR pathway (ATM, ATR, BRCA1, BRCA2, CDK12, CHEK2, FANCA, MLH1, MRE11A, NBN, PALB2, or RAD51C).
Randomisation was stratified by previous treatment with abiraterone or taxane-based chemotherapy versus no such prior treatment (both cohorts) and by tumour HRR gene mutation status (all comers cohort only).
The median age in the HRRm population was 70 years (range 41 to 90) in both arms; 68% were White, 21% were Asian, and 3% were Black. Most participants (62%) in both arms had an ECOG performance status of 0. In patients treated with Talzenna, the proportion of patients with RECIST 1.1 measurable disease at baseline per BICR was 36%. Thirty-seven percent (37%) of patients had received prior abiraterone or taxane-based chemotherapy. All patients (100%) had tumours with HRR gene mutations.
The primary efficacy outcome was radiographic progression-free survival (rPFS) evaluated according to RECIST version 1.1 and Prostate Cancer Clinical Trials Working Group Criteria 3 (PCWG3) (bone) criteria, as assessed by BICR. Key efficacy outcomes included OS and ORR assessed by BICR.
A statistically significant improvement in BICR-assessed rPFS was demonstrated for Talzenna in combination with enzalutamide compared to placebo in combination with enzalutamide for both all-comers cohort and HHRm gene-mutated cohort. The OS data were not mature at the time of the final rPFS analysis.
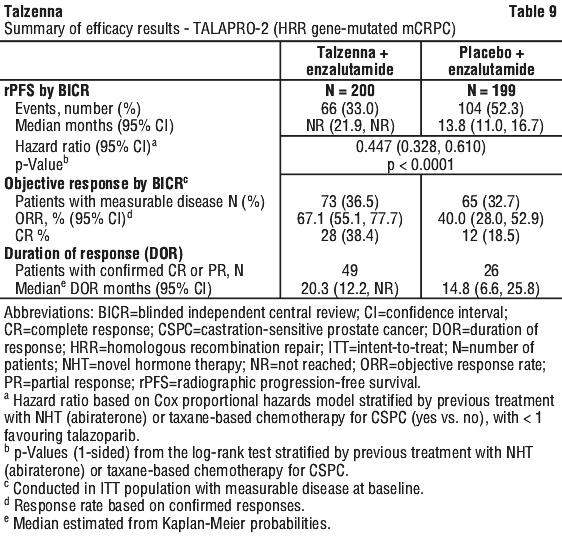
Efficacy results of TALAPRO-2 in HRR gene-mutated mCRPC are provided in Table 9 and Figure 3.
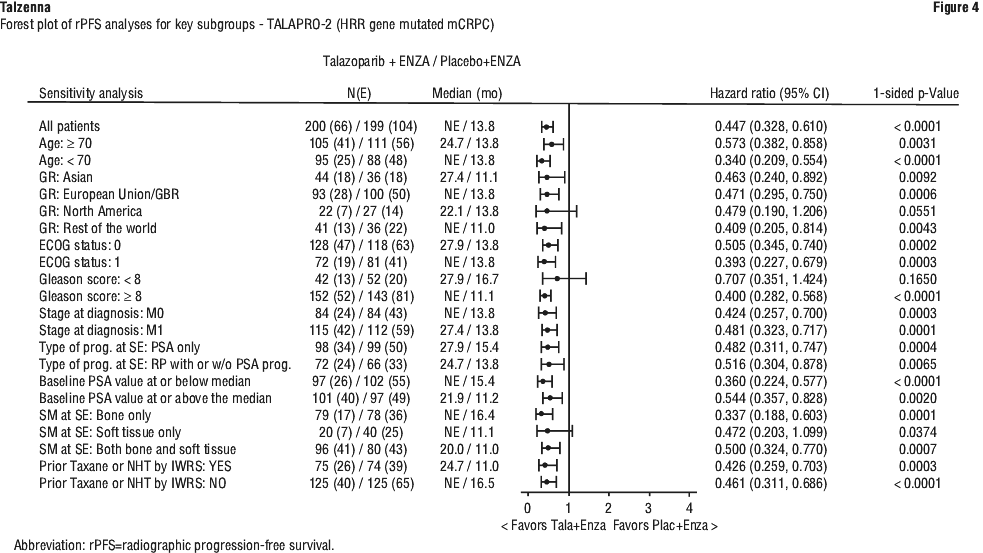
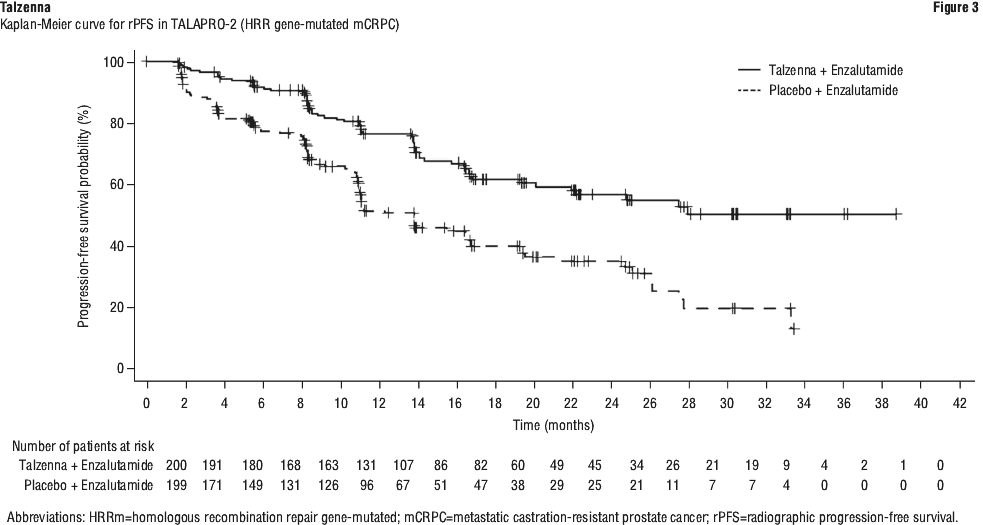 Prespecified subgroup rPFS analyses were performed based on prognostic factors and baseline characteristics to evaluate the internal consistency of the treatment effect. Consistent with the overall results, a reduction in the risk of disease progression or death in favour of talazoparib in combination with enzalutamide was observed in patient subgroups shown in Figure 4.
Prespecified subgroup rPFS analyses were performed based on prognostic factors and baseline characteristics to evaluate the internal consistency of the treatment effect. Consistent with the overall results, a reduction in the risk of disease progression or death in favour of talazoparib in combination with enzalutamide was observed in patient subgroups shown in Figure 4.
 Exploratory subgroup analyses of rPFS for patients with BRCA-mutated (BRCAm) and non-BRCAm HRRm are presented in Table 10.
Exploratory subgroup analyses of rPFS for patients with BRCA-mutated (BRCAm) and non-BRCAm HRRm are presented in Table 10.

5.2 Pharmacokinetic Properties
The pharmacokinetic profile of talazoparib is linear from 0.025 mg to 2 mg (double the 1 mg monotherapy recommended daily dose). After oral administration of 1 mg talazoparib monotherapy once daily in breast cancer patients, the geometric mean (% coefficient of variation [CV%]) area under the plasma concentration-time curve (AUC) and maximum observed plasma concentration (Cmax) of talazoparib at steady-state was 208 (37%) nanogram.hr/mL and 16.4 (32%) nanogram/mL, respectively.
After oral administration of 0.5 mg Talzenna once daily in combination with enzalutamide in mCRPC patients, the geometric mean (CV%) steady-state Ctrough across visits ranged from 3.29 to 3.68 nanogram/mL (45 to 48%), which is similar to the observed values of 3.53 (61%) nanogram/mL when Talzenna monotherapy was administered at 1 mg once daily in breast cancer patients.
The median accumulation ratio of talazoparib following repeated oral administration of 1 mg once daily was in the range of 2.3 to 5.2, and talazoparib plasma concentrations reached steady-state within 2 to 3 weeks when administered alone, and within 9 weeks when coadministered with enzalutamide.
Absorption.
Following oral administration of talazoparib, median time to Cmax (Tmax) was between 1 and 2 hours after dosing.
The effect of food.
Food intake decreased the rate but not the extent of talazoparib absorption. Following a single oral dose of talazoparib with high-fat, high-calorie food (approximately 827 calories, 57% fat), the mean Cmax of talazoparib was decreased by approximately 46%, the median Tmax was delayed from 1 to 4 hours, and AUCinf was not affected. Based on these results, Talzenna can be administered with or without food.
Distribution.
The population mean apparent volume of distribution (Vss/F) of talazoparib was 420 L. In vitro, talazoparib is approximately 74% bound to plasma proteins with no concentration dependence over the concentration range of 0.01 microM to 1 microM. Renal or hepatic impairment do not appear to impact talazoparib protein binding as there was no obvious trend in the mean talazoparib fraction of unbound drug (fu) in human plasma in vivo with worsening renal or hepatic function.
Metabolism.
Talazoparib undergoes minimal hepatic metabolism in humans. Following oral administration of a single 1 mg dose of [14C]talazoparib, no major circulating metabolites were identified in plasma and talazoparib was the only circulating drug-derived entity identified. No metabolites that individually represented more than 10% of the administered dose were recovered in the urine or faeces. The identified metabolic pathways of talazoparib in humans include: 1) mono-oxidation; 2) dehydrogenation; 3) cysteine conjugation of mono-desfluoro-talazoparib; and 4) glucuronide conjugation.
Excretion.
The mean terminal plasma half-life of talazoparib was 89.8 hours and the population mean apparent oral clearance (CL/F) was 6.45 L/h in cancer patients. Excretion of talazoparib in urine was the major route of elimination (69% of the administered dose, 55% unchanged), and 20% was recovered in the faeces (14% unchanged).
Special populations.
Age, sex, race and body weight. A population PK analysis was conducted using data from 490 patients with cancer who received talazoparib 1 mg daily as monotherapy to evaluate the impact of age (ranging from 18 to 88 years), sex (53 males and 437 females), race (361 White, 41 Asian, 16 Black, 9 Others and 63 Not reported) and body weight (ranging from 35.7 kg to 162 kg) on the PK of talazoparib. The results indicate that age, sex, race and body weight have no clinically relevant effect on the PK of talazoparib.
Paediatric population. Pharmacokinetics of talazoparib have not been evaluated in patients < 18 years of age.
Elderly population. Of the 494 patients who received Talzenna, 85 patients were ≥ 65 years of age. No overall differences in safety or effectiveness of Talzenna were observed between these patients and younger patients but greater sensitivity of some older individuals cannot be ruled out.
Hepatic impairment. Based on a population PK analysis that included 490 patients who received talazoparib 1 mg daily as monotherapy, where 118 patients had mild hepatic impairment (total bilirubin ≤ 1.0 x ULN and AST > ULN, or total bilirubin > 1.0 to 1.5 x ULN and any AST), mild hepatic impairment had no effect on the PK of talazoparib. The PK of talazoparib in patients with normal hepatic function, mild hepatic impairment, moderate hepatic impairment (total bilirubin > 1.5 to 3.0 x ULN and any AST) or severe hepatic impairment (total bilirubin > 3.0 x ULN and any AST) was studied in a PK trial. Population PK analysis using data from this PK trial indicated that mild, moderate or severe hepatic impairment had no significant impact on the PK of talazoparib.
Renal impairment. Talzenna monotherapy.
Data from a PK trial in advanced cancer patients with varying degrees of renal impairment indicate that talazoparib total exposure (AUC0-24) after multiple talazoparib once-daily doses increased by 12%, 43% and 163% in patients with mild (eGFR 60 - 89 mL/min/1.73 m2), moderate (eGFR 30 - 59 mL/min/1.73 m2) and severe (eGFR 15 - 29 mL/min/1.73 m2) renal impairment, respectively, relative to patients with normal renal function (eGFR ≥ 90 mL/min/1.73 m2). Talazoparib Cmax increased by 11%, 32% and 89% in patients with mild, moderate and severe renal impairment, respectively, relative to patients with normal renal function. Consistent with these findings, a population PK analysis that included 490 patients, where 132 patients had mild renal impairment (60 mL/min ≤ CrCl < 90 mL/min), 33 patients had moderate renal impairment (30 mL/min ≤ CrCl < 60 mL/min) and 1 patient had severe renal impairment (CrCl < 30 mL/min), showed that talazoparib CL/F was decreased by 14% and 37% in patients with mild and moderate renal impairment, corresponding to 17% and 59% increase in AUC, respectively, when compared to patients with normal renal function (CrCl ≥ 90 mL/min). The PK of talazoparib have not been studied in patients requiring haemodialysis.
Talzenna coadministered with enzalutamide.
Based on a population PK analysis that included 412 mCRPC patients who received talazoparib co-administered with enzalutamide, where 152 patients had mild renal impairment (60 mL/min ≤ CrCl < 90 mL/min), 72 patients had moderate renal impairment (30 mL/min ≤ CrCl < 60 mL/min), and 2 patients had severe renal impairment (CrCl < 30 mL/min). The median predicted talazoparib CL/F was decreased by 8.0%, 27.1% and 46.7%, corresponding to increases of 9%, 37% and 88% in AUC, in patients with creatinine clearance values of 75 mL/min (representative of mild renal impairment), 45 mL/min (moderate renal impairment) and 22.5 mL/min (severe renal impairment), respectively, compared to a typical patient with normal renal function (90 mL/min). The PK of talazoparib has not been studied in patients requiring haemodialysis (see Section 4.2 Dose and Method of Administration, Dosage adjustment, Renal impairment).
Cardiac electrophysiology. The effect of talazoparib on cardiac repolarisation was evaluated using time-matched electrocardiograms (ECGs) in assessing the relationship between the change of the QT interval corrected for heart rate (QTc) from baseline and the corresponding plasma talazoparib concentrations in 37 patients with advanced solid tumours. Talazoparib did not have a clinically relevant effect on QTc prolongation at the maximum clinically recommended dose of 1 mg once daily.
5.3 Preclinical Safety Data
Genotoxicity.
Talazoparib was clastogenic in an in vitro chromosomal aberration assay in human peripheral blood lymphocytes and in an in vivo micronucleus assay in rats at exposures similar to clinically relevant doses. This clastogenicity is consistent with genomic instability resulting from the primary pharmacology of talazoparib, indicating the potential for genotoxicity in humans. Talazoparib was not mutagenic in a bacterial reverse mutation (Ames) test.
Carcinogenicity.
Carcinogenicity studies have not been conducted with talazoparib.6 Pharmaceutical Particulars
6.1 List of Excipients
Capsule content.
Silicified microcrystalline cellulose.
Capsule shells.
White body (0.1 mg, 0.25 mg, 0.5 mg and 1 mg strengths) or cap (0.1 mg strength).
Hypromellose, titanium dioxide.
Ivory body (0.35 mg strength) or cap (0.25 mg and 0.35 mg strengths).
Hypromellose, titanium dioxide, yellow iron oxide.
Light pink cap (0.5 mg strength).
Hypromellose, titanium dioxide, red iron oxide.
Light red cap (1 mg strength).
Hypromellose, titanium dioxide, red iron oxide, yellow iron oxide.
Printing ink (TekPrint SW-9008 Black).
Shellac, propylene glycol, ammonium hydroxide, black iron oxide, potassium hydroxide.
6.2 Incompatibilities
Not applicable.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 30°C. Protect from light.
6.5 Nature and Contents of Container
Talzenna 0.1 mg strength.
High-density polyethylene (HDPE) bottles with child-resistant polypropylene closures containing 30 capsules.
Talzenna 0.25 mg strength.
HDPE bottles with child-resistant polypropylene closures containing 30 capsules.
Polyvinyl chloride/polyvinylidene chloride (PVC/PVdC) blister with an aluminium peel off foil lidding in cartons containing 30, 60 or 90 capsules.
Talzenna 0.35 mg strength.
HDPE bottles with child-resistant polypropylene closures containing 30 capsules.
Talzenna 0.5 mg strength.
HDPE bottles with child-resistant polypropylene closures containing 30 capsules.
Talzenna 1 mg strength.
HDPE bottles with child-resistant polypropylene closures containing 30 capsules.
PVC/PVdC blister with an aluminium peel off foil lidding in cartons containing 30 capsules.
Not all presentations may be marketed.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
Chemical structure.
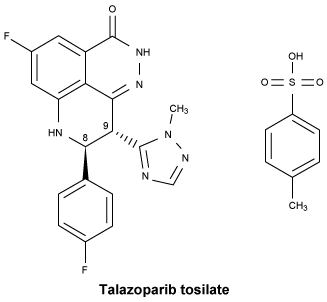
CAS number.
Talazoparib tosilate: 1373431-65-2.
Talazoparib: 1207456-01-6.7 Medicine Schedule (Poisons Standard)
S4 (Prescription Medicine).
Summary Table of Changes



 Refer to the enzalutamide Product Information for dose modifications for adverse reactions associated with enzalutamide.
Refer to the enzalutamide Product Information for dose modifications for adverse reactions associated with enzalutamide. The following adverse reactions have been identified in < 10% of the 286 patients receiving Talzenna and are not included in Table 4:
The following adverse reactions have been identified in < 10% of the 286 patients receiving Talzenna and are not included in Table 4:
 The following adverse reactions have been identified in < 10% of the 198 patients receiving Talzenna in combination with enzalutamide, and thus were not included in Table 6:
The following adverse reactions have been identified in < 10% of the 198 patients receiving Talzenna in combination with enzalutamide, and thus were not included in Table 6:




 Prespecified subgroup rPFS analyses were performed based on prognostic factors and baseline characteristics to evaluate the internal consistency of the treatment effect. Consistent with the overall results, a reduction in the risk of disease progression or death in favour of talazoparib in combination with enzalutamide was observed in patient subgroups shown in Figure 4.
Prespecified subgroup rPFS analyses were performed based on prognostic factors and baseline characteristics to evaluate the internal consistency of the treatment effect. Consistent with the overall results, a reduction in the risk of disease progression or death in favour of talazoparib in combination with enzalutamide was observed in patient subgroups shown in Figure 4. Exploratory subgroup analyses of rPFS for patients with BRCA-mutated (BRCAm) and non-BRCAm HRRm are presented in Table 10.
Exploratory subgroup analyses of rPFS for patients with BRCA-mutated (BRCAm) and non-BRCAm HRRm are presented in Table 10.

