What is in this leaflet
This leaflet answers some common questions about Tambocor tablets. It does not contain all the available information. It does not take the place of talking to your doctor or pharmacist.
All medicines have risks and benefits. Your doctor has weighed the risks of you taking Tambocor against the benefits they expect it can have for you.
If you have any concerns about taking this medicine, ask your doctor or pharmacist.
Keep this leaflet with the medicine. You may need to read it again.
What Tambocor is used for
Tambocor is used to treat a heart condition called arrhythmia. Arrhythmia is an irregular or abnormal heartbeat. The active ingredient in Tambocor, flecainide acetate, belongs to a group of drugs called antiarrhythmic medicines which slow down the impulses that cause the heart to beat in an irregular or abnormal way.
Your doctor may have prescribed Tambocor for another purpose.
Ask your doctor if you have any questions about why this medicine has been prescribed for you.
Tambocor is available only with a doctor's prescription.
Before using or being given Tambocor
When you must not use or have Tambocor
Do not use or have Tambocor if you have an allergy to:
- flecainide acetate
- any of the ingredients listed at the end of this leaflet.
Some of the symptoms of an allergic reaction may include:
- shortness of breath
- wheezing or difficulty breathing
- swelling of the face, lips, tongue or other parts of the body
- rash, itching or hives on the skin
Do not give Tambocor to children.
Do not take or have this medicine:
- if you have severe heart, liver or kidney disease unless your doctor says it is suitable for you
- after the USE BY date. The USE BY date can be found on the box and tablet blister. For the injection this can be found on the box and vial label
- if the packaging is torn or shows signs of tampering
If you are not sure whether you should start taking or be given this medicine, talk to your doctor.
Before you start to take or have Tambocor
Tell your doctor if you have allergies to any other medicines, foods, preservatives or dyes.
Tell your doctor or pharmacist if you are pregnant or plan to become pregnant or are breast-feeding. Your doctor or pharmacist can discuss with you the risks and benefits involved.
Tell your doctor if you have or have had any of the following:
- you have used Tambocor before and if you had any problems you think were caused by it
- you have or have had any kidney, liver or heart problems
Do not use or have Tambocor unless your doctor says it is suitable for you.
Taking other medicines
Tell your doctor about any other medicines you are taking, including those that you buy without a prescription from your pharmacy, supermarket or health food shop.
It is particularly important to tell your doctor or pharmacist if you are taking any medicines to:
- treat high blood pressure, angina or other heart conditions, for example digoxin, propranolol, atenolol, labetalol, alprenolol, pindolol, metoprolol, timolol, oxprenolol, sotalol, disopyramide, verapamil, amiodarone, diltiazem or nifedipine
- treat stomach ulcers, for example cimetidine
These medicines may be affected by Tambocor or may affect how well it works. You may need different amounts of your medicines, or you may need to take different medicines.
Your doctor and pharmacist may have more information on medicines to be careful of or avoid while taking or being given this medicine.
How to use Tambocor
Follow all directions given to you by your doctor or pharmacist carefully. They may differ from the information contained in this leaflet.
If you do not understand the instructions on the box, ask your doctor or pharmacist for help.
How much to take or have
In adults the usual starting dose of Tambocor tablets is 50 to 100 mg every twelve hours. Your doctor may gradually increase this dose. The usual maximum dose is 300 mg a day. However, some patients may need up to 400 mg a day.
It is very important to follow your doctor's instructions exactly. Never change the dose yourself. Do not stop taking this medicine unless your doctor tells you to.
Stopping Tambocor suddenly may make your condition worse.
Swallow your tablets with plenty of water. You can take the tablets before or after meals.
The dose for Tambocor IV injection is usually around 2mg/kg body weight and is given in a hospital. Your doctor may vary the dose to suit your condition.
Tambocor is not recommended for use in children.
If you forget to take Tambocor
If you forget to take Tambocor tablets, take them as soon as you remember. However, if it is almost time for your next dose skip the dose that you missed. Take your next dose at the normal time.
Never take a double dose to make up for a dose you have missed. If you miss two or more doses, please talk to your doctor.
For Tambocor injection, the doctor will assure that you are given the dose at the required time.
If you take too much Tambocor (overdose)
Immediately telephone your doctor or the Poisons Information Centre (in Australia call 131126; in New Zealand call 03 4747000) for advice, or go to Accident and Emergency at the nearest hospital, if you think you or anyone else may have used too much Tambocor. Do this even if there are no signs of discomfort or poisoning. You may need urgent attention.
Keep the telephone numbers of these places/services handy.
If anyone else takes some of your Tambocor, they should go to a hospital immediately.
Your doctor will ensure that you are not given too much Tambocor injection.
While you are taking Tambocor
Things you must do
If you are about to be started on any new medicines remind your doctor and pharmacist that you are taking or being given Tambocor.
Tell any other doctors, dentists and pharmacists who are treating you that you are taking Tambocor.
If you are going to have surgery, tell the surgeon or anaesthetist that you are taking this medicine. It may affect other medicines used during surgery.
If you become pregnant while taking or being given this medicine, tell your doctor immediately.
Things you must not do
Do not take Tambocor to treat any other complaints unless your doctor tells you to.
Tambocor is a prescription medicine and has been prescribed for you only.
Do not give it to anyone else even of their condition is the same as yours.
Do not stop taking Tambocor or lower the dose without first checking with your doctor.
Things to be careful of
If you feel dizzy for any reason do not drive or operate machinery.
Side effects
Tell your doctor or pharmacist as soon as possible if you do not feel well while you are taking or being given Tambocor. This medicine helps most people with arrhythmia, but it may have unwanted side effects in a few people. All medicines can have side effects. Sometimes they are serious, most of the time they are not. You may need medical attention if you get some of the side effects.
Do not be alarmed by the following lists of side effects. You may not experience any of them.
Ask your doctor or pharmacist to answer any questions you may have.
Tell your doctor as soon as possible if you notice any of the following:
- irregular or rapid heartbeat;
- dizziness - usually this is a minor and a temporary effect which goes away. If dizziness is severe, do not stop taking Tambocor but see your doctor or pharmacist
- blurred eyesight, double vision, cannot stand bright lights
- nausea
- difficulty in breathing
- tiredness
- headache, chest pain
If you experience any of these effects and find them troublesome, or if you experience any unwanted effects that are not listed here, contact your doctor or pharmacist immediately.
After using Tambocor
Storage
Keep Tambocor tablets and injection in a safe, dry, cool place where the temperature stays below 30°C.
Your injection will usually be stored in the hospital pharmacy.
Do not store in the fridge. Keep Tambocor tablets in their blister pack and carton until it is time to take them.
Keep Tambocor tablets where children cannot reach them. A locked cupboard at least one-and-a-half metres above the ground is a good place to store medicines.
The hospital pharmacy will ensure that Tambocor injection is safely stored before use.
Disposal
If your doctor tells you to stop taking the tablets, or if they have passed their USE BY date, ask your pharmacist what to do with any medicine that is left over.
Product description
What Tambocor looks like
Tambocor 50 mg tablets are white, round tablets marked TR50 on one side and plain on the other. Each pack contains 60 tablets.
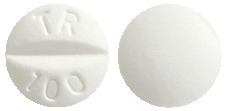
Tambocor 100 mg tablets are white, round tablets marked TR100 on one side and plain on the other. Each pack contains 60 tablets.
Tambocor IV injection is a clear, colourless solution in a clear glass ampoule. Each pack contains 5 ampoules.
Ingredients
Tambocor 50 mg tablets contain 50 mg of the active ingredient flecainide acetate.
Tambocor 100 mg tablets contain 100 mg of the active ingredient flecainide acetate.
Both tablets also contain the following inactive ingredients:
- starch
- croscarmellose sodium
- cellulose
- vegetable oil
- magnesium stearate
Tambocor tablets do not contain gluten.
Tambocor IV injection contains 150 mg of the active ingredient flecainide acetate. It also contains the following inactive ingredients:
- acetic acid
- sodium acetate
- water for injections
Sponsor
iNova Pharmaceuticals (Australia) Pty Limited
ABN: 13 617 871 539
Level 10, 12 Help Street
Chatswood NSW 2067
Toll Free: 1800 630 056
® = Registered
Australian Registration Numbers:
50 mg tablets: AUST R 41001.
100 mg tablets: AUST R 41002.
IV injection: AUST R 10592.
New Zealand Toll Free: 0508 375 394
This leaflet was updated in July 2019.
Published by MIMS October 2019