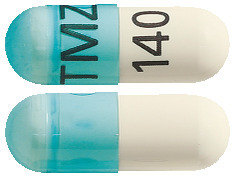What is in this leaflet
This leaflet answers some common questions about Temozolomide JUNO.
It does not contain all of the available information. It does not take the place of talking to your doctor or pharmacist.
All medicines have benefits and risks. Your doctor has weighed the risks of you taking Temozolomide JUNO against the benefits they expect it will have for you.
If you have any concerns about taking this medicine, talk to your doctor or pharmacist.
Keep this leaflet with your medicine. You may need to read it again.
What Temozolomide JUNO is used for
Temozolomide JUNO contains temozolomide as the active ingredient and belongs to a group of medicines called cytotoxics or chemotherapy medicines. It works by killing cancer cells and stopping cancer cells from growing and multiplying.
Temozolomide JUNO is used to treat:
- patients with brain tumours
- adult patients with advanced metastatic malignant melanoma.
Ask your doctor if you have any questions about why Temozolomide JUNO has been prescribed for you. Your doctor may have prescribed Temozolomide JUNO for another reason.
Use in children
Temozolomide JUNO can be used to treat children, 3 years of age and older, with specific forms of brain cancer (glioblastoma multiforme or anaplastic astrocytoma, showing recurrence or progression after standard therapy).
Temozolomide JUNO is available only with a doctor's prescription.
Before you take it
When you must not take it
Do not take Temozolomide JUNO if you are allergic to medicines containing temozolomide, dacarbazine or any of the ingredients listed at the end of this leaflet. Some of the symptoms of an allergic reaction may include skin rash, itching or hives, swelling of the face, lips or tongue which may cause difficulty in swallowing or breathing, wheezing or shortness of breath.
Do not take Temozolomide JUNO if you have very low levels of white blood cells, red blood cells or platelets.
Do not take Temozolomide JUNO if you are pregnant.
Do not take Temozolomide JUNO if you or your partner intend to become pregnant. Temozolomide JUNO may cause birth defects if either the male or female is using Temozolomide JUNO at the time of conception or during pregnancy. Therefore, female patients must have a negative pregnancy test before starting Temozolomide JUNO. Both male and female patients and their partners should each use some kind of birth control while taking Temozolomide JUNO. Male patients whose partners are already pregnant should use a condom to minimise exposure of the unborn baby to Temozolomide JUNO in the sperm.
Do not take Temozolomide JUNO if you are breastfeeding.
Do not take Temozolomide JUNO if the expiry date (Exp.) printed on the pack has passed.
Do not take Temozolomide JUNO if the packaging is torn or shows signs of tampering.
Before you start to take it
Tell your doctor if you are allergic to any other medicines, foods, dyes or preservatives.
Tell your doctor if you intend to have children. Temozolomide JUNO may cause infertility in men.
Tell your doctor if you have any medical conditions, especially the following:
- liver or kidney problems
- anaemia or blood clotting problems
Tell your doctor if you vomit frequently. Your doctor may give you medicine to control the vomiting.
If you have not told your doctor about any of the above, tell them before you start taking Temozolomide JUNO.
Taking other medicines
Tell your doctor if you are taking any other medicines, including any that you buy without a prescription from a pharmacy, supermarket or health food shop.
Some medicines may be affected by Temozolomide JUNO, or may affect how well it works. These include other medicines used to treat cancer or any other treatments that may affect your immune system. You may need different amounts of your medicine or you may need to use different medicines. Your doctor will advise you.
How to take it
Follow all directions given to you by your doctor carefully.
How much to take
Your doctor has worked out the exact dose of Temozolomide JUNO for you according to your individual needs. You may be given other medication to take before or after Temozolomide JUNO to help stop nausea.
Temozolomide JUNO in combination treatment with radiation (newly diagnosed patients):
If you are a patient with a newly diagnosed brain tumour, your doctor will start you on a dose of Temozolomide JUNO every day for 42 days (up to 49 days) in combination with radiation therapy. This is the first part of the treatment ("concomitant phase") in which you complete the radiation therapy. Your treatment will be interrupted for 4 weeks to give your body a chance to recover.
You will then start the next phase of treatment ("adjuvant phase") and your Temozolomide JUNO dose will change. In this phase, there are up to 6 treatment cycles. Each treatment cycle lasts 28 days. You will take your new dose of Temozolomide JUNO capsules once daily for the first five days ("dosing days") of each cycle, followed by 23 days without Temozolomide JUNO; this adds up to a 28 day treatment cycle. After day 28, the next cycle will begin, in which you will again take this medicine once daily for five days followed by 23 days without Temozolomide JUNO.
Before each new treatment cycle begins, your blood will be tested todetermine if the Temozolomide JUNO dose needs to be adjusted.
Taking Temozolomide JUNO alone (patients treated for recurrent brain tumour):
Take the dose the doctor has prescribed once a day for five days.
Depending on your response to Temozolomide JUNO, a new treatment cycle will begin each 28 days. You will then take this medicine again once daily for five days.
Before each new treatment cycle, your blood will be tested to see if the dose needs to be changed.
How to take it
Swallow the capsules whole with a glass of water. Do not open or chew the capsules.
Each time you start a new treatment cycle, be sure you understand exactly how many capsules of each strength you need to take on each day of dosing.
Temozolomide JUNO comes in different strength capsules (shown on the outer label in ‘mg’). Each strength is a different colour. Depending on the dose of Temozolomide JUNO that your doctor prescribes, you may have to take several capsules on each dosing day of the treatment cycle
Be sure you understand exactly how many capsules you need to take of each strength. Ask your doctor or pharmacist to write down the number of each strength (include colour) that you need to take on each dosing day.
Be sure you know exactly which days are your dosing days.
Be sure you review the dose with your health care provider each time you start a new cycle. Sometimes the dose or the mix of capsules you need to take will be different from the last cycle.
Once you take the medicine home, if you are confused or unsure about how to take your dose, call for re-instruction before beginning the treatment cycle. Errors in how you take this medicine may have serious health consequences.
When to take it
Take Temozolomide JUNO without food at least one hour before a meal. It is good practice to take it at about the same time each day.
If vomiting occurs after you take your capsules, do not take another dose that day.
How long to take it for
Keep taking Temozolomide JUNO exactly as your doctor recommends. Your doctor will tell you when your treatment should be stopped.
If you forget to take it
If you miss a dose, take the missed dose as soon as possible during the same day. If a full day has gone by, check with your doctor.
Do not take a double dose to make up for the dose you missed unless your doctor tells you to.
If you take too much (overdose)
Immediately telephone your doctor or the Poisons Information Centre (telephone 13 11 26) for advice, or go to Accident and Emergency at the nearest hospital, if you think you or anyone else may have taken too much Temozolomide JUNO. Do this even if there are no signs of discomfort or poisoning.
While you are taking it
Things you must do
Tell all doctors, dentists and pharmacists who are treating you that you are taking Temozolomide JUNO.
Tell your doctor if you feel sick or vomit while being treated with Temozolomide JUNO. Your doctor may give you another medicine to help with this.
Tell your doctor if you become unusually pale or tired, get blood clotting problems or frequent infections while being treated with Temozolomide JUNO. These could be caused by a low level of red blood cells, platelets or white blood cells in the blood. This is more common in patients over 70years of age. Your doctor may need to change your dose of Temozolomide JUNO.
Tell your doctor immediately if you or your partner becomes pregnant while taking Temozolomide JUNO.
Be sure to keep all your doctor's appointments so your progress can be checked. Your doctor may need to do some blood and other tests from time to time to check on your progress and detect any unwanted side effects.
Keep follow-up appointments with your doctor. It is important to have your follow-up doses of Temozolomide JUNO at the appropriate times to get the best effects from your treatment.
If you are about to be started on any new medicine, tell your doctor, dentist or pharmacist that you are being treated with Temozolomide JUNO.
Things you must not do
Do not open the capsules. If a capsule is damaged, avoid contact with your skin, eyes and nose. Avoid inhaling the powder. If you touch the powder or get some in your eyes or nose, wash the area with water.
Do not use Temozolomide JUNO to treat any other conditions unless your doctor tells you to.
Do not give Temozolomide JUNO to anyone else, even if they have the same condition as you.
Things to be careful of
Be careful driving or operating machinery until you know how Temozolomide JUNO affects you.
Temozolomide JUNO may cause drowsiness, in some people.
If this occurs, do not drive, operate machinery or do anything else that could be dangerous.
Side effects
Tell your doctor or pharmacist as soon as possible if you do not feel well while you are taking Temozolomide JUNO.
Like all other medicines, Temozolomide JUNO may have unwanted side effects in some people. Sometimes they are serious, most of the time they are not. You may need medical treatment if you get some of the side effects.
Ask your doctor or pharmacist to answer any questions you may have.
Tell your doctor if you notice any of the following and they worry you:
- nausea, vomiting, feeling unwell
- tiredness, sleepiness
- constipation
- headache
- loss of appetite or weight
- diarrhoea
- fever or high temperature
- rash, hair loss, itching
- dizziness, weakness
- general body pain
- stomach pain, indigestion
- different taste sensation
- mouth ulcers
- coughing
- sleeplessness.
These are the more common side effects of Temozolomide JUNO.
Tell your doctor as soon as possible if you notice any of the following:
- shortness of breath
- tingling or numbness in hands or feet
- bruising, bleeding or being unusually pale or tired. This could be caused by a low level of platelets or red blood cells in the blood
- any signs of infection, such as shivering that is associated with chills and fever, joint pain and loss of appetite. This could be a sign of an infection caused by a low level of white blood cells in the blood
- symptoms such as fever, headache, personality change, seizures and/or vomiting which could be associated with a brain infection caused by a herpes virus.
- fever, skin rash, swollen lymph nodes and/or facial swelling.
- symptoms of diabetes, such as passing large amounts of urine and constant thirst.
- development of red or purple spots under the skin.
These may be serious side effects. You may need medical attention.
Shivering associated with chills and fever or the development of red or purple spots under the skin may take some time to occur.
Therefore, even after you have finished your treatment with Temozolomide JUNO, you should tell your doctor immediately if you notice these side effects.
Tell your doctor if you notice anything else that is making you feel unwell. Other side effects not listed above may also occur in some patients.
Do not be alarmed by this list of possible side effects. You may not experience any of them.
After taking it
Storage
Keep Temozolomide JUNO where children cannot reach it. A locked cupboard at least one-and-a-half metres above the ground is a good place to store medicines.
Keep your capsules in the original sachets or bottles until it is time to take them.
Keep your capsules in a cool dry place where the temperature stays below 25°C.
Do not store Temozolomide JUNO or any other medicine in the bathroom or near a sink. Do not leave it in the car or on windowsills. Heat and dampness can destroy some medicines.
Disposal
Return any unused medicine to your pharmacist.
Product description
What it looks like
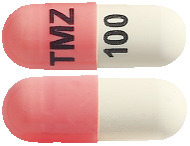
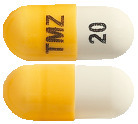
Temozolomide JUNO capsules come in 5 strengths:
- 5 mg– green and white capsule printed with “TMZ” and “5” in black
- 20 mg– yellow and white capsule printed with “TMZ” and “20” in black
- 100 mg– pink and white capsule printed with “TMZ” and “100” in black
- 140 mg– blue and white capsule printed with “TMZ” and “140” in black
- 180 mg – maroon and white capsule printed with “TMZ” and “180” in black
- 250 mg– white capsule printed with “TMZ” and “250” in black.
Sachet Pack: Each sachet contains 1 capsule. There are 5 sachets or 20 sachets in a pack.
Bottle Pack: Each bottle contains 5 capsules or 20 capsules.
Ingredients
The active ingredient in Temozolomide JUNO is temozolomide:
- each Temozolomide JUNO 5 capsule contains 5 mg of temozolomide
- each Temozolomide JUNO 20 capsule contains 20 mg of temozolomide
- each Temozolomide JUNO 100 capsule contains 100 mg of temozolomide
- each Temozolomide JUNO 140 capsule contains 140 mg of temozolomide
- each Temozolomide JUNO 180 capsule contains 180 mg of temozolomide
- each Temozolomide JUNO 250 capsule contains 250 mg of temozolomide.
Temozolomide JUNO contains sugars as lactose.
The capsules also contain:
- colloidal anhydrous silica
- sodium starch glycollate
- tartaric acid
- stearic acid.
The capsule shells contain:
- gelatin
- water
- titanium dioxide
- indigo carmine CI73015 (5 mg and 140 mg capsules only)
- iron oxide yellow (5 mg and 20 mg capsules only)
- iron oxide red (100 mg capsules only)
- sodium lauryl sulfate (250 mg capsules only)
- TekPrint SW-9008 black ink, (ARTG 2328).
Temozolomide JUNO does not contain sucrose, gluten, tartrazine or other azo dyes.
Sponsor
Juno Pharmaceuticals Pty Ltd
42 Kelso Street,
Cremorne,
VIC – 3121
Australian registration numbers (sachets):
- Temozolomide JUNO 5 - AUST R 206042
- Temozolomide JUNO 20 - AUST R 206043
- Temozolomide JUNO 100 - AUST R 206044
- Temozolomide JUNO 140 - AUST R 206215
- Temozolomide JUNO 180 - AUST R 206045
- Temozolomide JUNO 250 - AUST R 206046.
Australian registration numbers (bottles):
- Temozolomide JUNO 5 - AUST R 354885
- Temozolomide JUNO 20 - AUST R 354886
- Temozolomide JUNO 100 - AUST R 354887
- Temozolomide JUNO 140 - AUST R 354890
- Temozolomide JUNO 180 - AUST R 354888
- Temozolomide JUNO 250 - AUST R 354889.
This leaflet was revised in May 2021.
Published by MIMS July 2021