Notes
Distributed by Wagner Pharmaceuticals
1 Name of Medicine
Terbinafine (as hydrochloride).
2 Qualitative and Quantitative Composition
Each tablet contains 250 mg terbinafine (as hydrochloride) as the active ingredient.
For the full list of excipients see Section 6.1 List of Excipients.
3 Pharmaceutical Form
White, round, uncoated biconvex tablets with bevelled edges engraved with "APO" on one side and "TER" over "250" and scored through the centre of the other.
4.1 Therapeutic Indications
Terbinafine is indicated for:
Treatment in adults of ringworm (tinea corporis, tinea cruris and tinea pedis) due to infection caused by dermatophytes such as Trichophyton (e.g. T. rubrum, T. mentagrophytes, T. verrucosum, T. violaceum), Microsporum canis and Epidermophyton floccosum, where oral therapy is considered appropriate owing to the site, severity or extent of the infection, and the infection is not responsive to topical therapy.
Onychomycosis in adults (fungal infection of the nail) caused by dermatophyte fungi.
4.2 Dose and Method of Administration
Terbinafine-WGR tablets are intended for oral administration.
Dosage.
Skin infections.
250 mg once a day.
Onychomycosis.
250 mg once a day.
Administration.
Terbinafine tablets should be taken orally. The bioavailability of terbinafine is not affected by a light meal.
Duration of treatment.
Oral terbinafine should be prescribed only after topical therapy has failed and for the shortest time possible.
The duration of treatment varies according to the indication and the severity of the infection.
Skin infections.
Likely durations of treatment are as follows:
tinea pedis (interdigital, plantar/moccasin type): 2 to 6 weeks;
tinea corporis, cruris: 2 to 4 weeks.
Complete resolution of the signs and symptoms of infection may not occur until several weeks after mycological cure.
Onychomycosis.
For most patients the duration for successful treatment is between 6 weeks and 3 months.
Infections of finger and toenails (other than big toe) usually respond to the shorter duration of treatment, particularly in patients of younger age with a normal rate of nail outgrowth. In patients with slow nail growth, treatment for up to three months is usually adequate. However, infections in the big toe, or if nail growth is very poor, treatment for up to 6 months may be necessary.
Optimal clinical effect is seen some months after mycological cure and cessation of treatment. This is related to the period required for outgrowth of healthy nail tissue.4.3 Contraindications
Hypersensitivity to terbinafine or any of the excipients in the formulation (see Section 6.1 List of Excipients).
Severe, chronic, or active hepatic disease (see Section 4.4 Special Warnings and Precautions for Use).
4.4 Special Warnings and Precautions for Use
Use in hepatic impairment.
Terbinafine is contraindicated for patients with chronic or active hepatic disease. Before prescribing terbinafine tablets, liver function tests should be performed since hepatotoxicity may occur in patients with and without pre-existing liver disease. Therefore, periodic monitoring (after 4-6 weeks of treatment) of liver function tests is recommended. Terbinafine should be immediately discontinued in case of elevation of liver function tests. Very rare cases of liver failure, some leading to death or liver transplant, have been reported with the use of terbinafine. In the majority of liver failure case, the patients had underlying systemic conditions (see Section 4.8 Adverse Effects (Undesirable Effects)). Patients prescribed terbinafine tablets should be warned to report immediately any symptoms of persistent nausea, anorexia, fatigue, vomiting, right upper abdominal pain or jaundice, dark urine or pale stools. Patients with these symptoms should discontinue taking oral terbinafine and the patient's liver function should be immediately evaluated.
Use in renal impairment.
The use of terbinafine tablets in patients with impaired renal function (creatinine clearance less than 50 mL/min or serum creatinine of more than 300 micromol/L) has not been adequately studied and therefore is not recommended.
Effect on vision.
During high-dose studies in monkeys, refractile irregularities were observed in the retina at doses that were 30 to 60 times the human dose (non-toxic effect level 50 mg/kg). The clinical relevance of this observation is unknown. However, the ocular effects in monkeys were not confirmed in humans in the placebo-controlled trials, where the incidence of ophthalmic abnormalities was lower in the terbinafine tablet-treated patients (1.1%) compared with those who received placebo (1.5%).
Effect on blood.
Patients taking terbinafine tablets are at risk of developing agranulocytosis, thrombocytopenia, neutropenia and pancytopenia which are very rarely associated with terbinafine. The problem usually resolves within a few days to a week of withdrawal of terbinafine. Patients taking terbinafine should be advised to report any symptoms of infections. Prescribers should examine the patient to determine the correct aetiology of any blood dyscrasias that occurs in patients treated with terbinafine, and consideration should be given to a possible change in medication regimen, including discontinuation of treatment with terbinafine.
Cases of thrombotic thrombocytopenic purpura (TTP), some fatal, have been reported with terbinafine. Discontinue terbinafine if clinical symptoms and laboratory findings consistent with TTP occur. The findings of unexplained thrombocytopenia and anemia should prompt further evaluation and consideration of the diagnosis of TTP.
Dermatological effects.
Serious skin reactions (e.g. Stevens-Johnson syndrome, toxic epidermal necrolysis, drug rash with eosinophilia and systemic symptoms) have been very rarely reported in patients taking terbinafine tablets.
If progressive skin rash occurs, treatment with terbinafine tablets should be discontinued.
Terbinafine should be used with caution in patients with pre-existing psoriasis or lupus erythematosus as precipitation and exacerbation of psoriasis and cutaneous and systemic lupus erythematosus have been reported in a postmarketing setting.
Drug resistance.
Drug resistance has been reported with the use of terbinafine in dermatophytes, especially Trichophyton species. Prescribers should take into consideration the local prevalence of drug resistance and if an alternate treatment should be considered.
Paediatric use.
There is no experience with terbinafine in children and its use cannot be recommended.
This product should be kept out of reach of children.
Use in the elderly.
There is no evidence to suggest that elderly patients require different dosages or experience side effects different from those in younger patients. When using terbinafine tablets in this age group the possibility of impairment of liver or kidney function should be considered (see Section 4.4 Special Warnings and Precautions for Use).
Effects on laboratory tests.
Transient decreases in absolute lymphocyte counts (ALC).
Transient decreases in absolute lymphocyte counts (ALC) have been observed in controlled clinical trials. In placebo-controlled trials, 8/465 terbinafine-treated patients (1.7%) and 3/137 placebo-treated patients (2.2%) had decreases in ALC to below 1000/mm3 on two or more occasions. The clinical significance of this observation is unknown. However, in patients with known or suspected immunodeficiency, physicians should consider monitoring complete blood counts in individuals using terbinafine therapy for greater than six weeks.
Effect on lipids.
In chronic toxicity studies in rats, oral terbinafine, at a dose of 309 mg/kg per day, increased serum cholesterol levels.
This effect was more marked in female, than in male, rats. Effects on triglycerides levels were not consistent among the various studies. In monkeys a daily dose of 300 mg/kg increased triglyceride levels and chylomicron concentrations. In a small clinical study, a daily dose of 250 mg for 8 weeks did not result in detectable changes in the plasma lipid profile. In other clinical trials there was no evidence of a significant change in the plasma lipid profile of patients.4.5 Interactions with Other Medicines and Other Forms of Interactions
The plasma clearance of terbinafine may be accelerated by drugs that induce metabolism (see Rifampicin) and may be inhibited by drugs that inhibit (see Cimetidine) cytochrome P450.
Where co-administration of such agents is necessary, the dosage of terbinafine may need to be adjusted accordingly.
There have been spontaneous reports of increase or decrease in prothrombin times in patients concomitantly taking oral terbinafine and warfarin, however, a causal relationship between terbinafine tablets and these changes has not been established.
Cautious use of terbinafine is advised in women taking oral contraceptives since a few cases of menstrual disorders have been reported in patients taking this drug combination, although the incidence of these disorders remains within the background incidence of patients taking oral contraceptives alone.
The following medicinal products may increase the effect or plasma concentration of terbinafine.
Cimetidine.
Cimetidine decreased the clearance of terbinafine by 33%. Where co-administration of such agents is necessary, the dosage of terbinafine may need to be adjusted accordingly.
Fluconazole.
Fluconazole significantly increased the Cmax and AUC of terbinafine, due to inhibition of both CYP2C9 and CYP3A4 enzymes. Similar increase in exposure may occur when other drugs which may inhibit both CYP2C9 and CYP3A4, such as ketoconazole and amiodarone, are concomitantly administered with terbinafine.
The following medicinal products may decrease the effect or plasma concentration of terbinafine.
Rifampicin.
Rifampicin increased the clearance of terbinafine by 100 %. Where co-administration of such agents is necessary, the dosage of terbinafine may need to be adjusted accordingly.
Effect of terbinafine on other medicinal products.
In vitro and in vivo studies showed negligible potential for interaction with the drugs that are metabolised via the CYP450 system except those with CYP2D6-mediated metabolism.
Terbinafine does not interfere with the metabolism of antipyrine or digoxin. Terbinafine clearance is unaffected by cyclosporine.
There was no effect of terbinafine on the pharmacokinetics of fluconazole. Further there was no clinically relevant interaction between terbinafine and the potential comedications cotrimoxazole (trimethoprim and sulfamethoxazole), zidovudine or theophylline.
Terbinafine may increase the effect or plasma concentration of the following medicinal products. Compounds predominantly metabolised by CYP2D6.
Terbinafine inhibits the CYP2D6-mediated metabolism. Therefore, patients receiving concomitant treatment with drugs predominantly metabolised by this enzyme, such as tricyclic antidepressants (TCAs; e.g. Desipramine), beta-blockers, selective serotonin reuptake inhibitors (SSRIs), antiarrhythmics Class 1A, 1B and 1C and monoamine oxidase inhibitors (MAOIs) Type B, should be followed, if the co-administered drug has a narrow therapeutic window.
In studies in healthy subjects characterised as extensive metabolisers of dextromethorphan (antitussive drug and CYP2D6 probe substrate), terbinafine significantly increased the dextromethorphan/dextrorphan metabolic ratio in urine. Thus terbinafine may convert extensive CYP2D6 metabolisers to poor metaboliser status.
Caffeine.
Terbinafine decreased the clearance of caffeine administered intravenously by 19%.
Terbinafine may decrease the effect of plasma concentration of the following medicinal products. Cyclosporin.
Terbinafine increased the clearance of cyclosporin by 15%.4.6 Fertility, Pregnancy and Lactation
Effects on fertility.
No data available.
(Category B1)
Foetal toxicity and fertility studies in animals suggest no adverse effects.
Since clinical experience in pregnant women is not available, terbinafine tablets should not be used during pregnancy unless the potential benefits outweigh any potential risks.
Terbinafine is excreted in breast milk. Therefore, mothers receiving oral treatment with terbinafine tablets should not breastfeed.4.7 Effects on Ability to Drive and Use Machines
No studies on the effects of terbinafine tablets treatment on the ability to drive and use machines have been performed. Patients who experience dizziness as an undesirable effect should avoid driving vehicles or using machines.
4.8 Adverse Effects (Undesirable Effects)
In general terbinafine is well tolerated. In clinical trials, adverse events occurred in 10.4% of patients taking terbinafine and 5.6% of patients taking placebo. Most adverse events were mild to moderate in severity and of a short duration.
Adverse drug reactions from clinical trials experience are listed by MedDRA system organ class. Within each system organ class, the adverse drug reactions are ranked under heading by frequency, with the most frequent reactions first. The frequency category for each adverse drug reaction is based on the following convention (CIOMS III): very common (≥ 1/10); common (≥ 1/100 to, < 1/10); uncommon (≥ 1/1,000 to, < 1/100); rare (≥ 1/10,000 to, < 1/1,000); very rare (< 1/10,000).
Blood and lymphatic system disorders.
Uncommon: anaemia.
Very rare: haematological disorders such as neutropenia, agranulocytosis, pancytopenia, thrombocytopenia.
Gastrointestinal disorders.
Very common: nausea, vomiting, flatulence, mild abdominal discomfort, abdominal cramps, anorexia, diarrhoea, dyspepsia/gastritis, belching, abdominal distension, decreased appetite.
Uncommon: taste disturbances, including taste loss, which usually recover within several weeks after discontinuation of the drug. Isolated cases of prolonged taste disturbances have been reported. A decrease of food intake leading to significant weight loss was observed in very few cases.
Hepatobiliary disorders.
Rare: transient increases in liver enzymes, hepatobiliary dysfunction, cholestatic jaundice, liver failure (some leading to liver transplant or death). In the majority of liver failure cases, the patients had underlying systemic conditions (see Section 4.3 Contraindications).
Immune system disorders.
Very rare: anaphylactoid reactions (including angioedema), cutaneous and systemic lupus erythematosus.
Musculoskeletal and connective tissue disorders.
Very common: musculoskeletal reactions (arthralgia, myalgia).
Psychiatric disorders.
Common: depression.
Very rare: anxiety.
Skin and subcutaneous tissue disorders.
Very common: urticaria, rash.
Common: pruritus, erythema.
Uncommon: photosensitivity reactions.
Very rare: psoriaform eruptions or exacerbation of psoriasis, Stevens-Johnson syndrome, toxic epidermal necrolysis, erythema multiforme, acute generalised exanthematous pustulosis, cutaneous and systemic lupus erythematosus, anaphylactoid reactions (including angioedema), toxic skin eruption, dermatitis exfoliative, dermatitis bullous, alopecia. In the event of an allergic or severe skin reaction, terbinafine treatment should be discontinued.
Nervous system disorders.
Very common: headache.
Common: dysgeusia* including ageusia*, dizziness, tiredness/fatigue.
Uncommon: paraesthesia and hypoaesthesia.
Very rare: sedation, light-headedness, chest pain, hair loss and allergic reactions (including anaphylaxis).
Eye disorders.
Common: visual impairment.
Ear and labyrinth disorders.
Uncommon: tinnitus.
General disorders.
Uncommon: pyrexia.
Investigations.
Uncommon: weight decreased**.
Effect on laboratory tests.
Transient rises in serum urea/serum creatinine and liver enzymes.
Transient decreases in haematocrit, haemoglobin, and leucocytes.
* Hypogeusia, including ageusia, which usually recover within several weeks after discontinuation of the drug. Isolated cases of prolonged hypogeusia have been reported.
** Weight decreased secondary to dysgeusia.
Other adverse drug reactions from post-marketing spontaneous reports.
The following adverse drug reactions have been identified based on post-marketing experience with terbinafine tablets via spontaneous case reports and literature cases. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency which is therefore categorised as not known. Adverse drug reactions are listed according to system organ classes in MedDRA.
Blood and lymphatic system disorders.
Anaemia, thrombotic thrombocytopenic purpura.
Ear and labyrinth disorders.
Hypoacusis, impaired hearing, tinnitus.
Eye disorders.
Vision blurred, visual acuity reduced.
Gastrointestinal disorders.
Pancreatitis.
General disorders and administration site conditions.
Influenza-like illness, pyrexia.
Immune system disorders.
Anaphylactic reaction, serum sickness-like reaction.
Investigations.
Blood creatine phosphokinase increased.
Musculoskeletal and connective tissue disorders.
Rhabdomyolysis.
Nervous system disorders.
Anosmia including permanent anosmia, hyposmia.
Psychiatric disorders.
Anxiety and depressive symptoms secondary to taste disturbances.
Skin and subcutaneous tissue disorders.
Photosensitivity reactions (e.g. photodermatosis, photosensitivity allergic reaction and polymorphic light eruption), drug rash with eosinophilia and systemic symptoms.
Vascular disorders.
Vasculitis.
Reporting suspected adverse effects.
Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at http://www.tga.gov.au/reporting-problems.4.9 Overdose
A few cases of overdosage (up to 5 g) have been reported.
Signs and symptoms.
Studies in animals suggest that in a high dose situation, such as accidental overdose, central nervous symptoms (CNS) may appear. The relevance of those effects to man is unknown. However, these effects can be monitored.
Central nervous system.
Headache and dizziness.
Gastrointestinal system.
Nausea, and epigastric pain.
Treatment.
The recommended treatment of overdosage consists in eliminating the drug, primarily by the administration of activated charcoal, and giving symptomatic supportive therapy, if needed.
Activated charcoal may reduce the absorption of the drug if given within one or two hours of ingestion. In patients who are not fully conscious or have an impaired gag reflex, consideration should be given to administering activated charcoal via a nasogastric tube, once the airway is protected.
For information on the management of overdose, contact the Poisons Information Centre on 131126 (Australia).5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Pharmacotherapeutic group: Oral antifungal agent, ATC Code: D01B A02.
Terbinafine is an allylamine with antifungal activity mainly against dermatophytes, including Trichophyton (e.g. T. rubrum, T. mentagrophytes, T. verrucosum, T. violaceum), Microsporum canis, and Epidermophyton floccosum.
Mechanism of action.
Terbinafine interferes specifically with fungal sterol biosynthesis at an early step. This leads to a deficiency in ergosterol and to an intracellular accumulation of squalene, resulting in fungal cell death. Terbinafine acts by inhibition of squalene epoxidase in the fungal cell membrane. The enzyme squalene epoxidase is not linked to the cytochrome P450 system. When administered orally, the drug concentrates in skin and nails at levels associated with antifungal activity.
Drug resistance.
The potential terbinafine resistance in dermatophytes may be associated with mutations in erg1, the target gene for squalene epoxidase/monooxygenase (SQLE). There have been reports of some Trichophyton isolates (such as T. mentagrophytes, T. indotinae, T. rubrum, T. interdigitale) with reduced susceptibility to terbinafine, suggesting a potential for development of drug resistance (see Section 4.4 Special Warnings and Precautions for Use).
The clinical significance of this observation is not fully understood.
Clinical trials.
No data available.
5.2 Pharmacokinetic Properties
Absorption.
Following oral administration, terbinafine is well absorbed (> 70%) and the bioavailability of terbinafine hydrochloride from terbinafine as a result of first-pass metabolism is approximately 40%.
A single oral dose of 250 mg terbinafine results in peak plasma concentrations (Cmax) of 0.83 microgram/mL within 2 hours of administration. The absorption half-life is 0.8 hours and the distribution half-life is 4.6 hours.
An increase in the AUC of terbinafine, of less than 20% is observed when terbinafine is administrated with food. At steady-state, in comparison to a single dose, the peak concentration of terbinafine is 25% higher and plasma AUC increases by a factor of 2.5; the increase in plasma AUC is consistent with an effective half-life of ~ 36 hours.
Distribution.
Terbinafine binds strongly to plasma proteins (99%). It concentrates in the lipophilic stratum corneum. Terbinafine is also secreted in sebum, thus achieving high concentrations in hair follicles, hair and sebum-rich skins. There is also evidence from animal studies that terbinafine is distributed into the nail plate in the first few weeks after commencing therapy. Animal studies also indicate that terbinafine accumulates in all lipophilic tissues including the retinal and choroid tissues. In studies conducted so far, no ophthalmological abnormalities attributable to terbinafine have been reported in humans.
Metabolism.
Terbinafine is extensively metabolised in the body. Biotransformation results in metabolites with no antifungal activity.
Excretion.
Terbinafine and its metabolites are excreted predominantly in the urine. The elimination half-life is 17 hours. There is no evidence of accumulation in individuals with normal hepatic and renal function. No age-dependent changes in pharmacokinetics have been observed. In patients with renal impairment (creatinine clearance ≤ 50 mL/min) or with pre-existing liver disease, the clearance of terbinafine is decreased by approximately 50% compared to normal volunteers.
When administered orally, the drug concentrates in skin and nails at levels associated with antifungal activity.
5.3 Preclinical Safety Data
Genotoxicity.
No data available.
Carcinogenicity.
In a 2 year rat carcinogenicity study, small but significant increases in hepatocellular carcinomas, adenomas and combined tumours were seen in males at a dietary dose of 69 mg/kg per day. No increase in hepatic tumours was seen in female rats at a dietary dose of 97 mg/kg per day.6 Pharmaceutical Particulars
6.1 List of Excipients
Methylcellulose, croscarmellose sodium, magnesium stearate, colloidal anhydrous silica.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 25°C. Protect from light.
6.5 Nature and Contents of Container
250 mg tablets.
Terbinafine-WGR tablets.
Blister pack (PVC/PVDC/Al) of 42 tablets (AUST R 100028).
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
Terbinafine hydrochloride is a white to off-white, finely crystalline powder. It is soluble in isopropyl alcohol (> 70 mg/mL at 25°C) and ethanol (> 70 mg/mL at 25°C), and slightly soluble in water (6.3 mg/mL at 25°C).
Chemical structure.
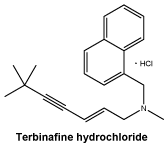 Chemical name: (E)-N-(6,6-dimethyl-2-hepten-4-ynyl)-N-methyl-1-naphthalenemethanamine hydrochloride.
Chemical name: (E)-N-(6,6-dimethyl-2-hepten-4-ynyl)-N-methyl-1-naphthalenemethanamine hydrochloride.
Molecular formula: C21H26ClN.
Molecular weight: Terbinafine base: 291.44.
Terbinafine hydrochloride: 327.90.
CAS number.
78628-80-5.7 Medicine Schedule (Poisons Standard)
S4 - Prescription Only Medicine.
Summary Table of Changes
