What is in this leaflet
This leaflet answers some common questions about TERIMIDE. It does not contain all the available information. It does not take the place of talking to your doctor or pharmacist.
All medicines have risks and benefits. Your doctor has weighed the risks of you taking TERIMIDE against the benefits they expect it will have for you.
If you have any concerns about taking this medicine, ask your doctor or pharmacist.
Keep this leaflet with the medicine. You may need to read it again.
What TERIMIDE is used for
This medicine is used to treat relapsing forms of multiple sclerosis (MS).
This medicine belongs to a group of medicines called immunomodulating drugs.
The cause of MS is not yet known. MS affects the central nervous system (CNS) - the brain and spinal cord. In MS, the body's immune system reacts against its own myelin (the 'insulation' or the protective sheath surrounding nerve fibres).
With relapsing forms of MS, people can have repeated attacks or relapses of inflammation of the CNS from time to time. Symptoms vary from patient to patient and may include blurred vision, weakness in the legs or arms, or loss of control of bowel or bladder function. These are followed by periods of recovery.
This medicine works by selectively interfering with the ability of white blood cells (lymphocytes) to produce the disease response and nerve damage that ultimately leads to relapses.
TERIMIDE has been shown to reduce or decrease the number of relapses and slow down the progression of physical disability in patients with relapsing forms of MS. Although it is not a cure, patients treated with TERIMIDE generally find they will have fewer relapses.
Ask your doctor if you have any questions about why this medicine has been prescribed for you. Your doctor may have prescribed it for another reason.
This medicine is available only with a doctor's prescription.
Before you take TERIMIDE
When you must not take it
Do not take TERIMIDE if you have an allergy to:
- any medicine containing teriflunomide
- any of the ingredients listed at the end of this leaflet
- any other similar medicines
Some of the symptoms of an allergic reaction may include:
- shortness of breath
- wheezing or difficulty breathing
- swelling of the face, lips, tongue or other parts of the body
- rash, itching or hives on the skin
Do not take this medicine if you have:
- any diseases which reduce your body's natural defences
- any diseases of the blood
- any serious skin disorders such as rash with blisters and peeling of the skin or extended rashes with the liver, blood and other body organs involvement
- severe liver disease
- a condition called hypoproteinaemia (when you do not have enough protein in your blood)
Do not take this medicine if you are not using reliable birth control.
Do not take this medicine if you are pregnant. It may affect your developing baby if you take it during pregnancy.
Do not breast-feed if you are taking this medicine. The active ingredient in TERIMIDE passes into breast milk and there is a possibility that your baby may be affected.
You must not become pregnant while taking TERIMIDE and for a certain period of time after stopping TERIMIDE. TERIMIDE may increase the risk of birth defects.
Do not give this medicine to a child under the age of 18 years. Safety and effectiveness in children younger than 18 years have not been established.
Do not take this medicine after the expiry date printed on the pack or if the packaging is torn or shows signs of tampering. If it has expired or is damaged, return it to your pharmacist for disposal.
If you are not sure whether you should start taking this medicine, talk to your doctor.
Before you start to take it
Tell your doctor if you have allergies to any other medicines, foods, preservatives or dyes.
Tell your doctor if you have or have had any of the following medical conditions:
- liver problems
- high blood pressure
- chronic or serious infection
- a decrease in the number of white blood cells or an illness which lowered your body's resistance to disease
- tuberculosis
- lung problems, such as interstitial lung disease (an inflammation of lung tissue) which is a serious and potentially fatal disease
- kidney disease
Before you start TERIMIDE, your doctor will need to take blood samples to check the health of your liver and blood cells. This must be done in the last 6 months before starting this medicine.
Tell your doctor if you are pregnant or plan to become pregnant or are breast-feeding, or if you intend to father a child. TERIMIDE may increase the risk of birth defects. To reduce any risk to the developing baby, you will need to stop taking TERIMIDE and may need to undergo a wash-out procedure. Your doctor will discuss the wash-out procedure with you.
Tell your doctor if you plan to have surgery.
If you have not told your doctor about any of the above, tell them before you start taking TERIMIDE.
Taking other medicines
Tell your doctor or pharmacist if you are taking any other medicines, including any that you get without a prescription from your pharmacy, supermarket or health food shop.
Some medicines and TERIMIDE may interfere with each other. These include:
- antibiotics such as rifampicin, cefaclor, penicillin G and ciprofloxacin
- medicines used to treat epilepsy such as carbamazepine and phenytoin
- some medicines used for diabetes such as repaglinide or pioglitazone
- some medicines used to treat cancer such as paclitaxel, doxorubicin, methotrexate
- some medicines used to treat depression such as duloxetine
- ondansetron, a medicine used to prevent and treat nausea
- theophylline, a medicine used to treat asthma
- warfarin, a medicine used to prevent blood clots
- some medicines used to lower cholesterol such as statins, rosuvastatin
- St. John's Wort (Hypericum perforatum)
These medicines may be affected by TERIMIDE or may affect how well it works. You may need different amounts of your medicines, or you may need to take different medicines.
In certain situations, for example, if you experience a serious side effect, you change your medication or you want to fall pregnant, your doctor will ask you to take medication that will help your body get rid of TERIMIDE faster.
Your doctor and pharmacist have more information on medicines to be careful with or avoid while taking this medicine.
How to take TERIMIDE
Follow all directions given to you by your doctor or pharmacist carefully. They may differ from the information contained in this leaflet.
If you do not understand the instructions on the box, ask your doctor or pharmacist for help.
How much to take
The usual dose for this medicine is one 14 mg tablet per day.
Your doctor may have prescribed a different dose.
How to take it
Swallow the tablets whole with a full glass of water.
When to take it
Take your medicine at about the same time each day. Taking it at the same time each day will have the best effect. It will also help you remember when to take it.
It does not matter if you take this medicine before or after food.
How long to take it
Continue taking your medicine for as long as your doctor tells you.
This medicine helps to control your condition, but does not cure it. It is important to keep taking your medicine even if you feel well.
If you forget to take it
Skip the dose you missed and take your next dose when you are meant to.
Otherwise, take it as soon as you remember, and then go back to taking your medicine as you would normally.
Do not take a double dose to make up for the dose that you missed. This may increase the chance of you getting an unwanted side effect.
If you are not sure what to do, ask your doctor or pharmacist.
If you have trouble remembering to take your medicine, ask your pharmacist for some hints.
If you take too much (overdose)
Immediately telephone your doctor or the Poisons Information Centre (telephone 13 11 26) for advice, or go to Accident and Emergency at the nearest hospital, if you think that you or anyone else may have taken too much TERIMIDE. Do this even if there are no signs of discomfort or poisoning. You may need urgent medical attention.
While you are using TERIMIDE
Things you must do
If you are about to be started on any new medicine, remind your doctor and pharmacist that you are taking TERIMIDE.
Tell any other doctors, dentists and pharmacists who treat you that you are taking this medicine.
If you are going to have surgery, tell the surgeon or anaesthetist that you are taking this medicine. It may affect other medicines used during surgery.
If you become pregnant while taking this medicine, stop taking it and tell your doctor immediately.
If you need to stop treatment, the levels of TERIMIDE in your blood can be lowered rapidly using a special wash-out procedure.
If you are female talk to your doctor about birth control that you must use during treatment. You must use reliable methods of birth control while taking TERIMIDE.
Talk to your doctor if you plan to become pregnant or before you stop taking this medicine.
Tell your doctor immediately if you:
- have an infection or notice chills or a fever or signs of an infection while taking this medicine
- skin becomes itchy or yellow, the whites of your eyes become yellow, you start to bleed or bruise easily, or your urine becomes very dark or you have stomach pain, nausea or vomiting, stop taking it immediately - you may be developing a liver problem
- experience numbness or tingling in the hands or feet or numbness or weakness of the arms and legs
Tell your doctor if you need to have a vaccination during treatment with this medicine or for 6 months after stopping treatment.
Keep all of your doctor's appointments so that your progress can be checked. Your doctor may do some tests from time to time to make sure the medicine is working and to prevent unwanted side effects.
Things you must not do
Do not take TERIMIDE to treat any other complaints unless your doctor tells you to.
Do not give your medicine to anyone else, even if they have the same condition as you.
Do not stop taking your medicine or lower the dosage without checking with your doctor.
Things to be careful of
Be careful when drinking alcohol while you are taking this medicine. The effects of alcohol could be made worse while taking TERIMIDE. It is recommended you minimise your alcohol intake while taking this medicine.
Side effects
Tell your doctor or pharmacist as soon as possible if you do not feel well while you are taking TERIMIDE.
This medicine helps most people with MS, but it may have unwanted side effects in a few people. All medicines can have side effects. Sometimes they are serious, most of the time they are not. You may need medical attention if you get some of the side effects.
If you are over 65 years of age you may have an increased chance of getting side effects.
Do not be alarmed by the following lists of side effects. You may not experience any of them.
Ask your doctor or pharmacist to answer any questions you may have.
Tell your doctor or pharmacist if you notice any of the following and they worry you:
- diarrhoea
- rashes, itchy skin
- hair loss
- weight loss
- unusual tiredness or weakness
- numbness or tingling of hands or feet
- thickened, inflamed, red patches on the skin, which may be accompanied with whitish scaly skin, or accompanied by white pus-filled bumps
- nail disorders
The above list includes the more common side effects of your medicine.
Tell your doctor as soon as possible if you notice any of the following:
- signs and symptoms of severe infection, such as fever
- severe upper stomach pain, often with nausea and vomiting
- severe skin rash or sores in your mouth
- pale skin, fatigue, or proneness to bruising
- if you develop new or worsening symptoms such as cough or trouble breathing, with or without a fever
- inflammation of the colon, symptoms include diarrhoea usually with blood and mucus
The above list includes serious side effects that may require medical attention.
If any of the following happen, tell your doctor immediately or go to Accident and Emergency at your nearest hospital:
- swelling of the face, lips, mouth or throat, which may cause difficulty in swallowing or breathing
- hives
- fainting
- yellowing of the skin and eyes (jaundice)
- thickened patches of red skin
The above list includes very serious side effects. You may have had a serious allergic reaction to TERIMIDE. You may need urgent medical attention or hospitalisation.
Tell your doctor or pharmacist if you notice anything that is making you feel unwell.
Other side effects not listed above may also occur in some people.
After using TERIMIDE
Storage
Keep your tablets in the pack until it is time to take them. If you take the tablets out of the pack they may not keep well.
Keep your tablets in a cool dry place where the temperature stays below 25°C.
Do not store TERIMIDE or any other medicine in the bathroom or near a sink. Do not leave it on a window sill or in the car. Heat and dampness can destroy some medicines.
Keep it where children cannot reach it. A locked cupboard at least one-and-a-half metres above the ground is a good place to store medicines.
Disposal
If your doctor tells you to stop taking this medicine or the expiry date has passed, ask your pharmacist what to do with any medicine that is left over.
Product description
What it looks like
TERIMIDE tablets are pale blue to pastel blue, pentagonal film-coated tablets debossed with "L" on one side and "598" on the other side.
TERIMIDE tablets are available in blister packs of 28 tablets.
Ingredients
TERIMIDE contains 14 mg of teriflunomide as the active ingredient.
The tablets also contain the following inactive ingredients:
- colloidal anhydrous silica
- hyprolose
- lactose monohydrate
- magnesium stearate
- maize starch
- microcrystalline cellulose
- OPADRY complete film coating system 03F505071 BLUE
- purified water
- sodium starch glycollate type A
TERIMIDE contains lactose.
Distributor
TERIMIDE is distributed in Australia by:
Alphapharm Pty Ltd trading as Viatris
Level 1, 30 The Bond
30-34 Hickson Road
Millers Point NSW 2000
www.viatris.com.au
Phone: 1800 274 276
This leaflet was prepared in October 2021.
AUST R 299149
terimide_cmi\Oct21/00
Published by MIMS December 2021


 Headache, palpitations, arthralgia, myalgia, increased blood creatine phosphokinase and decreased white blood cell count were also observed.
Headache, palpitations, arthralgia, myalgia, increased blood creatine phosphokinase and decreased white blood cell count were also observed.
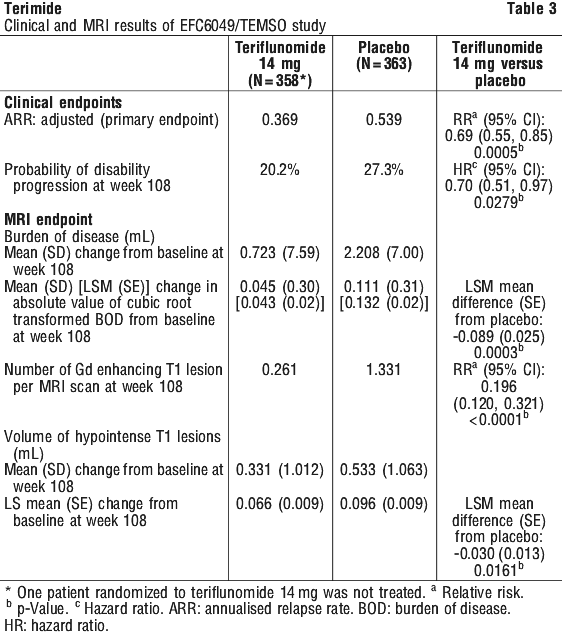
 The probability of disability progression at 108 weeks (with 90% CIs) was 0.273 (0.223, 0.323) for placebo and 0.202 (0.156, 0.247) for teriflunomide 14 mg.
The probability of disability progression at 108 weeks (with 90% CIs) was 0.273 (0.223, 0.323) for placebo and 0.202 (0.156, 0.247) for teriflunomide 14 mg.
 Study 3 (EFC6260/TOPIC) was a double-blind, placebo-controlled study that evaluated once daily doses of teriflunomide 7 mg and 14 mg for up to 108 weeks in patients with early MS (i.e. a first clinical episode). Patients had a first neurological event occurring within 90 days of randomization, with 2 or more T2 lesions at least 3 mm in diameter that were characteristic of MS. The primary endpoint was time to a second clinical episode (relapse).
Study 3 (EFC6260/TOPIC) was a double-blind, placebo-controlled study that evaluated once daily doses of teriflunomide 7 mg and 14 mg for up to 108 weeks in patients with early MS (i.e. a first clinical episode). Patients had a first neurological event occurring within 90 days of randomization, with 2 or more T2 lesions at least 3 mm in diameter that were characteristic of MS. The primary endpoint was time to a second clinical episode (relapse). The teriflunomide MRI activity was also shown in a phase 2 study (study 4 (Study 2001)). A total of 179 patients received 7 mg (n=61) or 14 mg (n=57) of teriflunomide or placebo (n= 61) for 36 weeks duration. Baseline demographics were consistent across treatment groups. The mean number of unique active lesions per brain MRI scan during the 36-week treatment period was lower in patients treated with teriflunomide 14 mg (0.98) as compared to placebo (2.69), the difference being statistically significant (p=0.0052).
The teriflunomide MRI activity was also shown in a phase 2 study (study 4 (Study 2001)). A total of 179 patients received 7 mg (n=61) or 14 mg (n=57) of teriflunomide or placebo (n= 61) for 36 weeks duration. Baseline demographics were consistent across treatment groups. The mean number of unique active lesions per brain MRI scan during the 36-week treatment period was lower in patients treated with teriflunomide 14 mg (0.98) as compared to placebo (2.69), the difference being statistically significant (p=0.0052). Molecular formula: C12H9F3N2O2.
Molecular formula: C12H9F3N2O2.