What is in this leaflet
This leaflet answers some common questions about this medicine. It does not contain all the available information. It does not take the place of talking to your doctor or pharmacist.
All medicines have risks and benefits. Your doctor has weighed the risks of you taking this medicine against the benefits they expect it will have for you.
If you have any concerns about taking this medicine, ask your doctor or pharmacist.
Keep this leaflet with the medicine. You may need to read it again.
What this medicine is used for
The name of your medicine is Terry White Chemists Mirtazapine. It contains the active ingredient mirtazapine.
It is used to treat and prevent the reoccurrence of depression.
Depression is longer lasting or more severe than the "low moods" everyone has from time to time due to the stress of everyday life. It is thought to be caused by a chemical imbalance in parts of the brain. This affects your whole body and can cause emotional and physical symptoms, such as feeling low in spirit, loss of interest in activities, unable to enjoy life, poor appetite or overeating, disturbed sleep, often waking up early, loss of sex drive, lack of energy and feeling guilty over nothing.
Mirtazapine corrects this chemical imbalance and may help relieve the symptoms of depression.
Ask your doctor if you have any questions about why this medicine has been prescribed for you. Your doctor may have prescribed this medicine for another reason.
This medicine is available only with a doctor's prescription.
There is no evidence that this medicine is addictive.
Use in children
This medicine must not be used in children or adolescents under 18 years of age.
Before you take this medicine
When you must not take it
Do not take this medicine if you have an allergy to:
- any medicine containing mirtazapine
- any of the ingredients listed at the end of this leaflet
Some of the symptoms of an allergic reaction may include:
- shortness of breath
- wheezing or difficulty breathing
- swelling of the face, lips, tongue or other parts of the body
- rash, itching or hives on the skin
Do not take this medicine if you are taking other medicines called monoamine oxidase inhibitor (MAOI) or have been taking a MAOI within the last 14 days. MAOIs may be used for the treatment of depression (phenelzine, tranylcypromine, moclobemide), Parkinson's disease (selegiline), infections (linezolid), or diagnosis of certain conditions/treatment of certain blood disorders (methylene blue).
There may be other MAOIs not listed above, so check with your doctor or pharmacist.
Do not take this medicine if you are pregnant. Mirtazapine is not recommended to be used during pregnancy, as it may affect your developing baby. Your doctor will discuss the risks and benefits of taking mirtazapine when pregnant. Adequate methods of contraception should be used when taking mirtazapine.
Do not breastfeed if you are taking this medicine. Mirtazapine may pass into breast milk and there is a possibility that your baby may be affected. Do not take this medicine whilst breastfeeding until you and your doctor have discussed the risks and benefits involved.
Do not take this medicine after the expiry date printed on the pack or if the packaging is torn or shows signs of tampering. If it has expired or is damaged, return it to your pharmacist for disposal.
If you are not sure whether you should start taking this medicine, talk to your doctor.
Before you start to take it
Tell your doctor if you have allergies to any other medicines, foods, preservatives or dyes.
Tell your doctor if you have or have had any of the following medical conditions:
- thoughts of suicide or self-harm
- epilepsy (fits or convulsions)
- liver problems, such as jaundice
- kidney problems
- heart disease
- certain kinds of heart conditions that may change your heart rhythm, a recent heart attack, heart failure, or take certain medicines that may affect the heart's rhythm
- low blood pressure
- any mental illness (e.g. schizophrenia, bipolar disorder)
- diabetes
- glaucoma (increased pressure in the eyes)
- difficulties in urination due to an enlarged prostate
- unexplained high fever, sore throat and mouth ulcers
- galactose or lactose intolerance
- glucose-galactose malabsorption
- low sodium levels in your blood (hyponatremia).
Tell your doctor if you are pregnant or plan to become pregnant or are breastfeeding.
Your doctor can discuss with you the risks and benefits involved.
If you have not told your doctor about any of the above, tell them before you start taking this medicine.
Taking other medicines
Tell your doctor or pharmacist if you are taking any other medicines, including any that you get without a prescription from your pharmacy, supermarket or health food shop.
Some medicines and mirtazapine may interfere with each other. These include:
- other medicines (e.g. SSRIs, venlafaxine, nefazodone) for depression, anxiety, obsessive compulsive disorders or pre-menstrual dysphoric disorder
- MAOIs (such as phenelzine, tranylcypromine, moclobemide, linezolid, and selegiline)
- medicines to help you sleep or calm down (e.g. benzodiazepines and tranquillisers)
- other sedatives especially antipsychotics, antihistamine and opioids
- medicines containing St. John's wort (hypericum perforatum)
- phenytoin or carbamazepine, used to treat epilepsy
- warfarin, used to prevent blood clotting
- rifampicin, linezolid and erythromycin, antibiotics used to treat infections
- medicines to treat fungal infections, such as ketoconazole
- HIV/AIDS medicines
- cimetidine, used to treat reflux or stomach ulcers
- triptans used to treat migraines, such as sumatriptan, naratriptan and zolmitriptan
- tramadol, strong painkiller
- methylene blue, an injectable diagnostic dye
- tryptophan, found in some preparations bought in health food shops
These medicines may be affected by mirtazapine or may affect how well it works. You may need different amounts of your medicines, or you may need to take different medicines.
Your doctor and pharmacist have more information on medicines to be careful with or avoid while taking this medicine.
How to take this medicine
Follow carefully all directions given to you by your doctor or pharmacist. Their instructions may be different to the information in this leaflet.
How much to take
This will depend on your condition and whether you are taking any other medicines.
The usual starting dose is 15 mg per day. Your doctor may slowly increase your dose depending on how you respond to mirtazapine.
The effective dose for most people is usually between 30 mg and 45 mg per day.
Do not stop taking your medicine or change your dosage without first checking with your doctor.
How to take it
Swallow the tablets whole with a full glass of water.
When to take it
Take this medicine at the same time each day, preferably as a single night-time dose before going to bed.
If recommended by your doctor, your dose may be taken in sub-doses equally divided over the day (e.g. once in the morning and once at night-time before going to bed).
It does not matter if you take it before, with or after food.
How long to take it for
Continue taking your medicine for as long as your doctor tells you.
For depression, the length of treatment will depend on how quickly your symptoms improve. Most antidepressants take time to work, so do not be discouraged if you don't feel better right away.
Some of your symptoms may improve in 1-2 weeks, but it can take up to 2-4 weeks to feel the full benefit of the medicine.
Even when you feel well, you will usually have to take your medicine for 4-6 months, or even longer, to make sure the benefits will last.
If you forget to take it
Once-Daily Dosing:
If you forget to take your dose before going to bed, do not take the missed dose the next morning, because it may cause drowsiness or sleepiness during the day. Wait until the next evening before taking your normal dose.
Do not take a double dose to make up for the missed dose.
Twice-Daily Dosing:
If you forget your morning dose, simply take it together with your evening dose.
If you forget your evening dose, do not take it with the next morning dose. Skip the missed dose and continue treatment with your normal morning and evening doses.
If you have forgotten both doses, you should not make up for the missed doses; the next day, just continue with your normal morning and evening doses.
If you are not sure what to do, ask your doctor or pharmacist.
If you have trouble remembering to take your medicine, ask your pharmacist for some hints.
If you take too much (overdose)
Immediately telephone your doctor or the Poisons Information Centre (telephone 13 11 26) for advice or go to Accident and Emergency at the nearest hospital, if you think that you or anyone else may have taken too much of this medicine. Do this even if there are no signs of discomfort or poisoning. You may need urgent medical attention.
If you take too much mirtazapine you may feel drowsy, dizzy, confused, agitated, have increased heart rate or lose consciousness.
While you are taking this medicine
Things you must do
If you are about to be started on any new medicine, remind your doctor and pharmacist that you are taking this medicine.
Tell any other doctors, dentists, and pharmacists who treat you that you are taking this medicine.
If you are going to have surgery, tell the surgeon or anaesthetist that you are taking this medicine. It may affect other medicines used during surgery.
If you become pregnant or start to breastfeed while taking this medicine, tell your doctor immediately.
If you are about to have any blood tests, tell your doctor that you are taking this medicine. It may interfere with the results of some tests.
Keep all your doctor's appointments so that your progress can be checked. Your doctor may occasionally do tests to make sure the medicine is working and to prevent side effects.
Go to your doctor regularly for a check-up.
Tell your doctor immediately if you develop fever, chills, sore throat or mouth ulcers or experience other signs of infections. In rare cases mirtazapine can cause a shortage of white blood cells, resulting in lowering body resistance to infection. These symptoms may appear after 2-6 weeks of treatment.
Tell your doctor immediately or go to the nearest hospital for treatment if you have any suicidal thoughts or other mental/mood changes.
Young adults aged 18 to 24 are at particularly high risk of having this happen to them.
Occasionally, the symptoms of depression or other psychiatric conditions may include thoughts of harming yourself or committing suicide. Until the full antidepressant effect of the medicine becomes apparent, it is possible that these symptoms may increase in the first few weeks of treatment.
If you or someone you know is demonstrating any of the following warning signs of suicide while taking this medicine, contact your doctor or health professional immediately or go to the nearest hospital for treatment:
- thoughts or talk of death or suicide
- thoughts or talk of self-harm or harm to others
- any recent attempts to self-harm
- increase in aggressive behaviour, irritability or agitation.
All mention of suicide or violence must be taken seriously.
If you have diabetes take care to monitor your blood sugar levels. Your diabetes medication may need to be adjusted.
Tell your doctor if, for any reason, you have not taken your medicine exactly as prescribed. Otherwise your doctor may think that it was not effective and change your treatment unnecessarily.
Tell your doctor if you feel this medicine is not helping your condition.
Things you must not do
Do not take this medicine to treat any other complaints unless your doctor tells you to.
Do not give your medicine to anyone else, even if they have the same condition as you.
Do not stop taking your medicine or lower the dosage without checking with your doctor. Stopping your medicine suddenly may cause nausea, headache, dizziness, anxiety or agitation.
Your doctor may want to gradually reduce the amount of medicine you are taking before stopping completely.
Things to be careful of
Be careful when driving or operating machinery until you know how this medicine affects you. Mirtazapine may cause drowsiness, sleepiness or dizziness in some people and affect alertness and concentration.
If any of these occur, do not drive, operate machinery or do anything else that could be dangerous.
Be careful when drinking alcohol while taking mirtazapine. Taking mirtazapine with alcohol can make you sleepier and less alert.
Your doctor may suggest you avoid alcohol while being treated with this medicine.
Side effects
Tell your doctor or pharmacist as soon as possible if you do not feel well while you are taking mirtazapine or if you have any questions or concerns.
All medicines can have side effects. Sometimes they are serious but most of the time they are not.
Do not be alarmed by the following lists of side effects. You may not experience any of them.
Tell your doctor or pharmacist if you notice any of the following and they worry you:
- lethargy, drowsiness or sleepiness
- headache
- tiredness
- increase in appetite and weight gain
- dry mouth or increased salivation
- diarrhoea or constipation
- nausea or vomiting
- dizziness or faintness, especially when getting up quickly from a lying or sitting position (hypotension)
- swollen ankles or feet, as a result of fluid accumulation (oedema)
- fluid retention with weight gain
- mild rash or skin eruptions
- nightmares/vivid dreams
- tingling fingers or toes
- painful joints or back
- muscle aches and pains
- restless legs
- reduced energy or enthusiasm
- anxiety or inability to sleep or stay asleep (insomnia) – these may be symptoms of depression
- difficulty in passing urine (urinary retention)
- increased prolactin hormone levels in blood (hyperprolactinaemia, including symptoms such as enlarged breasts and/or milky nipple discharge)
- sleepwalking
Tell your doctor as soon as possible if you notice any of the following:
- abnormal sensations in the skin e.g. burning, stinging, tickling, tingling, itching or any type of skin rash or blistering, peeling or flaking skin
- low sodium levels (feeling weak, sick, confused, exhausted, with muscle weakness or cramps)
- feeling extremely restless and having an overwhelming urge to always be moving
- abnormal sensations in the mouth sensations of numbness in the mouth or swelling in the mouth
- unusual changes in behaviour
- eye pain (a possible sign of glaucoma or raised pressure in the eye)
The above list includes serious side effects that may require medical attention. Serious side effects are rare.
If any of the following happen, tell your doctor immediately or go to Accident and Emergency at your nearest hospital:
- thoughts or actions relating to suicide or self-harm
- severe skin reaction which starts with painful red areas, then large blisters and ends with peeling of layers of skin - this may be accompanied by fever and chills, aching muscles and generally feeling unwell
- chest pain, fast or irregular heartbeat
- epilepsy or fits (seizures)
- shaking or tremors
- sudden muscle contractions (myoclonus)
- fainting, cough and problems breathing (signs of a blood clot on the lung)
- attack of excessive excitability (mania)
- agitation
- confusion
- hallucinations (hearing, seeing or feeling things that are not there)
- yellow colouring of eyes or skin; this may suggest problems with your liver
- generalised fluid retention with weight gain
- fever, sore throat, mouth ulcers, gastrointestinal (stomach, bowels) disturbances
- fever, sweating, increased heart rate, uncontrollable diarrhoea, muscle contractions, shivering, overactive reflexes, restlessness, mood changes and unconsciousness (serotonin syndrome).
- abdominal pain and nausea; this may suggest inflammation of the pancreas
- skin rash, itching or hives; swelling of the face, lips or tongue which may cause difficulty breathing
- a combination of symptoms such as fever, sweating, increased heart rate, diarrhoea, (uncontrollable) muscle contractions, shivering, overactive reflexes, restlessness, mood changes unconsciousness and increased salivation (serotonin syndrome)
- muscle pain, stiffness and/or weakness, darkening or discolouration of the urine (rhabdomyolysis)
The above list includes very serious side effects. You may need urgent medical attention or hospitalisation. These side effects are very rare.
Tell your doctor or pharmacist if you notice anything that is making you feel unwell.
Other side effects not listed above may occur in some patients.
Some side effects may occur with no symptoms and may only be identified by tests.
For example, high blood pressure, high cholesterol or fat levels, changes in levels of white or red blood cells.
Storage and disposal
Storage
Keep your medicine in its original packaging until it is time to take it. If you take your medicine out of its original packaging it may not keep well.
Keep your medicine in a cool dry place where the temperature will stay below 30°C.
Do not store your medicine, or any other medicine, in the bathroom or near a sink. Do not leave it on a window sill or in the car. Heat and dampness can destroy some medicines.
Keep this medicine where children cannot reach it. A locked cupboard at least one-and-a-half metres above the ground is a good place to store medicines.
Disposal
If your doctor or pharmacist tells you to stop taking this medicine or it has passed its expiry date, your pharmacist can dispose of the remaining medicine safely.
Product description
What Terry White Chemists Mirtazapine looks like
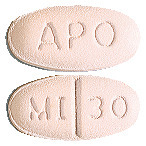
30 mg tablets
Light pink, oval shaped, scored, film coated tablets, imprinted "APO" on one side and "MI" bisect "30" on the other side.
Blister packs of 30 tablets. AUST R 127683.
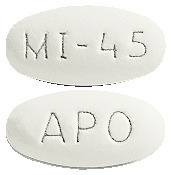
45 mg tablets
White to off-white, oval shaped, unscored, film coated tablets, imprinted "APO" on one side and "MI-45" on the other side.
Blister packs of 30 tablets. AUST R 127699.
* Not all strengths and/or pack types may be available.
Ingredients
Each tablet contains 30 mg or 45 mg of mirtazapine as the active ingredient.
It also contains the following inactive ingredients:
- lactose monohydrate microcrystalline cellulose
- croscarmellose sodium
- magnesium stearate
- hypromellose
- hyprolose
- macrogol 8000
- titanium dioxide
- iron oxide red CI 77491 (30 mg only)
- iron oxide yellow CI 77492 (15 mg and 30 mg only).
This medicine is gluten-free, sucrose-free, tartrazine-free and free of other azo dyes.
Sponsor
Apotex Pty Ltd
16 Giffnock Avenue
Macquarie Park NSW 2113
This leaflet was last updated in: July 2019.
Published by MIMS September 2019



 Some secondary parameter results have been excluded from Table 3. These were number of:
Some secondary parameter results have been excluded from Table 3. These were number of:

