1 Name of Medicine
Tetrabenazine.
2 Qualitative and Quantitative Composition
Each tablet contains 25 mg tetrabenazine.
Excipients with known effects.
Contains sugars as lactose.
For the full list of excipients, see Section 6.1 List of Excipients.3 Pharmaceutical Form
Tablet, uncoated.
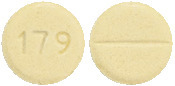
Yellow, circular, flat faced beveled edge uncoated tablet debossed with "179" on one side and scored on the other side.
4.1 Therapeutic Indications
May be useful for the control of chorea, hemiballismus, tardive and buccolingual dyskinesias and certain dystonic syndromes.
4.2 Dose and Method of Administration
Dosage.
Dosage given below is a guide only. For each patient, the dose of tetrabenazine should be titrated to determine the most appropriate dose. Treatment should be reassessed periodically in the context of the patient's underlying condition and concomitant medications.
Method of administration.
Adults.
An initial dosage in adults of 25 mg twice a day is recommended. This can be increased by 25 mg a day every 3 or 4 days until the desired therapeutic effect is achieved, or until 200 mg/day is given, or unwanted side effects intervene.
Children.
In children, 12.5 mg twice a day has been used as an initial dose with increments of 12.5 mg every 3 to 4 days until the desired therapeutic effect is obtained, or an upper limit of 3 mg/kg/day is reached, or unwanted side effects intervene.
Note.
Only very limited information on use in children is available.
Dosage adjustment.
Dosage may need to be reduced in patients with impaired renal or hepatic function or in elderly patients. It is reported that if no improvement is found after 7 to 10 days at the maximum dose then it is unlikely that a higher dose or a longer duration of therapy will benefit the patient.4.3 Contraindications
Tetrabenazine should not be given closer than one day before or in combination with levodopa or reserpine as it blocks the action of these drugs, particularly the central action.
Tetrabenazine should not be administered to persons with a known sensitivity to tetrabenazine or to any of the excipients.
Tetrabenazine should not be given to patients with Parkinsonism or depression, as it may worsen these conditions. It should not be administered within two weeks of treatment with a monoamine oxidase inhibitor (MAOI).
Tetrabenazine is contraindicated during breast-feeding.
4.4 Special Warnings and Precautions for Use
Identified precautions.
As for other CNS active drugs, the effect of combination of tetrabenazine and other central depressants including alcohol should be considered. Tetrabenazine may potentiate the action of antihypertensive drugs.
Depression.
Tetrabenazine may cause depression or worsen pre-existing depression. Cases of suicidal ideation and behaviour have been reported in patients taking the product. Particular caution should be exercised in treating patients with a history of depression or prior suicide attempts or ideation.
If depression or suicidal ideation occurs it should be controlled by reducing the dose and/or initiating antidepressant therapy. If depression or suicidal ideation is profound, or persists, discontinuation of tetrabenazine and initiation of antidepressant therapy should be considered.
MAOI antidepressants should not be used until at least two weeks have elapsed since the last tetrabenazine dose, to avoid restlessness, disorientation and confusion, as well as a potentially serious drug interaction resulting in hypertensive crisis.
Parkinsonism.
Tetrabenazine can induce Parkinsonism and exacerbate pre-existing symptoms of Parkinson's disease. The tetrabenazine dose should be adjusted as clinically indicated to minimise this side effect.
Neuroleptic malignant syndrome.
Neuroleptic malignant syndrome is a rare complication of tetrabenazine therapy. Neuroleptic malignant syndrome most often occurs early in treatment or in response to changes in dose. The main symptoms of this condition are mental changes, rigidity, hyperthermia, autonomic dysfunction (sweating and fluctuations in blood pressure) and elevated creatinine phosphokinase levels. If neuroleptic malignant syndrome is suspected tetrabenazine should be withdrawn immediately and appropriate treatment initiated.
QTc.
Tetrabenazine causes a small increase (about 8 msec) in the corrected QT interval. In slow metabolisers this increase may be greater (30 msec). Tetrabenazine should be used with caution with other drugs known to prolong QTc and in patients with congenital long QT syndromes and a history of cardiac arrhythmias (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
Dysphagia and choking.
Dysphagia and choking attacks with a possibly consequent bronchopneumonia appear to be the only acutely dangerous adverse effects of tetrabenazine reported so far. If these occur, therapy should be discontinued.
Lactose.
Patients with rare hereditary problems of galactose intolerance, the Lapp lactase deficiency or glucose-galactose malabsorption, should not take tetrabenazine tablets as they contain lactose.
Orthostatic hypotension.
Tetrabenazine may induce postural hypotension at therapeutic doses and symptoms may include postural dizziness and fainting. These should be considered in patients who may be vulnerable to hypotension or its effects. Monitoring of vital signs on standing should be considered in patients who are vulnerable to hypotension.
Use in renal impairment.
The use of tetrabenazine in patients with renal insufficiency has not been studied.
Use in the elderly.
No specific studies have been performed in the elderly.
Paediatric use.
No adequately controlled clinical studies have been performed in children.
Effects on laboratory tests.
There are no special requirements to monitor effects on laboratory tests.4.5 Interactions with Other Medicines and Other Forms of Interactions
Interaction may occur when the following medications are administered with tetrabenazine (see Section 4.3 Contraindications; Section 4.4 Special Warnings and Precautions for Use):
Reserpine and levodopa.
Inhibit the action of these drugs and thereby attenuate their effects.
MAOIs.
Possible serious interactions resulting in hypertensive crisis. At least 14 days should elapse between the discontinuation of a MAOI and initiation of treatment with tetrabenazine.
Tricyclic antidepressants.
Have been reported to antagonise the locomotor activity induced by tetrabenazine in animals.
CNS stimulants and depressants.
Possible additive sedative effects should be considered when used in conjunction with CNS depressants (including alcohol, neuroleptics, hypnotics and opioids).
Neuroleptic agents.
Potential for significant dopamine depletion when administering with neuroleptic agents e.g. haloperidol, chlorpromazine and metoclopramide. Patients should be monitored clinically for the development of Parkinsonism. Neuroleptic malignant syndrome has been observed in isolated cases.
Antihypertensives.
May increase risk of orthostatic hypotension.
Beta-blockers.
May increase risk of orthostatic hypotension.
CYP2D6 inhibitors.
In vitro and in vivo studies indicate that tetrabenazine and its metabolites, α- and β-dihydotetrabenazine and are substrates for CYP2D6. β-HTBZ was also an inhibitor of CYP2D6 at clinically relevant concentrations. Caution should be used when adding a CYP2D6 inhibitor (such as fluoxetine, paroxetine, quinidine, duloxetine, terbinafine, amiodarone, or sertraline) to a patient already receiving a stable dose of tetrabenazine, or if the dose is influenced by the patient's CYP2D6 metaboliser status. A reduction in the dose of tetrabenazine should be considered.
Antipsychotics, antibiotics and class IA and III antiarrhythmic medications.
Tetrabenazine causes a small increase (about 8 msec) in the corrected QT interval.4.6 Fertility, Pregnancy and Lactation
Effects on fertility.
There are no data available on the potential of tetrabenazine to affect fertility.
(Category B3)
There is inadequate evidence of safety of the drug in human pregnancy, and the potential risk to humans is unknown. Tetrabenazine crosses the placenta and, because of the lack of data, tetrabenazine should not be used during pregnancy. In the developmental toxicity tests there was no evidence of in utero mortality, growth retardation or teratogenicity in either rats or rabbits at oral doses up to 5 and 27 times the clinical dose based on body surface area. When tetrabenazine was administered orally to female rats from the beginning of organogenesis to weaning, an increase in stillbirths and postnatal mortality was observed from 15 mg/kg/day, while delayed development was seen at 30 mg/kg/day. The no-effect dose for neonatal effects (5 mg/kg/day) was approximately equivalent to the clinical dose based on body surface area. The relative contributions of in utero and neonatal exposure and postnatal maternal neglect to these effects are unclear.
Tetrabenazine is excreted in milk. Oral administration to rats from early gestation to weaning was associated with increased stillbirths, hypothermia and neonatal mortality in pups (15 mg/kg/day, twice the clinical dose based on body surface area), and delayed pup development (30 mg/kg/day, 5 fold the clinical dose). The relative contributions of in utero and neonatal exposure and postnatal maternal neglect to these effects are unclear. Tetrabenazine is contraindicated during breast-feeding.4.7 Effects on Ability to Drive and Use Machines
As drowsiness may occur in up to 20% of patients, caution should be used when driving or operating machines until competence to do so under treatment has been established. It might be possible to reduce the drowsiness by careful dosage adjustment, especially initially.
4.8 Adverse Effects (Undesirable Effects)
Side effects include drowsiness, depression (which has on occasion been reported to be associated with suicidal ideation and behaviour) and Parkinsonism. The most commonly reported side effects are as follows (see Table 1):
 Neuroleptic malignant syndrome (NMS) associated with the use of tetrabenazine has been reported rarely. This may occur soon after initiation of therapy, following an increase in dosage or after prolonged treatment. The main symptoms are mental changes, rigidity, hyperthermia, autonomic dysfunction and elevated creatinine phosphokinase levels. If NMS is suspected tetrabenazine should be withdrawn and appropriate supportive therapy instituted; treatment with dantrolene and bromocriptine may be effective.
Neuroleptic malignant syndrome (NMS) associated with the use of tetrabenazine has been reported rarely. This may occur soon after initiation of therapy, following an increase in dosage or after prolonged treatment. The main symptoms are mental changes, rigidity, hyperthermia, autonomic dysfunction and elevated creatinine phosphokinase levels. If NMS is suspected tetrabenazine should be withdrawn and appropriate supportive therapy instituted; treatment with dantrolene and bromocriptine may be effective.
Reporting of suspected adverse effects.
Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.4.9 Overdose
Signs and symptoms of overdosage may include nausea, vomiting, diarrhoea, confusion, hallucinations, sedation, drowsiness, sweating, hypotension and hypothermia.
Management should be supportive. There is no information available on the effect of pharmacological antagonists or of dialysis.
For information on the management of overdose, contact the Poisons Information Centre on 131126 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action.
Tetrabenazine is a synthetic derivative of benzylquinolizine that causes depletion of dopamine and other monoamines in the central nervous system.
Studies conducted in vitro have shown that tetrabenazine is an inhibitor of monoamine transportation into pre-synaptic neuronal vesicles, by inhibition of VMAT2 (vesicular monoamine transporter 2), which is principally located in the central nervous system. Dihydrotetrabenazine, the principal metabolite of tetrabenazine, has a similar affinity and more significant selectivity for VMAT2, and both compounds are believed to contribute to the pharmacological effect.
At a synaptic level tetrabenazine creates a reversible depletion of monoamines in the presynaptic vesicles. Animal studies have shown that tetrabenazine causes preferential depletion of dopamine from nerve terminals in the CNS but neurotransmitter depletion by a single dose of tetrabenazine is reversible and lasts only a few hours. This pharmacological effect explains the therapeutic benefit of tetrabenazine in patients' suffering from hyperkinetic movement disorders.
Clinical trials.
No data available.
5.2 Pharmacokinetic Properties
Absorption.
Based on metabolite levels, tetrabenazine is quickly and mostly absorbed after oral administration. Its absorption is not affected by the taking of food. After administration of single doses from 12.5 to 50 mg of tetrabenazine, for the metabolites alpha and beta dihydrotetrabenazine, the maximum plasma concentration and the area under the curve increased approximately in proportion to the dose, indicating a linear kinetic. Clinical testing has shown that a single oral dose of tetrabenazine undergoes extensive (> 75%) absorption from the gastro-intestinal tract.
Metabolism.
The metabolism of tetrabenazine is complex, initially proceeding via the formation of alpha and beta dihydrotetrabenazine. The majority of the observed metabolites appear to be formed from these dihydrotetrabenazines as a result of O-dealkylation, hydroxylation and conjugation. No significant build-up has been observed after daily administration. The elimination half-life of dihydrotetrabenazine is approximately 5 to 6 hours.
Excretion.
Tetrabenazine is mostly eliminated in metabolised form in urine.
5.3 Preclinical Safety Data
Genotoxicity.
Tetrabenazine and its major metabolites, α- and β-dihydrotetrabenazine, were negative in the in vitro bacterial reverse mutation assay. Tetrabenazine was clastogenic in the in vitro chromosome aberration assay in Chinese hamster ovary cells in the presence of metabolic activation, while α- and β-dihydrotetrabenazine were clastogenic in Chinese hamster lung cells in the presence and absence of metabolic activation. In vivo micronucleus tests in rats and mice with tetrabenazine were negative. The genotoxic potential of tetrabenazine is considered to be low.
Carcinogenicity.
A 6 month study in transgenic p53 (+/-) heterozygous mice and a long term study in male rats at oral doses up to 30 mg/kg/day and 6 mg/kg twice daily, respectively, did not provide any evidence of carcinogenic potential. These doses correspond to exposures approximately 3 to 4 times the clinical exposure, based on plasma AUC or dose based on body surface area. Mammary gland hyperplasia was observed in female rats that received twice daily oral doses of 7.5 mg/kg or greater for 6 months associated with exposures (plasma AUC) similar to the clinical exposure. As the effect of tetrabenazine on prolactin levels is not known, the relevance of this finding is uncertain.6 Pharmaceutical Particulars
6.1 List of Excipients
Lactose, maize starch, sodium starch glycollate type A, purified talc, magnesium stearate, iron oxide yellow, and colloidal anhydrous silica.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Storage conditions.
Store below 25°C. Store in original container. Discard 4 weeks after opening.
6.5 Nature and Contents of Container
Container type.
White round high-density polyethylene (HDPE) tablet container with a child-resistant, tamper-evident polypropylene (PP) screw cap with mounted desiccant containing 112 tablets.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
Tetrabenazine is white to slightly yellow crystalline powder. Tetrabenazine is soluble in hot water, but practically insoluble in acetone. UV max (alcohol): 230, 284 nanometre (ε 7780, 3820).
CAS number.
58-46-8.
Chemical name.
1,3,4,6,7,11b-Hexahydro-9,10-dimethoxy-3-(2-methylpropyl)- 2H-benzo-[a]quinolizin-2-one.
Chemical structure.

7 Medicine Schedule (Poisons Standard)
Prescription only medicine - Schedule 4 (S4).
Summary Table of Changes


 Neuroleptic malignant syndrome (NMS) associated with the use of tetrabenazine has been reported rarely. This may occur soon after initiation of therapy, following an increase in dosage or after prolonged treatment. The main symptoms are mental changes, rigidity, hyperthermia, autonomic dysfunction and elevated creatinine phosphokinase levels. If NMS is suspected tetrabenazine should be withdrawn and appropriate supportive therapy instituted; treatment with dantrolene and bromocriptine may be effective.
Neuroleptic malignant syndrome (NMS) associated with the use of tetrabenazine has been reported rarely. This may occur soon after initiation of therapy, following an increase in dosage or after prolonged treatment. The main symptoms are mental changes, rigidity, hyperthermia, autonomic dysfunction and elevated creatinine phosphokinase levels. If NMS is suspected tetrabenazine should be withdrawn and appropriate supportive therapy instituted; treatment with dantrolene and bromocriptine may be effective.
