1 Name of Medicine
Fluorouracil.
2 Qualitative and Quantitative Composition
Tolak 4% Once Daily cream contains fluorouracil (5-FU) 4% w/w (equivalent to 40 mg/g).
Excipients with known effect.
Methyl hydroxybenzoate, propyl hydroxybenzoate, and, arachis oil (peanut oil).
For the full list of excipients, see Section 6.1 List of Excipients.3 Pharmaceutical Form
Cream, white to off-white in appearance.
4.1 Therapeutic Indications
Tolak 4% Once Daily is indicated for the topical treatment of actinic keratosis (AK) of the face, ears, and/or scalp in adults.
4.2 Dose and Method of Administration
Tolak 4% Once Daily should be applied once daily in an amount sufficient to cover the whole area of the face and/or ears and/or scalp where AK lesions have been identified with a thin film, using the fingertips to gently massage the medication uniformly into the skin.
Duration of treatment.
Apply Tolak 4% Once Daily for a period of 4 weeks as tolerated.
The development of an inflammatory response is associated with the pharmacological action of 5-FU on dysplastic AK cells. This is characterised by local skin reactions that peaks at 4 weeks of treatment and resolves within 2-4 weeks after the end of treatment (see Section 4.4 Special Warnings and Precautions for Use; Section 4.8 Adverse Effects (Undesirable Effects) for normal pattern of response).
In case of severe discomfort during treatment or where skin reactions last more than 4 weeks, symptomatic treatment (such as emollients or topical corticosteroids) should be offered.
The number of retreatments with Tolak 4% Once Daily is the decision of the treating physician. When assessing options to treat recurrent lesions, the physician should consider that repeated treatment with Tolak 4% Once Daily in case of recurrence has not been formally assessed. The duration between initial treatment and retreatment with Tolak 4% Once Daily in clinical studies ranged between 7 and 13 months (mean: 9.4 months). The number of retreatments with Tolak 4% Once Daily is the decision of the treating physician (see Section 5.1 Pharmacodynamic Properties, Clinical trials).
Evaluation of the therapeutic effect can be assessed approximately 4 weeks after end of treatment.
Method of administration.
Treatment areas should be washed, rinsed and dried prior to applying Tolak 4% Once Daily.
Thoroughly wash hands after applying Tolak 4% Once Daily (see Section 4.4 Special Warnings and Precautions for Use).
Special populations.
Paediatric population.
No data are available in the paediatric population since children do not have actinic keratosis.
Elderly population.
No dedicated studies in elderly patients have been conducted. No dose adjustment is required for elderly patients (65 years and older) based on the results of the clinical studies (see Section 5.1 Pharmacodynamic Properties).
Hepatic and renal impairment.
No dosage adjustment is required for patients with hepatic or renal impairment.4.3 Contraindications
Tolak 4% Once Daily is contraindicated:
In patients with known hypersensitivity to the active substance or to any of the excipients, see Section 6.1 List of Excipients.
In patients with allergy to peanut or soya (see Section 6.1 List of Excipients).
During pregnancy (see Section 4.6 Fertility, Pregnancy and Lactation).
During breastfeeding (see Section 4.6 Fertility, Pregnancy and Lactation).
In coadministration with brivudine, sorivudine and analogues as they may lead to a substantial increase in plasma levels of 5-FU and associated toxicity. The antiviral nucleoside drugs brivudine and sorivudine are potent inhibitors of dihydropyrimidine dehydrogenase (DPD), a 5-FU metabolising enzyme (see Section 4.4 Special Warnings and Precautions for Use; Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
In patients with dihydropyrimidine dehydrogenase (DPD) deficiency (see Section 4.4 Special Warnings and Precautions for Use).
4.4 Special Warnings and Precautions for Use
Do not apply Tolak 4% Once Daily directly into eyes, nose, mouth, or other mucous membranes because irritation, local inflammation and ulceration can occur. Patients should wash hands well after applying Tolak 4% Once Daily.
Tolak 4% Once Daily should not be applied to open wounds or damaged skin where the skin barrier is compromised.
The normal pattern of response includes: an early inflammatory phase (typically characterised by erythema, which may become intense and blotchy); an apoptotic phase (characterised by skin erosion); and finally healing (when epithelialisation occurs).
The clinical manifestation of response usually occurs in the second week of treatment. However, these effects can sometimes be more severe (see Section 4.8 Adverse Effects (Undesirable Effects)). In case of severe discomfort during treatment or for skin reactions lasting more than 4 weeks, symptomatic treatment (such as emollients or topical corticosteroids) should be offered (see Section 4.2 Dose and Method of Administration).
Occlusive dressings may increase inflammatory skin reactions.
Ophthalmic adverse reactions.
Corneal and conjunctival disorders have occurred with topical 5-FU. Avoid applying to the periocular area. To avoid transfer of the drug into the eyes and/or contact lenses and to the periocular area during and after application, patients should wash hands well after applying Tolak 4% Once Daily. If accidental exposure occurs, patients should flush eye(s) with large amounts of water.
Hypersensitivity reactions.
Allergic contact dermatitis (delayed type hypersensitivity reaction) has been observed with topical 5-FU preparations. Delayed type hypersensitivity should be suspected in the event of severe pruritus or eczema at the application site or at a distant site.
Although the potential for a delayed hypersensitivity reaction to 5-FU exists, patch testing to confirm hypersensitivity may be inconclusive.
Tolak 4% Once Daily contains peanut oil. The peanut oil used in Tolak 4% Once Daily is tested for residual protein through amino acid analysis; the acceptance criterion for total protein is no more than 0.5 parts per million. Physicians should use caution in prescribing Tolak 4% Once Daily for peanut-sensitive individuals (see Section 4.3 Contraindications).
Tolak 4% Once Daily contains:
Methyl hydroxybenzoate and propyl hydroxybenzoate which may cause allergic reactions (possibly delayed).
Butylated hydroxytoluene which may cause local skin reactions (e.g. contact dermatitis), or irritation to the eyes and mucous membranes.
Cetyl alcohol and stearyl alcohol which may cause local skin reactions (e.g. contact dermatitis).
Photosensitivity.
Topical 5-FU is associated with photosensitivity reactions. Exposure to ultraviolet rays including sunlight, sun lamps, and tanning beds should be avoided during treatment with Tolak 4% Once Daily.
Dihydropyrimidine dehydrogenase (DPD) deficiency.
Tolak 4% Once Daily must not be used on patients with DPD deficiency (see Section 4.3 Contraindications). Life-threatening systemic toxicity has been reported with the topical use of fluorouracil in a patient with DPD deficiency. Symptoms of serious side effects included severe stomach-area abdominal pain, bloody diarrhea, vomiting, fever, and chills. Physical examination revealed stomatitis, erythematous skin rash, neutropenia, thrombocytopenia, and inflammation of the esophagus, stomach and small bowel.
A large percentage of fluorouracil is catabolized by the DPD enzyme. DPD enzyme deficiency may result in increased availability of fluorouracil to the anabolic pathway, which may lead to increased interference with DNA and RNA synthesis and increased cytotoxic activity and potential toxicities. Therefore, Tolak 4% Once Daily is contraindicated in patients with DPD deficiency.
Patients should discontinue Tolak 4% Once Daily if symptoms of 5-FU systemic toxicity develop (see Section 4.9 Overdose).4.5 Interactions with Other Medicines and Other Forms of Interactions
Systemic concentrations of fluorouracil after topical dermal administration of Tolak 4% Once Daily appear to be low and as such are considered unlikely under normal circumstances to significantly influence the pharmacokinetics of other concomitantly administered systemic drugs (see Section 5.2 Pharmacokinetic Properties). Because life-threatening systemic toxicity has been reported with the topical use of fluorouracil in a patient with DPD deficiency (see Section 4.4 Special Warnings and Precautions for Use), concomitant use of substances that decrease the activity of DPD (such as capecitabine, brivudine, sorivudine, and interferon-α) could result in a pronounced increase in systemic fluorouracil concentrations and associated toxicity and is therefore not advised (see Section 4.4 Special Warnings and Precautions for Use; Section 5.2 Pharmacokinetic Properties).
The concomitant use of brivudine and sorivudine with Tolak 4% Once Daily is contraindicated (see Section 4.3 Contraindications). Treatment with the antiviral nucleoside analogues brivudine or sorivudine and topical application of Tolak 4% Once Daily should be separated by an interval of at least four weeks.
Systemic administration of fluorouracil or its prodrugs has led to signs of increased activity and adverse effects of CYP2C9 substrates such as warfarin and phenytoin. Bleeding complications have been observed upon concomitant administration with warfarin, and symptoms of phenytoin intoxication have been observed upon concomitant administration with phenytoin. The possibility of drug interactions between Tolak 4% Once Daily and CYP2C9 substrates, particularly those with a narrow therapeutic index, should be considered.
Systemic interactions between Tolak 4% Once Daily and other concomitantly administered systemic drugs may be more pronounced in patients with DPD deficiency. When DPD deficiency is suspected, the possibility of drug interactions between systemically absorbed fluorouracil and the concomitant medicines should be considered. The possibility of DPD deficiency when drug interactions are suspected should also be considered.
Concomitant administration of leucovorin may enhance both the activity and toxicity of fluorouracil by increasing the inhibition by fluorouracil of the enzyme thymidylate synthase. The relevance of this activity to the topical dermal use of Tolak 4% Once Daily in the treatment of actinic keratosis lesions is likely to be minimal due to the low systemic absorption of fluorouracil after topical dermal administration as Tolak 4% Once Daily.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility.
No clinical data in humans are available on the effects of topical 5-FU on fertility. Fertility studies with systemic 5-FU resulted in impairment of male fertility and in a reduction of pregnancy rates in female rodents. The use of topical 5-FU may impair female and male fertility. Topical 5-FU is not recommended in women and men attempting to have a child.
(Category D)
There are no adequate data from the use of topical 5-FU in pregnant women. Systemic administration of high doses of 5-FU have teratogenic and/or embryotoxic effects in mice, rats, hamsters and monkeys.
Cases of miscarriage and birth defects (including cleft lip and cleft palate) have been reported when pregnant women were exposed to a topical or parenteral fluorouracil product. In addition, ventricular septal defect and cases of miscarriage occurred when pregnant women applied a topical fluorouracil product to mucous membranes (Tolak 4% Once Daily is not indicated for use on the mucous membrane).
The potential risk for humans is unknown, hence Tolak 4% Once Daily must not be used during pregnancy (see Section 4.3 Contraindications).
Women of childbearing potential should not become pregnant during topical 5-FU therapy and should use effective method of contraception during treatment with 5-FU therapy. If a pregnancy occurs during treatment the patient should be advised about the risk for the child of adverse effects associated with the treatment and genetic counselling is recommended.
Contraception in males and females.
Due to the genotoxic potential of fluorouracil, women of childbearing potential should not become pregnant during topical 5-FU therapy and must use an effective method of contraception during treatment with 5-FU therapy and for 6 months following completion of treatment. Men must use an effective method of contraception and not father a child during treatment with 5-FU therapy and for 3 months following completion of treatment.
No information is available on the excretion of 5-FU into breast milk. As risk to nursing infants cannot be excluded, Tolak 4% Once Daily must not be used in nursing mothers (see Section 4.3 Contraindications). If use of Tolak 4% Once Daily is absolutely necessary, breastfeeding must be discontinued.4.7 Effects on Ability to Drive and Use Machines
It is unlikely that treatment with Tolak 4% Once Daily will have any effect on the ability to drive and use machines when used according to the dosage instructions.
4.8 Adverse Effects (Undesirable Effects)
Summary of safety profile.
The most frequently reported events in subjects treated with Tolak 4% Once Daily in the primary clinical studies were application site reactions. Tolerability at the application site was evaluated in the primary clinical studies. Local reactions related to tolerability, associated with the pharmacological action of 5-FU included erythema, scaling/dryness, oedema, crusting, erosions, stinging/burning, and pruritus with an incidence of 62% to 99% by symptom. Most of these local reactions were reported as mild (17% to 37% by symptom) or moderate (22% to 44% by symptom); severe reactions were less frequent (6% to 38% by symptom, with overall lower occurrence of severe reactions compared to active comparator (5% 5-FU cream). They were transient with a peak at 4 weeks of treatment and resolved within 2-4 weeks after the end of treatment (also see Section 4.4 Special Warnings and Precautions for Use for normal pattern of response). Application site reactions are presented in Table 1.
All other adverse effects (including insomnia, nasal discomfort, pharyngitis, nausea, periorbital oedema, impetigo, rash, and lip blister) were reported at a frequency below 1%.
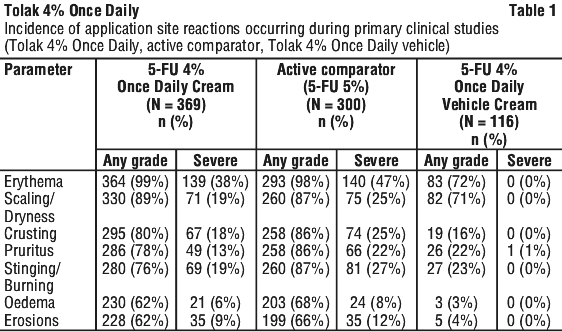 Table 2 lists the adverse reactions reported in AK patients treated with Tolak 4% Once Daily for 4 weeks during the primary clinical studies.
Table 2 lists the adverse reactions reported in AK patients treated with Tolak 4% Once Daily for 4 weeks during the primary clinical studies.
Their frequency is defined using the following convention: very common (≥ 1/10), common (≥ 1/100 to < 1/10); uncommon (≥ 1/1,000 to < 1/100); rare (≥ 1/10,000 to < 1/1,000); very rare (< 1/10,000).
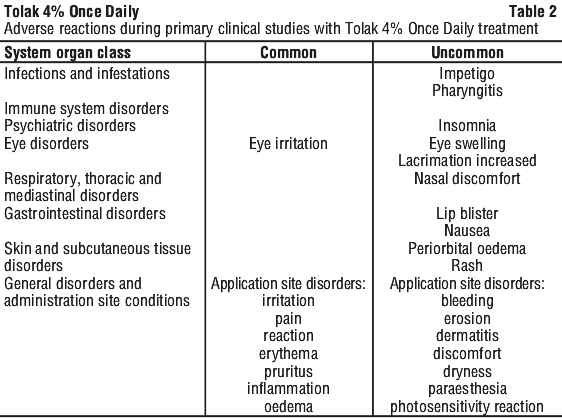
Description of selected adverse reactions.
Photosensitivity.
Topical 5-FU is associated with photosensitivity reactions including severe sunburn. Photosensitivity reaction was reported in one subject (0.3%) in the primary clinical studies of Tolak 4% Once Daily. It should be noted that photosensitivity reaction was also reported by a subject in the vehicle group.
Post-marketing experience.
Hypersensitivity reactions.
Although no case has been reported in the primary clinical trials of Tolak 4% Once Daily, allergic contact dermatitis (delayed type hypersensitivity reaction) has been reported with topical 5-FU drugs and with Tolak 4% Once Daily since first marketing approval.
Reporting suspected adverse effects.
Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.4.9 Overdose
When applied to the skin as recommended, systemic intoxication with 5-FU is unlikely. Application of significantly higher doses than recommended may result in an increase in the frequency and severity of reactions at the application site.
There is no known clinical case of accidental ingestion of Tolak 4% Once Daily, however if this occurs signs of 5-FU overdosage may include nausea, vomiting, diarrhea and stomatitis.
Blood dyscrasias may occur in severe cases. Daily white cell counts should be performed, and appropriate measures should be taken accordingly for the prevention of systemic infection.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Pharmacotherapeutic group: pyrimidine analogues. ATC code: L01BC02.
Mechanism of action.
The active substance fluorouracil (5-FU) is a cytostatic agent that has an antimetabolite effect. Due to its structural similarity with the thymine (5-methyluracil) occurring in nucleic acids, 5-FU prevents its formation and utilisation and, in this way, inhibits both DNA and RNA synthesis which results in growth inhibition.
Clinical trials.
The safety and the efficacy of Tolak 4% Once Daily were evaluated in two primary, multi-centre, randomised, controlled studies (Trial 1 and Trial 2) in subjects with at least 5 visible actinic keratosis lesions on the face, scalp, and/or ear (not exceeding 1 cm). Trial 1 compared Tolak 4% Once Daily to an active comparator (5-FU 5%) (twice daily) and a negative placebo control (vehicle). Trial 2 was a placebo-controlled study. Application of the medication, once daily for 4 weeks, involved field treatment of the whole area of the face and/or ears and/or scalp where actinic keratosis lesions were identified at baseline. A high proportion of patients in these studies applied Tolak 4% Once Daily cream on a large area of skin between 240 cm2 and 961 cm2. All efficacy endpoints were evaluated at 4 weeks post-treatment. Subjects were all Caucasian with a mean age of approximately 68 years (33-89 years). The mean number of actinic keratosis lesions was 14.4 and 16.2 (Trial 1) and 19.2 and 23.2 (Trial 2), in the Tolak 4% Once Daily group and the placebo group, respectively.
As shown in Table 3, in both trials, superiority of Tolak 4% Once Daily was demonstrated versus vehicle. In Trial 1, the difference in "100% complete clearance rate" of Tolak (5-FU 4%; cream once daily) (54.4%) minus active comparator (5-FU 5% cream; twice daily) (57.9%) was 3.5% with a lower 97.5% confidence limit of -11.11%. The difference in "75% complete clearance rate" of Tolak (80.5%) minus active comparator (80.2%) was 0.3% with a lower 97.5% confidence limit of -5.94% in the intention-to-treat population (with similar results in the per-protocol population).
 The safety of 4-week treatment Tolak 4% Once Daily was assessed up to 4 weeks post-treatment. The majority of the reported adverse reactions and local skin responses were mild to moderate in intensity and resolved without sequelae.
The safety of 4-week treatment Tolak 4% Once Daily was assessed up to 4 weeks post-treatment. The majority of the reported adverse reactions and local skin responses were mild to moderate in intensity and resolved without sequelae.
Long term efficacy - lesion recurrence.
After completing the two primary clinical studies, patients treated with Tolak 4% Once Daily were followed up for 12 months for lesion recurrence. Of the 184 patients included in the analysis of recurrence, 83 (45.1%) patients remained clear 12 months after treatment and 101 (54.9%) patients had a recurrence within 12 months.
The duration between initial treatment and retreatment with Tolak 4% Once Daily in clinical studies ranged between 7 and 13 months (mean: 9.4 months). There was no formal assessment of repeated treatment with Tolak 4% Once Daily in case of recurrence.
Paediatric population.
No data are available in paediatric patients.
Elderly population.
Of the 403 subjects treated with Tolak 4% Once Daily in the phase III clinical trials, 204 subjects were 68 years and older while 199 subjects were below 68 years of age. No overall differences in efficacy were observed between the two groups.
5.2 Pharmacokinetic Properties
5-FU shows low absorption following topical application. In a systemic absorption study of topically applied Tolak 4% Once Daily, 8 of 21 patients had undetectable (1 nanogram/mL) levels of 5-FU. Among patients with detectable plasma 5-FU levels, the highest level of plasma 5-FU was generally observed at 1-hour post-dose and the observed maximum concentration ranged between 1.1 - 7.4 nanogram/mL.
5-FU may be metabolised by catabolic or anabolic routes which are similar to those of endogenous uracil. The rate-limiting step in the metabolism of 5-FU is conversion to 5-6- dihydrofluorouracil by the enzyme DPD.
5.3 Preclinical Safety Data
Genotoxicity.
5-FU has no potential to induce point mutations in bacteria. 5-FU induced chromosome aberrations and/or micronuclei in vitro in several cell lines and was clastogenic after IP or oral administrations in mice and rats and after dermal application to mice.
Carcinogenicity.
No evidence of carcinogenicity was found in several studies in rats or mice after intravenous or oral administration.6 Pharmaceutical Particulars
6.1 List of Excipients
Tolak 4% Once Daily cream is for topical use only and contains fluorouracil (5-FU) 4% w/w (equivalent to 40 mg/g). The cream also contains the excipients: stearoyl macrogolglycerides, butylated hydroxytoluene, cetyl alcohol, citric acid, glycerol, isopropyl myristate, methyl gluceth-10, methyl hydroxybenzoate, propyl hydroxybenzoate, purified water, arachis oil (peanut oil), sodium hydroxide, stearic acid and stearyl alcohol.
6.2 Incompatibilities
Not applicable.
6.3 Shelf Life
In Australia, information on the shelf-life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
Shelf-life after first opening the immediate packaging: 4 weeks.
6.4 Special Precautions for Storage
Store below 25°C.
6.5 Nature and Contents of Container
Aluminium tube with membrane, internally coated with an epoxyphenolic lacquer, with a polypropylene piercing screw cap.
Pack sizes: 20 g and 40 g.
Not all pack sizes may be marketed.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
Chemical structure.

CAS number.
Chemical Abstracts Service (CAS) registry number: 51-21-8.
Chemical name: 5-Fluoropyrimidine-2,4(1H,3H)-dione.
Molecular formula: C4H3FN2O2. Relative molecular weight: 130.1.
5-FU is a white or almost white, crystalline powder. It is sparingly soluble in water, slightly soluble in alcohol, practically insoluble in chloroform. Its dissociation constants (pKa) are 8.0 and 13.0.7 Medicine Schedule (Poisons Standard)
Schedule 4 (Prescription Only Medicine).
Summary Table of Changes


 Table 2 lists the adverse reactions reported in AK patients treated with Tolak 4% Once Daily for 4 weeks during the primary clinical studies.
Table 2 lists the adverse reactions reported in AK patients treated with Tolak 4% Once Daily for 4 weeks during the primary clinical studies.
 The safety of 4-week treatment Tolak 4% Once Daily was assessed up to 4 weeks post-treatment. The majority of the reported adverse reactions and local skin responses were mild to moderate in intensity and resolved without sequelae.
The safety of 4-week treatment Tolak 4% Once Daily was assessed up to 4 weeks post-treatment. The majority of the reported adverse reactions and local skin responses were mild to moderate in intensity and resolved without sequelae.
