Notes
Distributed by Clinect Pty Ltd
1 Name of Medicine
Tranexamic acid.
2 Qualitative and Quantitative Composition
Each Tranexamic Acid Waymade tablet contains 500 mg of tranexamic acid.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Tranexamic Acid Waymade is available as film coated tablets. Tranexamic Acid Waymade is a white to off white, oblong shaped, film-coated biconvex tablet with a break-line and marked "TA" on one side of the break-line and the other side plain.
4.1 Therapeutic Indications
Oral administration.
Hereditary angioneurotic oedema.
Short term use in the treatment of hyphaema and in patients with established coagulopathies who are undergoing minor surgery.
Menorrhagia.4.2 Dose and Method of Administration
Oral administration.
Traumatic hyphaema.
1.0 to 1.5 g every 8 hours for six to seven days.
Menorrhagia.
Two tablets (1 g) four times a day, increasing to three tablets (1.5 g) four times a day if needed, for four days. Treatment should be initiated at the onset of visible bleeding, and continued for the first 4 days of the menstrual cycle. Patients should be assessed after three months of treatment.
No efficacy data are available from randomised, controlled clinical trials for treatment beyond three menstrual cycles.
Hereditary angioneurotic oedema.
Patients who can sense the onset of attacks are best treated intermittently with 2 - 3 tablets, 2 - 3 times a day until symptoms subside. Others should be treated continuously with the same dose.
Prostatectomy.
1 g orally six hours pre-operatively followed by 1 g orally 3 to 4 times a day until macroscopic haematuria is no longer present. Treatment beyond two weeks is not recommended.
Patients with established coagulopathies undergoing minor surgery conisation of the cervix.
1.0 to 1.5 g (2 to 3 tablets) every 8 to 12 hours for 12 days post-operatively.
Dental operations/extraction.
25 mg/kg is given orally two hours before operation. Factor VIII and Factor IX should be given as well as tranexamic acid. After the operation, 25 mg/kg of tranexamic acid is given 3 to 4 times a day for 6 to 8 days.
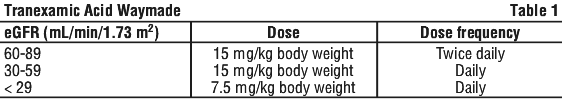
Dosage adjustment in renal impairment for orally administered Tranexamic Acid Waymade.
See Table 1.
Method of administration.
Orally administered.4.3 Contraindications
Patients with a history or risk of thrombosis should not be given Tranexamic Acid Waymade unless at the same time it is possible to give treatment with anticoagulants.
Active thromboembolic disease such as deep vein thrombosis (DVT), pulmonary embolism and cerebral thrombosis.
The preparation should not be given to patients with acquired disturbances of colour vision. If disturbances of colour vision arise during the course of treatment the administration of the preparation should be discontinued.
Patients with subarachnoid haemorrhage should not be given Tranexamic Acid Waymade as anecdotal experience indicates that cerebral oedema and cerebral infarction may be caused in such cases.
Hypersensitivity to Tranexamic Acid Waymade or any of its excipients.
4.4 Special Warnings and Precautions for Use
The dose of Tranexamic Acid Waymade should be reduced in patients with renal impairment because of the risk of accumulation (see Section 4.2 Dose and Method of Administration). Isolated cases of obstruction of the urinary tract due to blood clots have been observed when tranexamic acid has been used to treat severe bleeding from the upper urinary tract.
Tranexamic Acid Waymade therapy is not indicated in haematuria caused by diseases of the renal parenchyma. Intravascular precipitation of fibrin frequently occurs in these conditions and may aggravate the disease. In addition, in cases of massive renal haemorrhage of any cause, antifibrinolytic therapy carries the risk of clot retention in the renal pelvis.
Although clinical evidence shows no significant increase in thrombosis, possible risk of thrombotic complications cannot be ruled out. Venous and arterial thrombosis or thromboembolism has been reported in patients treated with tranexamic acid. In addition, cases of central retinal artery and central retinal vein obstruction have been reported. A few patients have developed intracranial thrombosis with tranexamic acid but further investigation is needed to assess the significance of this potential hazard.
Patients with a high risk for thrombosis (a previous thromboembolic event and a family history of thromboembolic disease) should use Tranexamic Acid Waymade only if there is a strong medical indication and under strict medical supervision. The risk for thromboembolic events may be increased in patients using hormonal contraceptives. If Tranexamic Acid Waymade has to be used in these patients, advise them to use an effective alternative (nonhormonal) contraceptive method.
Tranexamic Acid Waymade should not be administered concomitantly with Factor IX Complex Concentrates or Anti-inhibitor Coagulant Concentrates, as the risk of thrombosis may be increased.
Blood in body cavities such as pleural space, joint spaces and urinary tract (e.g. renal, pelvis, bladder) may develop 'indissoluble clots' in these cavities due to extravascular blood clots which may be resistant to physiological fibrinolysis.
Patients with irregular menstrual bleeding should not use Tranexamic Acid Waymade until the cause of the irregularity has been established. If menstrual bleeding is not adequately reduced by Tranexamic Acid Waymade, an alternative treatment should be considered.
There are no data on the use of tranexamic acid in women taking oral contraceptive agents.
Patients with disseminated intravascular coagulation (DIC) who require treatment with Tranexamic Acid Waymade must be under the strict supervision of a physician experienced in treating this disorder.
Focal areas of retinal degeneration have developed in cats, dogs and rats following oral or intravenous tranexamic acid at doses between 250 to 1600 mg/kg/day (6 to 40 times the recommended usual human dose) from 6 days to 1 year. The incidence of such lesions has varied from 25% to 100% of animals treated and was dose related. At lower doses some lesions appeared to be reversible.
Limited data in cats and rabbits showed retinal changes in some animals with doses as low as 126 mg/kg/day (about 3 times the recommended human dose) administered for several days to two weeks.
No retinal changes have been reported or noted in eye examinations in patients treated with tranexamic acid for weeks to months in clinical trials. However, visual abnormalities, often poorly characterised, represent the most frequently reported postmarketing adverse event in Sweden. For patients who are to be treated continually for longer than several days, an ophthalmological examination, including visual acuity, colour vision, eye-ground and visual fields, is advised before commencing and at regular intervals during the course of treatment. Tranexamic Acid Waymade should be discontinued if changes in examination results are found.
Convulsions have been reported in association with tranexamic acid treatment.
Use in hepatic impairment.
Pharmacokinetic data from patients with pre-existing hepatic impairment, who were treated with tranexamic acid, are not available. As tranexamic acid is excreted unchanged, dose adjustment due to hepatic impairment is not required.
Use in renal impairment.
Patients with impaired renal function may experience an increased elimination half life for the drug. The need for dose reduction is recommended in adult patients with renal impairment.
Dose reduction is recommended in children ≥ 2 years old who are mildly or moderately renally impaired. Tranexamic Acid Waymade is not recommended in children who are severely impaired (see Section 5.1 Pharmacodynamic Properties, Clinical trials; Section 4.2 Dose and Method of Administration).
For both the adult and the paediatric patient, an eGFR ≥ 90 mL/min/1.73 m2 usually indicates kidney function within a 'normal range', but does not exclude patients with early kidney damage. If renal impairment is suspected, informed dose alterations decision may include other estimates of renal function including consultation with an experienced renal physician.
Use in the elderly.
No reduction in dosage is necessary unless there is evidence of renal failure.
Paediatric use.
Clinical experience with tranexamic acid in menorrhagic females under 15 years of age is not available.
Effects on laboratory tests.
No data available.4.5 Interactions with Other Medicines and Other Forms of Interactions
Clinically important interactions have not been observed with tranexamic acid tablets. There are no specific drug-drug interactions data for tranexamic acid. Because of the absence of interaction studies, simultaneous treatment with anticoagulants must take place under the strict supervision of a physician experienced in this field.
Avoid concomitant use of Tranexamic Acid Waymade with medical products that are prothrombotic because concomitant use can further increase the risk of thromboembolic adverse reactions associated with tranexamic acid (see Section 4.4 Special Warnings and Precautions for Use).
4.6 Fertility, Pregnancy and Lactation
Effects on fertility.
Fertility was not affected in male or female rats at high oral doses up to 850 - 880 mg/kg/day (~ 1.7 times the maximum recommended daily human oral dose on a body surface area basis).
There are no clinical data in humans supporting the impact of tranexamic acid on fertility.
(Category B1)
Drugs which have been taken by only a limited number or pregnant women and women of childbearing age, without an increase in the frequency of malformation or other direct or indirect harmful effects on the human foetus having been observed.
Available data from published studies, case series and case reports with tranexamic acid use in pregnant women in the second and third trimester and at the time of delivery have not clarified whether there is a drug-associated risk of miscarriage or adverse maternal or fetal outcomes. There are cases of fetal structural abnormalities that resulted in death of the newborn following administration of tranexamic acid to the mother during conception or the first trimester of pregnancy; however, due to other confounding factors the risk of major birth defects with use of Tranexamic Acid Waymade during pregnancy is not clear.
The estimated background risk for major birth defects and miscarriage for the indicated human population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes.
It is not known whether Tranexamic Acid Waymade use in pregnant women may cause a drug-associated risk of miscarriage or adverse maternal or fetal outcomes. For decisions regarding the use of Tranexamic Acid Waymade during pregnancy, the potential risk of Tranexamic Acid Waymade administration on the fetus should always be considered along with the mother's clinical need for Tranexamic Acid Waymade; an accurate risk-benefit evaluation should drive the treating physician's decision.
Tranexamic acid passes through the placenta. The concentration in cord blood after an intravenous injection of 10 mg/kg to pregnant women is about 30 mg/L, as high as in the maternal blood.
There were 13 clinical studies that described fetal and/or neonatal functional issues such as low Apgar score, neonatal sepsis, cephalohematoma and 9 clinical studies that discussed alterations to growth including low birth weight and preterm birth at 22-36 weeks of gestation in fetuses and infants exposed to tranexamic acid in utero.
Studies in rats (at up to 1000 mg/kg/day PO; 2 times the maximum recommended daily human oral dose on a body surface area basis) showed no effects of tranexamic acid on embryonic or neonatal development. In rabbits, increased fetal losses and lower litter weights were noted at 200 mg/kg IV and 400 mg/kg PO (1.6 times the maximum recommended daily human oral dose on a body surface area basis) (but not at 100 mg/kg IV or 200 mg/kg PO). There was no effect on rat or rabbit young survival (including one IV teratology study in rabbits at 50-200 mg/kg).
Published literature reports the presence of tranexamic acid in human milk. There are no data on the effects of tranexamic acid on the breastfed child or the effects on milk production. The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for Tranexamic Acid Waymade and any potential adverse effects on the breastfed child from Tranexamic Acid Waymade or from the underlying maternal condition.4.7 Effects on Ability to Drive and Use Machines
Tranexamic Acid Waymade may cause dizziness and therefore may influence the ability to drive or use machines.
4.8 Adverse Effects (Undesirable Effects)
Oral administration.
Gastrointestinal discomfort occurs in more than 30% of patients after oral administration of 6 g/day. The discomfort disappears when the dose is reduced.
Common side effects (≥ 1 to < 10%). Gastrointestinal disorders.
Nausea, vomiting, diarrhoea.
Uncommon side effects (≥ 0.1 to < 1%). Immune system disorders.
Dermatitis allergic.
Post-marketing experience.
Immune system disorders.
Hypersensitivity including anaphylactic reaction.
Central nervous system disorders.
Convulsion, dizziness.
Eye disorders.
Chromatopsia, visual impairment.
Vascular disorders.
Embolism.
Reporting suspected adverse effects.
Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.4.9 Overdose
Overdose data are limited. There is one report of overdosage in which a seventeen year old ingested 37 g of tranexamic acid and after receiving treatment with gastric lavage, mild intoxication was reported.
Symptoms of overdose may include dizziness, headache, nausea, vomiting, diarrhoea, orthostatic symptoms, hypotension and convulsions.
There is no known antidote for Tranexamic Acid Waymade overdose. In the event of overdose, the patient should be treated symptomatically and supportive measures should be instituted as required.
Activated charcoal may reduce absorption of tranexamic acid if given within one or two hours after ingestion. In patients who are not fully conscious or have impaired gag reflex, consideration should be given to administering activated charcoal via a nasogastric tube once the airway is protected.
In addition to this, monitor vital signs to detect a possible hypotensive episode. Monitor fluid and electrolyte status in patients with severe vomiting or diarrhoea and administer IV fluids and replace electrolytes as necessary. Monitor urine output and maintain adequate diuresis. Monitor for clinical evidence of thromboembolic complications (e.g. chest pain, shortness of breath, flank pain, extremity pain). Because there is a risk of thrombosis in predisposed individuals, anticoagulant therapy should be considered in these patients.
In symptomatic patients, support respiratory and cardiac function. Monitor blood count, renal function, pulse oximetry and/or blood gases and obtain a chest x-ray. Obtain an ECG and institute continuous cardiac monitoring.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action.
Tranexamic acid is a competitive inhibitor of plasminogen activation and at much higher concentrations a noncompetitive inhibitor of plasmin, thus implying that tranexamic acid interferes with the fibrinolytic process in the same way as aminocaproic acid. Tranexamic acid is about 10 times more potent in vitro than aminocaproic acid.
Tranexamic acid binds more strongly than aminocaproic acid to both the strong and weak sites of the plasminogen molecule in a ratio corresponding to the difference in potency between the compounds.
Tranexamic acid in a concentration of 1 mg/mL does not aggregate platelets in vitro. Tranexamic acid in concentrations up to 10 mg/mL blood has no influence on the platelet count, the coagulation time or various coagulation factors in whole blood or citrated blood in normal subjects. On the other hand tranexamic acid in concentrations of 1 mg/mL and 10 mg/mL blood prolongs the thrombin time.
Clinical trials.
No data available.
5.2 Pharmacokinetic Properties
Absorption.
Absorption from the gastrointestinal tract is only about 50% at reasonably low oral doses. However, a parallel intake of food has no effect on the gastrointestinal absorption of the drug following a dose of 2 g or on the maximum plasma concentration.
Distribution.
Tranexamic acid does not bind to serum albumin. The plasma protein binding is about 3% at therapeutic plasma levels and seems to be fully accounted for by its binding to plasminogen.
Three hours after a single oral dose of 25 mg/kg, the peak serum level was 15.4 g/L and the aqueous humour level was 1.6 g/L. The plasma peak level after 1 g orally is 8 mg/L and after 2 g, 15 mg/L, both obtained three hours after dosing.
When administered 36 to 48 hours before surgery in 4 doses of 10 to 20 mg/kg, an antifibrinolytically active concentration (10 microgram/mL) of tranexamic acid remains in different tissues for about 17 hours and in the serum for up to seven or eight hours.
Tranexamic acid passes through to the placenta. The concentration in cord blood after an intravenous injection of 10 mg/kg to women could be fairly high, about 30 microgram/mL of foetal serum.
The concentration in breast milk is about one hundredth of the serum peak concentration obtained.
Tranexamic acid passes to semen and inhibits its fibrinolytic activity but does not influence the sperm migration.
Tranexamic acid crosses the blood-brain barrier.
Tranexamic acid concentration in cerebrospinal fluid is about one tenth that of plasma. The drug passes into the aqueous humour, the concentration being about one tenth of the plasma concentration.
Tranexamic acid diffuses rapidly to the joint fluid and the synovial membrane, and in the joint fluid the same concentration is obtained as in the serum. The biological half-life in the joint fluid is about three hours.
Metabolism.
Only a small fraction of the drug is metabolised. The total amount of metabolites excreted in urine during 72 hours is less than 5%. Possible routes of biotransformation are acetylation or deamination followed by oxidation or reduction. After oral administration approximately 50% of the parent compound, 2% of the deaminated dicarboxylic acid and 0.5% of the acetylated product are excreted.
Excretion.
After an intravenous dose of 1 g, the plasma concentration time curve shows a triexponential decay with a half life of about 2 hours for the terminal elimination phase. The initial volume of distribution is about 9 to 12 litres.
Urinary excretion is the main route of elimination via glomerular filtration. Overall renal clearance is equal to overall plasma clearance (110 to 116 mL/min) and more than 95% of the dose is excreted in urine as the unchanged drug. Excretion of tranexamic acid by glomerular filtration is about 90% at 24 hours after intravenous administration of 10 mg/kg bodyweight.
After oral administration of 10 to 15 mg/kg body weight the urinary excretion at 24 hours is 39% and at 48 hours is 41% of the ingested dose or 78% of the absorbed material.
Special populations.
Renal impairment. Adults.
Tranexamic acid is eliminated unchanged in urine. Patients with impaired renal function may experience an increased elimination half life for the drug. Immediately after a dose of tranexamic acid was given, plasma levels of tranexamic acid were similar in all cardiac surgery patients. This reflects distribution into body fluid. A linear increase in plasma levels was observed with decreasing renal function (increasing serum creatinine levels) at 24 hours, confirming the need for dose reduction in renally impaired patients (see Section 4.2 Dose and Method of Administration).
Hepatic impairment. Pharmacokinetic data from patients with pre-existing hepatic impairment, who were treated with tranexamic acid, are not available. As tranexamic acid is excreted unchanged, dose adjustment due to hepatic impairment is not required.
5.3 Preclinical Safety Data
Genotoxicity.
Tranexamic acid was not mutagenic in B. subtilis and had no chromosomal effects in Chinese hamster cells. The incidence of chromosomal breakage was increased at 3 g/kg in rat bone marrow. No lethal mutagenicity was detected in a dominant lethal test at 100 mg/kg and 3 g/kg. The weight of evidence in a limited range of mutagenicity tests suggests that tranexamic acid is not mutagenic.
Carcinogenicity.
A dietary carcinogenicity study in Shermann-Wyckoff rats showed an increase in the incidence of biliary hyperplasia, cholangioma and adenocarcinoma of the liver at high doses. However, these findings have not been reproduced in a number of other lifetime studies in either SD or CDF1 mice. A possible treatment-related increase in the incidence of leukaemia was noted in mice receiving dietary tranexamic acid at doses equivalent to up to 5 g/kg/day for 20 months.6 Pharmaceutical Particulars
6.1 List of Excipients
Each Tranexamic Acid Waymade tablet contains the following inactive ingredients: microcrystalline cellulose, purified talc, magnesium stearate, colloidal anhydrous silica, povidone, croscarmellose sodium, titanium dioxide, macrogol 8000, vanillin and the proprietary ingredient, Eudragit E100 (ID Number 1753).
6.2 Incompatibilities
No data available.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 25°C. Protect from light.
6.5 Nature and Contents of Container
Tranexamic Acid Waymade Tablets are available in a PVC/aluminium blister packs of 100 tablets.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of in accordance with local requirements.
6.7 Physicochemical Properties
Tranexamic acid is a white crystalline powder that is odourless or almost odourless. It is freely soluble in water and in glacial acetic acid, practically insoluble in methanol, ethanol, acetone, diethyl ether and benzene.
The pKa: 4.3 and 10.6.
Chemical structure.
The chemical structure of tranexamic acid is:
 Chemical name: trans-4-aminomethylcyclohexane-carboxylic acid.
Chemical name: trans-4-aminomethylcyclohexane-carboxylic acid.
The molecular formula of tranexamic acid is C8H15NO2 and its molecular weight is 157.2.
CAS number.
1197-18-8.7 Medicine Schedule (Poisons Standard)
Prescription Only Medicine (S4).
Summary Table of Changes



 Chemical name: trans-4-aminomethylcyclohexane-carboxylic acid.
Chemical name: trans-4-aminomethylcyclohexane-carboxylic acid.