1 Name of Medicine
Treosulfan.
2 Qualitative and Quantitative Composition
Trecondi 1 g powder for solution for infusion.
One vial of powder contains 1 g of treosulfan.
Trecondi 5 g powder for solution for infusion.
One vial of powder contains 5 g of treosulfan.
When reconstituted according to Section 4.2 Dose and Method of Administration, 1 mL of the solution for infusion contains 50 mg treosulfan.
For the full list of excipients, see Section 6.1 List of Excipients.3 Pharmaceutical Form
Powder for solution for infusion.
White crystalline powder.
4.1 Therapeutic Indications
Adults with acute myeloid leukaemia (AML) or myelodysplastic syndrome (MDS).
Trecondi (treosulfan) is indicated in combination with fludarabine as part of conditioning treatment prior to allogeneic hematopoietic stem cell transplantation (alloHSCT) in adult patients with AML or MDS at increased risk for standard conditioning therapies.
Paediatric patients aged 1 month and older with malignant and non-malignant haematological diseases.
Trecondi (treosulfan) is indicated in combination with fludarabine, with or without thiotepa, as part of conditioning treatment prior to allogeneic haematopoietic stem cell transplantation (alloHSCT) in paediatric patients older than one month with malignant and non-malignant diseases.4.2 Dose and Method of Administration
Administration of treosulfan should be supervised by a physician experienced in conditioning treatment followed by alloHSCT.
Dosage.
Adults with AML or MDS.
Treosulfan is given in combination with fludarabine.
The recommended dose and schedule of administration is:
Treosulfan 10 g/m2 body surface area (BSA) per day as a two-hour intravenous infusion, given on three consecutive days (day -4, -3, -2) before stem cell infusion (day 0). The total treosulfan dose is 30 g/m2;
Fludarabine 30 mg/m2 BSA per day as a 0.5-hour intravenous infusion, given on five consecutive days (day -6, -5, -4, -3, -2) before stem cell infusion (day 0). The total fludarabine dose is 150 mg/m2;
Treosulfan should be administered before fludarabine on days -4, -3, -2 (FT10 regimen).
Dosage adjustment.
Paediatric population aged 1 month and older with malignant and non-malignant haematological diseases.
Treosulfan is given in combination with fludarabine, with thiotepa (intensified regimen; FT10-14TT regimen) or without thiotepa (FT10-14 regimen).
The recommended dose and schedule of administration is:
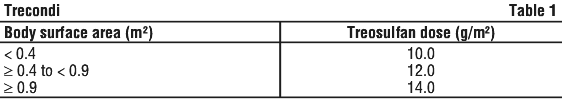
Treosulfan 10-14 g/m2 body surface area (BSA) per day as a two-hour intravenous infusion, given on three consecutive days (day -6, -5, -4) before stem cell infusion (day 0). The total treosulfan dose is 30-42 g/m2;
The dose of treosulfan should be adapted to the patient's BSA as follows (see Section 5.2 Pharmacokinetic Properties) (see Table 1):
 Fludarabine 30 mg/m2 BSA per day as a 0.5-hour intravenous infusion, given on five consecutive days (day -7, -6, -5, -4, -3) before stem cell infusion (day 0). The total fludarabine dose is 150 mg/m2;
Fludarabine 30 mg/m2 BSA per day as a 0.5-hour intravenous infusion, given on five consecutive days (day -7, -6, -5, -4, -3) before stem cell infusion (day 0). The total fludarabine dose is 150 mg/m2;
Treosulfan should be administered before fludarabine;
Thiotepa (intensified regimen 5 mg/kg twice a day), given as two intravenous infusions over 2-4 hours on day -2 before stem cell infusion (day 0).
The safety and efficacy of treosulfan in children less than 1 month of age has not yet been established.
Elderly.
No dose adjustment is necessary in any subset of the elderly population.
Renal and hepatic impairment.
No dose adjustment is necessary for mild or moderate impairment, but treosulfan is contraindicated in patients with severe impairment (see Section 4.3 Contraindications).
Method of administration.
Treosulfan is for intravenous use as a two-hour infusion.
Precautions to be taken before handling or administering the medicinal product.
When handling treosulfan, inhalation, skin contact or contact with mucous membranes should be avoided. Pregnant personnel should be excluded from handling cytotoxics.
Intravenous administration should be performed using a safe technique to avoid extravasation (see Section 4.4 Special Warnings and Precautions for Use).
Instructions on reconstitution of the medicine before administration.
As with all cytotoxic substances, appropriate precautions should be taken when handling treosulfan.
Trained personnel should reconstitute the medicinal product. When handling treosulfan, inhalation, skin contact or contact with mucous membranes should be avoided (the use of adequate protective disposable gloves, goggles, gown and mask is recommended). Contaminated body parts should be carefully rinsed with water and soap, the eyes should be rinsed with sodium chloride 9 mg/mL (0.9%) solution. If possible it is recommended to work on a special safety workbench, equipped with laminar flow, with liquid-impermeable, absorbent disposable foil. Adequate care and precautions should be taken in the disposal of items (syringes, needles, etc.) used to reconstitute cytotoxic medicinal products. Use Luer-lock fittings on all syringes and sets. Large bore needles are recommended to minimise pressure and the possible formation of aerosols. The latter may also be reduced by the use of a venting needle.
Pregnant personnel should be excluded from handling cytotoxics.
Instructions for reconstitution of treosulfan:
1. Treosulfan is reconstituted in its original glass container. Reconstituted solutions of treosulfan may be combined into a larger glass vial, EVA bag or PE bag. Do not dilute reconstituted product.
2. To avoid solubility problems, warm the solvent, sodium chloride 4.5 mg/mL (0.45%) solution, to 25°C - 30°C (not higher), for example by using a water bath.
3. Remove the treosulfan powder carefully from the inner surface of the vial by shaking. This procedure is very important, because moistening of powder that sticks to the surface results in caking. If this happens, vigorously shake the vial to redissolve the cake.
4. Reconstitute each vial of Trecondi containing 1 g treosulfan in 20 mL of pre-warmed (maximum 30°C) sodium chloride 4.5 mg/mL (0.45%) solution by shaking.
Reconstitute each vial of Trecondi containing 5 g treosulfan in 100 mL of pre-warmed (maximum 30°C) sodium chloride 4.5 mg/mL (0.45%) solution by shaking.
For preparation of sodium chloride 4.5 mg/mL (0.45%) solution equivalent volumes of sodium chloride 9 mg/mL (0.9%) solution and water for injections can be mixed.
The reconstituted solution contains 50 mg treosulfan per mL and appears as a clear colourless solution. Solutions showing any sign of precipitation should not be used.4.3 Contraindications
Hypersensitivity to the active substance.
Active non-controlled infectious disease.
Severe concomitant cardiac, lung, liver, and renal impairment.
Fanconi anaemia and other DNA breakage repair disorders.
Pregnancy and lactation (see Section 4.6 Fertility, Pregnancy and Lactation).
Administration of live vaccine.
4.4 Special Warnings and Precautions for Use
Myelosuppression.
Profound myelosuppression with pancytopenia is the desired therapeutic effect of treosulfan-based conditioning treatment, occurring in all patients. It is therefore recommended to monitor blood cell counts frequently until recovery of the haematopoietic system.
During phases of severe neutropenia (median duration of neutropenic period is 14-17.5 days in adults and 20-22 days in paediatric patients) the risk of infection is increased. Prophylactic or empiric anti-infective treatment (bacterial, viral, fungal) should therefore be considered. Growth factors (G-CSF, GM-CSF), platelet and/or red blood cell support should be given as indicated.
Secondary malignancies.
Secondary malignancies are well-established complications in long-term survivors after alloHSCT. How much treosulfan contributes to their occurrence is unknown. The possible risk of a second malignancy should be explained to the patient. On the basis of human data, treosulfan has been classified by the International Agency for Research on Cancer (IARC) as a human carcinogen.
Mucositis.
Oral mucositis (including high-grade severity) is a very common undesirable effect of treosulfan-based conditioning followed by alloHSCT (see Section 4.8 Adverse Effects (Undesirable Effects)). Use of mucositis prophylaxis (e.g. topical antimicrobials, barrier protectants, ice and adequate oral hygiene) is recommended.
Vaccines.
Concomitant use of live attenuated vaccines is not recommended.
Embryofetal toxicity and contraception.
Treosulfan can impair fertility (see Section 4.6 Fertility, Pregnancy and Lactation).
Ovarian suppression and amenorrhoea with menopausal symptoms commonly occur in pre-menopausal patients (see Section 4.6 Fertility, Pregnancy and Lactation).
Pregnant women should be informed of the potential risk to a fetus (see Section 4.6 Fertility, Pregnancy and Lactation). Females of reproductive potential should be advised to have a pregnancy test within 7 days prior to starting treatment with Trecondi and to use effective contraception during treatment and for at least 6 months after stopping treatment.
Males with female partners of reproductive potential should be advised to use effective contraception during treatment and for at least 6 months after the last dose of Trecondi.
Use in the elderly.
No data available.
Paediatric use.
Seizures.
There have been isolated reports of seizures in infants (≤ 4 months of age) with primary immunodeficiencies after conditioning treatment with treosulfan in combination with fludarabine or cyclophosphamide. Therefore, infants ≤ 4 months of age should be monitored for signs of neurological adverse reactions. Although it cannot be proved that treosulfan was the cause, the use of clonazepam prophylaxis for children younger than 1 year might be considered.
Respiratory, thoracic and mediastinal disorders.
There was a significant association between age and respiratory toxicity in paediatric patients treated with treosulfan-based conditioning.
Children younger than one year (mainly non-malignant diseases, especially immunodeficiencies) experienced more respiratory grade III/IV toxicity, possibly due to pulmonary infections already existing before the start of conditioning treatment.
Skin disorders.
An increase of skin disorders (e.g. rash, dermatitis) was observed when patients received sodium bicarbonate-containing hydration in the course of treosulfan infusion, potentially because of acceleration of the pH-dependent formation of alkylating epoxides.
Diaper dermatitis may occur in small children because of excretion of treosulfan in the urine. Therefore, nappies should be changed frequently up to 6-8 hours after each infusion of treosulfan.
Keep skin clean and dry on days of treosulfan infusion.
Effects on laboratory tests.
No data available.
Extravasation.
Treosulfan is considered an irritant. Intravenous application should be performed using a safe technique. If extravasation is suspected, general safety measures should be implemented. No specific measure has been proven to be recommendable.4.5 Interactions with Other Medicines and Other Forms of Interactions
No interaction of treosulfan was observed in high-dose chemotherapy.
In in vitro studies treosulfan did not inhibit CYP1A2, 2B6, 2C8, 2C9, 2D6, or 3A4 using testosterone as substrate. However, treosulfan was a reversible inhibitor of CYP2C19, and of CYP3A4 using midazolam as the substrate. Treosulfan did not inhibit substrate transport via various transport proteins at clinically relevant Cmax ratios, with the exception of P-glycoprotein (P-gp), which demonstrated potential for inhibition at ≥ 1.2-fold the clinical Cmax.
Detailed in vitro studies did not completely exclude potential interactions between high plasma concentrations of treosulfan and CYP3A4, CYP2C19, or P-gp substrates. Physiologically-based pharmacokinetic modelling predicted a weak (AUC ratio ≥ 1.25 and < 2) to moderate (AUC ratio ≥ 2 and < 5) interaction for CYP3A4, a weak interaction for CYP2C19, and a negligible (AUC ratio < 1.25) interaction for P-gp. Therefore, medicinal products with a narrow therapeutic index that are substrates for CYP3A4 or CYP2C19 should not be given during treatment with treosulfan.
Considering overall timing of treatments and the respective pharmacokinetic properties of concomitantly used medicinal products (e.g. half-life), the interaction potential can be reduced to "no interaction" (AUC ratio < 1.25), if all concomitantly used medicinal products are dosed 2 hours before or 8 hours after the 2-hour intravenous infusion of treosulfan.
The effect of treosulfan on the pharmacokinetics of fludarabine and thiotepa is not known.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility.
In common with other alkylating conditioning agents, treosulfan may impair fertility in men and women (see Section 4.4 Special Warnings and Precautions for Use).
Males of reproductive potential should seek advice on cryo-conservation of sperm prior to treatment with Trecondi because of the possibility of irreversible infertility due to therapy with treosulfan. Treosulfan can cause ovarian suppression and amenorrhoea with menopausal symptoms in premenopausal women.
Because of the potential risk to the fetus, both sexually active men and women of childbearing potential have to use effective contraception during and up to 6 months after treatment (see 'Use in pregnancy').
Specific reproductive toxicity studies with treosulfan in animals were not conducted. However, during chronic oral toxicity tests in rats, spermatogenesis and ovarian function were significantly affected. Data on impairment of fertility from the published literature includes reports of impaired spermatogenesis and uterine-ovarian and sperm development, and permanent reduction of reserve primordial ovarian follicles mice and rats treated with treosulfan or its active moieties.
(Category D)
There are no adequate or well-controlled studies with Trecondi in pregnant women.
Animal studies are insufficient with respect to developmental toxicity. Based on the cytotoxicity and genotoxicity of treosulfan and its active moieties, a potential for fetal toxicity and teratogenicity exists. Trecondi is contraindicated during pregnancy (see Section 4.3 Contraindications).
It is unknown whether treosulfan or its active moieties are excreted in human milk. No adequate or well-controlled studies have been conducted to assess the impact of Trecondi on milk production or its presence in breast milk. Since many drugs are excreted in human milk and because of the potential for serious adverse reactions in breastfeeding infants due to Trecondi, a risk to breast-fed children cannot be excluded. Breastfeeding should be discontinued during treatment with Trecondi and for at least 6 months after the last dose.4.7 Effects on Ability to Drive and Use Machines
Treosulfan has moderate influence on the ability to drive and use machines. It is likely that certain adverse reactions of treosulfan like nausea, vomiting or dizziness could affect these functions.
4.8 Adverse Effects (Undesirable Effects)
Summary of the safety profile.
Profound myelosuppression/pancytopenia is the desired therapeutic effect of conditioning therapy and occurs in all patients. Blood cell counts usually recover after HSCT.
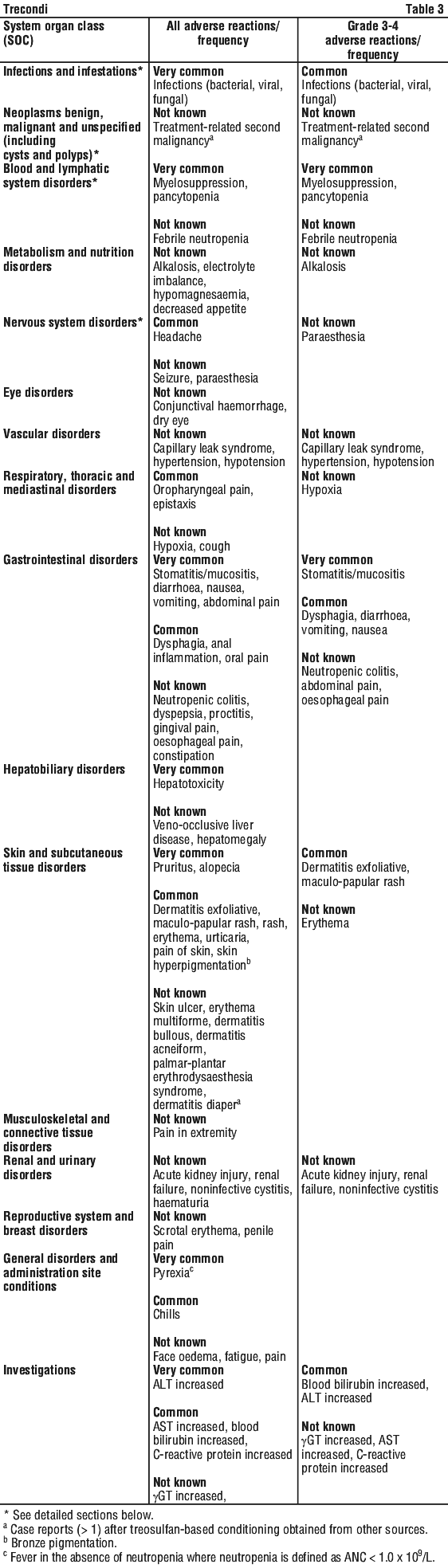
The most commonly observed adverse reactions (adults/ paediatric patients) after treosulfan-based conditioning followed by alloHSCT include overall infections (10.1% /11.6%), gastrointestinal disorders (nausea [38.0%/26.4%], stomatitis [36.4%/66.1%], vomiting [22.5%/42.1%], diarrhoea [14.4%/33.1%], abdominal pain [9.6%/17.4%]), fatigue (14.4%/1.7%), hepatotoxicity [0.3%/26.4%], febrile neutropenia (10.1%/1.7%), decreased appetite [8.0%/0.8%], maculopapular rash (5.2%/7.4%), pruritus (2.8%/10.7%), alopecia (1.5%/9.9%), pyrexia [4.1%/13.2%], oedema (6.2%/0.8%), rash (0.7%/5.8%), and increases of alanine transaminase (ALT [4.9%/10.7%]), aspartate transaminase (AST [4.1%/6.6%]) and bilirubin (17.1%/6.6%).
Adults.
Tabulated list of adverse reactions. The frequencies of adverse reactions reported in the table below are derived from 5 clinical trials (including a total of 613 patients) where treosulfan combined with fludarabine was investigated as conditioning treatment prior to alloHSCT in adult patients. Treosulfan was administered in a dose range of 10-14 g/m2 BSA on 3 consecutive days.
Adverse reactions are listed below, by system organ class and by frequency: very common (≥ 1/10), common (≥ 1/100 to < 1/10), uncommon (≥ 1/1,000 to < 1/100), rare (≥ 1/10,000 to < 1/1,000), very rare (< 1/10,000) and not known (cannot be estimated from the available data). Within each frequency group, undesirable effects are presented in order of decreasing seriousness. (See Table 2.)

 Description of selected adverse reactions.
Description of selected adverse reactions. Overall infections.
The overall incidence of infections was 10.1% (62/613). This includes the incidence for bacterial, viral and fungal infections (50/613; 8.1%) and for overall sepsis (12/613; 2%). The most frequent type of infection was lung infection (10/62 [16.1%]). Pathogens included bacteria (e.g. Staphylococcus, Enterococcus, Corynebacterium), viruses (e.g. cytomegalovirus [CMV], Epstein-Barr virus [EBV]) as well as fungi (e.g. candida). Overall sepsis includes sepsis (9/613; 1.5%), staphylococcal sepsis (2/613; 0.3%) and enterococcal sepsis (1/613; 0.2%). The infection rate was lowest in patients treated with the dose regimen of 10 g/m2 of treosulfan per day, from day -4 to -2 (8.1%).
Neoplasms benign, malignant and unspecified (including cysts and polyps).
One of 613 adult patients (0.2%) developed a second malignancy (breast cancer). A few further cases of second malignancies after treosulfan-based conditioning have been reported by other investigators. After long-term therapy with conventional doses of oral treosulfan in patients with solid tumours acute myeloid leukaemia was observed in 1.4% of 553 patients.
Blood and lymphatic system disorders.
Blood disorders were observed in 62 of 613 adult patients (10.1%). The most frequent adverse reaction was febrile neutropenia (10.1%). The lowest incidence was noted with the dose regimen of 10 g/m2/day, day -4 to -2 (4.4%).
The median (25%/75% percentiles) duration of neutropenia was 14 (12, 20) days with the 10 g/m2; treosulfan dose and 17.5 (14, 21) days with the 14 g/m2 treosulfan dose.
Cardiac disorders.
Cardiac disorders were observed in 21 patients (3.4%). The most frequent adverse reactions were cardiac arrhythmias, e.g. atrial fibrillation (1.0%), sinus tachycardia (0.8%), supraventricular tachycardia (0.3%), and ventricular extrasystole (0.3%). Isolated cases of cardiac arrest, cardiac failure, and myocardial infarction occurred. The lowest frequency of cardiac disorders was seen with the dose regimen of 10 g/m2/day, day -4 to -2 (2.6%).
Gastrointestinal disorders.
Gastrointestinal disorders were observed in 379 patients (61.8%). The most frequent adverse reactions reported were nausea (38.0%), stomatitis (36.4%), vomiting (22.5%), diarrhoea (14.4%), and abdominal pain (9.6%). The lowest frequencies of these adverse reactions were seen with the dose regimen of 10 g/m2 per day, day -4 to -2 (21.5%, 32.2%, 14.8%, 5.9%, and 6.7% respectively).
Hepatobiliary disorders.
The overall incidence of veno-occlusive liver disease (VOD) was 0.8% (5/613). VOD occurred only with the dose regimen of 14 g/m2/day treosulfan. None of these cases were fatal or life-threatening.
Paediatric population.
Tabulated list of adverse reactions. The adverse reactions reported in the table below are derived from two clinical trials (including a total of 121 patients; median age 7 years [range 0-17 years]) where treosulfan combined with fludarabine (and mostly with additional thiotepa) was administered as conditioning treatment prior to alloHSCT in paediatric patients with malignant or non-malignant diseases. Treosulfan was administered in a dose range of 10-14 g/m2 BSA on three consecutive days.
Adverse reactions are listed below, by system organ class and by frequency: very common (≥ 1/10), common (≥ 1/100 to < 1/10), uncommon (≥ 1/1,000 to < 1/100), rare (≥ 1/10,000 to < 1/1,000), very rare (< 1/10,000) and not known (cannot be estimated from the available data). Within each frequency group, undesirable effects are presented in order of decreasing seriousness. (See Table 3.)
 Description of selected adverse reactions.
Description of selected adverse reactions. Infections.
The overall incidence of infections in 121 paediatric patients was 11.6% (14/121) and thus comparable to that seen in adults. The frequency was higher in the paediatric age group 12-17 years (6/39 [15.4%]) compared to younger children (7/59 [11.9%]).
Neoplasms benign, malignant and unspecified (including cysts and polyps).
One case of a second malignancy (myelodysplastic syndrome) was reported in a child about 12 months after treosulfan-based conditioning for sickle cell disease.
Six cases of a second malignancy were reported by other investigators after treosulfan-based conditioning. Five paediatric patients received alloHSCT for primary immunodeficiencies, i.e. diseases with an increased risk for neoplasias per se. They developed myelodysplastic syndrome, acute lymphoblastic leukaemia, and Ewing's sarcoma. One patient with haemophagocytic lymphohistiocytosis developed secondary juvenile chronic myeloid leukaemia.
Blood and lymphatic system disorders.
The median (25%/75% percentiles) duration of neutropenia was 22 (17, 26) days in paediatric patients with malignant diseases and 20 (15, 25) days in patients with non-malignant disorders.
Nervous system disorders.
Seizure in the context of an encephalitis infection was reported in one of 121 paediatric patients. A report from an investigator-initiated trial performed in children with primary immunodeficiencies lists five cases of seizures occurring after other treosulfan-based conditioning regimens (see Section 4.4 Special Warnings and Precautions for Use).
Reporting of suspected adverse effects.
Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.4.9 Overdose
The principal toxic effect of treosulfan is profound myeloablation and pancytopenia. In addition, acidosis, skin toxicity, nausea, vomiting and gastritis may occur. In the absence of haematopoietic stem cell transplantation, the recommended dose of treosulfan would constitute an overdose. No specific antidote of treosulfan overdose is known. The haematologic status should be closely monitored and vigorous supportive measures instituted as medically indicated.
For information on the management of overdose, contact the Poisons Information Centre on 131126 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Pharmacotherapeutic group: Antineoplastic agents, alkylating agents, ATC code: L01AB02.
Mechanism of action.
Treosulfan is a pro-drug of a bifunctional alkylating agent with cytotoxic activity to haematopoietic precursor cells. The activity of treosulfan is due to the spontaneous conversion into a mono-epoxide intermediate and L-diepoxybutane (see Section 5.2 Pharmacokinetic Properties).
The epoxides formed alkylate nucleophilic centres of deoxyribonucleic acid (DNA) and are able to induce DNA cross-links which are considered responsible for the stem cell depleting and antineoplastic effects.
Pharmacodynamic effects.
Treosulfan has a broad antineoplastic and antileukaemic activity. This was demonstrated against transplanted mouse and rat lymphomas/leukaemias, sarcomas and hepatomas, human tumour xenografts, human tumour biopsies and cell lines.
The immunosuppressive effects of treosulfan are attributed to its toxicity against primitive and committed progenitor cells, T and NK cells, reduction of cellularity of primary and secondary lymphatic organs and a preclusive effect on the 'cytokine storm' that precedes the development of Graft-versus-Host-Disease (GvHD) and is involved in the pathogenesis of veno-occlusive disease.
Clinical trials.
In the pivotal phase III trial, adult patients with acute myeloid leukaemia (AML) or myelodysplastic syndrome (MDS) and increased risk for standard conditioning therapies because of higher age (≥ 50 years) or comorbidities (haematopoietic cell transplantation comorbidity index [HCT-CI] score > 2) were randomised to receive a conditioning regimen with 3 x 10 g/m2 treosulfan combined with fludarabine (FT10; n = 268) or a regimen of intravenous busulfan (total dose 6.4 mg/kg) combined with fludarabine (FB2; n = 283), followed by alloHSCT. 64% of patients had AML and 36% MDS. The median age of patients was 60 years (range 31-70 years); 25% of patients were older than 65 years.
The primary endpoint of this study was event-free survival (EFS) after 2 years. Events were defined as relapse of disease, graft failure or death (whatever occurred first). Non-inferiority of FT10 versus the reference FB2 was statistically proven. The p-value of 0.0005787 indicates superiority of treosulfan compared to busulfan (Figure 1).
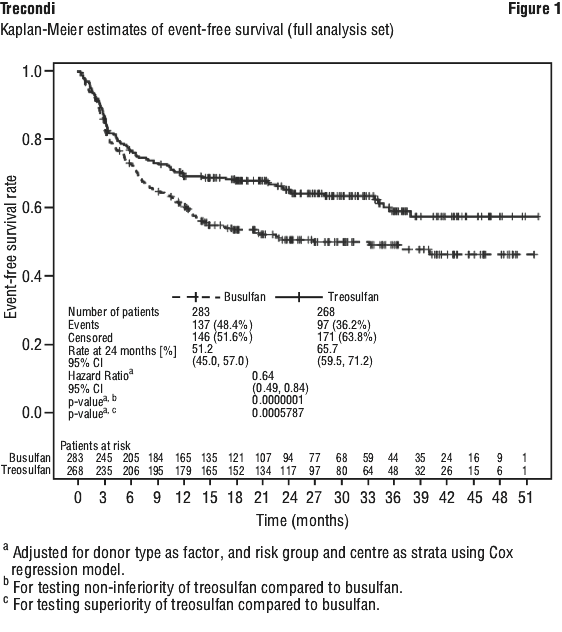 Analyses of EFS at 2 years for various pre-defined subgroups (donor type, risk group, disease, age group, HCT-CI score, remission status at study entry, and various combinations of these parameters) were always in favour of the treosulfan regimen (hazard ratio [HR] of FT10 vs. FB2 < 1), with only one exception (risk group II of matched related donor [MRD] patients; HR 1.18 [95% CI 0.61, 2.26]).
Analyses of EFS at 2 years for various pre-defined subgroups (donor type, risk group, disease, age group, HCT-CI score, remission status at study entry, and various combinations of these parameters) were always in favour of the treosulfan regimen (hazard ratio [HR] of FT10 vs. FB2 < 1), with only one exception (risk group II of matched related donor [MRD] patients; HR 1.18 [95% CI 0.61, 2.26]).
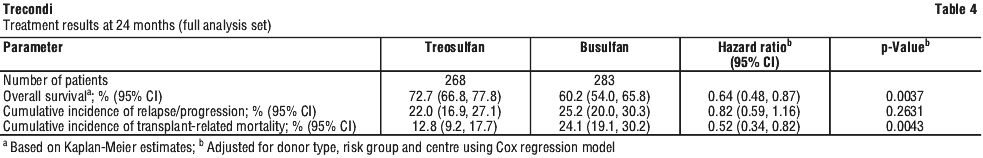
Further results are shown in Table 4.
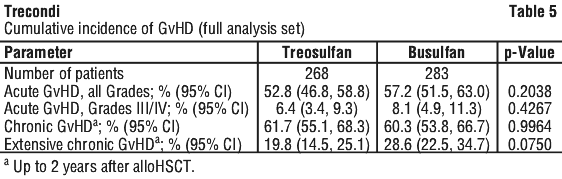
 Results of GvHD are shown in Table 5.
Results of GvHD are shown in Table 5.
Paediatric population.
The efficacy and safety of treosulfan-based conditioning was evaluated in 70 patients with acute lymphoblastic leukaemia (ALL), AML, MDS, or juvenile myelomonocytic leukaemia (JMML) who received a conditioning regimen with treosulfan and fludarabine with (n = 65) or without (n = 5) thiotepa. Treosulfan dose was adapted to the patient's BSA and 10, 12, or 14 g/m2 body surface area per day was administered as a two-hour intravenous infusion on day -6, -5, and -4 prior to stem cell infusion (day 0). A total of 37 patients (52.9%) were younger than 12 years.
No patient experienced a primary graft failure but one patient with ALL experienced a secondary graft failure. The incidence of complete donor-type chimerism was 94.2% (90% CI 87.2-98.0%) at day +28 visit, 91.3% (90% CI 83.6-96.1%) at day +100 visit and 91.2% (90% CI 82.4-96.5%) at month 12 visit.
The overall survival at 24 months was 85.7% (90% CI 77.1-91.2%). A total of 12 out of 70 patients (17.1%) died, 8 patients attributed to relapse/progression and 4 patients to transplant-related causes. Freedom from transplant-related mortality until day +100 after HSCT (primary endpoint) was achieved in 98.6% of patients (90% CI 93.4-99.9%). One transplant/treatment-related death occurred within this period. Transplant-related mortality at 24 months was 4.6% (90% CI 1.8 - 11.4%). Relapse or progression occurred in
16 patients, with a cumulative incidence of relapse/progression of 23.0% (90%
CI 14.7-31.3%) at month +24.
The efficacy and safety of treosulfan/fludarabine ± thiotepa-based conditioning was further evaluated in 51 patients with non-malignant diseases (primary immunodeficiency, haemoglobinopathy, inborn error of metabolism and bone marrow failure syndromes). Treosulfan dose was adapted to the patient's BSA and 10, 12, or 14 g/m2 body surface area per day was administered as a two-hour intravenous infusion on day -6, -5, and -4 prior to stem cell infusion (day 0). The dosing scheme was adapted during the trial in
terms of the BSA categories applied for the different doses, as a consequence 2 patients received a higher dose compared to the
initial dosing scheme. Fifty evaluable patients treated with the reference conditioning regimen busulfan/fludarabine ± thiotepa served as active control group. Busulfan dose was adapted to the patient's body weight and 3.2 to 4.8 mg/kg/day were administered on days -7, -6, -5, and -4. Most trial subjects (84% in both arms) received the intensified regimen with thiotepa given in 2 single doses of 5 mg/kg/body weight on day -2. Most patients were 28 days to 11 years of age (88.2% in the treosulfan arm and 80% in the busulfan arm). Alpha was not controlled for multiple testing in
this trial. The incidence of freedom from transplantation (treatment) related mortality until day +100 (primary endpoint) was 100.0% (90% CI 94.3%-100.0%) in the treosulfan arm and 90.0% (90% CI 80.1%-96.0%) in the busulfan arm. Overall survival at 1 year was 96.1% (90% CI 88.0%-98.8%) with treosulfan and 88.0% with busulfan (90% CI 77.9%-93.7%). In total, 2 patients (3.9%) in the treosulfan arm and 2 patients (4.0%) in the busulfan arm experienced primary graft failure, while secondary graft failures were reported for 9 patients (18.4%) receiving treosulfan-based conditioning. The incidence of complete donor type chimerism was comparable between the groups.
5.2 Pharmacokinetic Properties
Treosulfan is a pro-drug that is spontaneously converted under physiological conditions (pH 7.4; 37°C) into a monoepoxide intermediate and L-diepoxybutane with a half-life of 2.2 hours.
Absorption.
After intravenous administration, peak plasma levels are reached at the end of the infusion time. Maximum plasma levels (mean ± SD) in adult patients after a 2-hour intravenous infusion of 10, 12, or 14 g/m2 treosulfan were 306 ± 94 microgram/mL, 461 ± 102 microgram/mL, and 494 ± 126 microgram/mL, respectively.
Distribution.
Treosulfan is rapidly distributed in the body. A very low penetration of blood-brain-barrier by treosulfan was observed in rats. The treosulfan concentrations in brain tissue were 95%-98% lower than in plasma. However, an approximately 3-fold higher exposure in brain tissue of juvenile rats in comparison to young adults was found. The volume of distribution in adult patients is about 20-30 litres. No dose accumulation with the recommended daily treatment on three consecutive days was observed. Treosulfan does not bind to plasma proteins.
Metabolism.
Under physiological conditions (pH 7.4, temperature 37°C), the pharmacologically inactive treosulfan is converted spontaneously (non-enzymatically) into the active monoepoxide intermediate (S,S-EBDM = (2S,3S)-1,2-epoxybutane-3,4-diol-4-methanesulfonate) and finally to L-diepoxibutane (S,S-DEB = (2S,3S)-1,2:3,4-diepoxybutane).
In vitro data indicates that treosulfan is weakly metabolised by CYP2D6.
Excretion.
Plasma concentrations of treosulfan decline exponentially and are best described by a first order elimination process fitted by a two-compartment model.
The terminal half-life (T1/2ß) of intravenously administered treosulfan (up to 47 g/m2) is approximately 2 hours. Approximately 25-40% of the treosulfan dose is excreted unchanged with the urine within 24 hours, nearly 90% of which within the first 6 hours after administration.
Linearity/non-linearity.
Regression analysis of the area under the curve (AUC0-∞) versus treosulfan dose indicated a linear correlation.
Renal and hepatic impairment.
No pharmacokinetic studies with treosulfan were done in patients with severe renal or hepatic impairment, because such patients are generally excluded from alloHSCT. About 25-40% of treosulfan is excreted in urine; however, an influence of renal function on renal clearance of treosulfan was not observed.
Paediatric population.
Conventional dose calculation simply based on BSA results in a significantly higher exposure (AUC) of smaller children and infants with low BSA compared to adolescents or adults. Therefore, dosing of treosulfan in paediatric patients should be adapted to the BSA (see Section 4.2 Dose and Method of Administration), which results in a comparable treosulfan exposure in children of all age groups, corresponding to an exposure of a 3 x 14 g/m2 dose in adults.
Mean apparent terminal half-life of treosulfan was comparable between the different age groups and ranged between 1.3 and 1.6 hours.
PK/PD evaluation did not show a significant change of time to engraftment as function of AUC (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
5.3 Preclinical Safety Data
Genotoxicity.
Due to its alkylating mechanism of action treosulfan is characterised as a genotoxic compound with carcinogenic potential. Results from bacterial mutagenesis assays, in vitro chromosome aberration assays with human lymphocytes and reports from the published literature indicated that treosulfan and its active moieties were mutagenic in bacterial and mammalian assays and clastogenic in vitro and in vivo.
Carcinogenicity.
Due to its alkylating mechanism of action treosulfan is characterised as a genotoxic compound with carcinogenic potential. Treosulfan has been classified by the International Agency for Research on Cancer (IARC) as a human carcinogen.6 Pharmaceutical Particulars
6.1 List of Excipients
None.
6.2 Incompatibilities
In the absence of compatibility studies, this medicinal product must not be mixed with other medicinal products.
6.3 Shelf Life
Unopened vial.
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
Reconstituted solution for infusion.
After reconstitution with sodium chloride 4.5 mg/mL (0.45%) solution, chemical and physical stability has been demonstrated for 1 day (24 hours) at 25°C.
From a microbiological point of view, unless the method of reconstitution precludes the risk of microbial contamination, the product should be used immediately. If not used immediately, in-use storage times and conditions are the responsibility of the user.
Do not store in a refrigerator (2°C-8°C) as this might cause precipitation.
6.4 Special Precautions for Storage
Store below 25°C.
For storage conditions after reconstitution of the medicinal product, see Section 6.3 Shelf Life.
6.5 Nature and Contents of Container
Trecondi 1 g powder for solution for infusion.
Colourless type I glass vial, with rubber stopper and aluminium cap containing 1 g of treosulfan.
Trecondi 5 g powder for solution for infusion.
Colourless type I glass vial, with rubber stopper and aluminium cap containing 5 g of treosulfan.
Trecondi is available in packs of 1 or 5 vials.
Not all pack sizes may be marketed.
6.6 Special Precautions for Disposal
As with all cytotoxic substances, appropriate precautions should be taken when handling treosulfan (see Section 4.2 Dose and Method of Administration).
Treosulfan has mutagenic and carcinogenic potential. Remnants of the medicinal product as well as all materials that have been used for reconstitution and administration must be destroyed according to standard procedures applicable to antineoplastic agents.
In Australia, any unused medicine or waste material should be disposed of in accordance with local requirements.
6.7 Physicochemical Properties
Chemical structure.

CAS number.
299-75-2.7 Medicine Schedule (Poisons Standard)
S4 - Prescription Only Medicine.
Summary Table of Changes


 Fludarabine 30 mg/m2 BSA per day as a 0.5-hour intravenous infusion, given on five consecutive days (day -7, -6, -5, -4, -3) before stem cell infusion (day 0). The total fludarabine dose is 150 mg/m2;
Fludarabine 30 mg/m2 BSA per day as a 0.5-hour intravenous infusion, given on five consecutive days (day -7, -6, -5, -4, -3) before stem cell infusion (day 0). The total fludarabine dose is 150 mg/m2;


 Analyses of EFS at 2 years for various pre-defined subgroups (donor type, risk group, disease, age group, HCT-CI score, remission status at study entry, and various combinations of these parameters) were always in favour of the treosulfan regimen (hazard ratio [HR] of FT10 vs. FB2 < 1), with only one exception (risk group II of matched related donor [MRD] patients; HR 1.18 [95% CI 0.61, 2.26]).
Analyses of EFS at 2 years for various pre-defined subgroups (donor type, risk group, disease, age group, HCT-CI score, remission status at study entry, and various combinations of these parameters) were always in favour of the treosulfan regimen (hazard ratio [HR] of FT10 vs. FB2 < 1), with only one exception (risk group II of matched related donor [MRD] patients; HR 1.18 [95% CI 0.61, 2.26]). Results of GvHD are shown in Table 5.
Results of GvHD are shown in Table 5.

