What is in this leaflet
This leaflet answers some of the more common questions about TRIMBOW. It does not contain all the available information. It does not take the place of talking to your doctor or pharmacist.
All medicines have risks and benefits. Your doctor has weighed the risks of you being given this medicine against the benefits this medicine is expected to have for you.
If you have any concerns about taking this medicine, ask your doctor or pharmacist.
Keep this leaflet with the medicine. You may need to read it again.
What TRIMBOW is used for
TRIMBOW is a pressurised metered-dose inhaler that contains medicines to help you breathe.
TRIMBOW contains the three active ingredients:
- beclometasone dipropionate,
- formoterol (eformoterol) fumarate dihydrate
- glycopyrronium bromide (glycopyrrolate).
Beclometasone dipropionate belongs to a group of medicines called corticosteroids which act to reduce the swelling and irritation in your lungs.
Formoterol (eformoterol) fumarate dihydrate and glycopyrronium bromide (glycopyrrolate) are medicines called long-acting bronchodilators. They act in different ways to relax the muscles in your airways, helping to open the airways wider and allowing you to breathe more easily.
Regular treatment with this medicine helps to relieve and prevent symptoms such as shortness of breath, wheezing and cough in adult patients with chronic obstructive pulmonary disease (COPD) and asthma.
COPD is a serious long-term disease in which the airways become blocked and air sacs inside the lungs become damaged, leading to difficulty in breathing.
Asthma is a serious, long-term disease where the muscles surrounding the airways become tight (bronchoconstriction), and swollen and irritated (inflammation). Symptoms can come and go, which can include shortness of breath, wheezing, chest tightness and cough.
Do not use this medicine to relieve a sudden attack of breathlessness or wheezing.
You will use TRIMBOW every day and not only when you have breathing problems or other symptoms of COPD and asthma. This will ensure that it controls your symptoms properly.
Ask your doctor if you have any questions about why this medicine has been prescribed for you.
This medicine is available only with a doctor’s prescription.
TRIMBOW is unlikely to affect your ability to drive and use machines.
Before you use TRIMBOW
When you must not use it
Do not take this medicine if you have an allergy to:
- any medicine containing beclometasone dipropionate, formoterol (eformoterol) fumarate dihydrate or glycopyrronium bromide (glycopyrrolate)
- any of the other ingredients listed at the end of this leaflet.
Some of the symptoms of an allergic reaction may include:
- shortness of breath
- wheezing or difficulty breathing
- swelling of the face, lips, tongue, or other parts of the body
- rash, itching or hives on the skin.
Do not give this medicine to children and adolescents below the age of 18 years.
Do not use this medicine after the expiry date printed on the pack or if the packaging is torn or show signs of tampering. If it has expired or is damaged, return to your pharmacist for disposal.
If you are not sure whether you should start using this medicine, talk to your doctor.
Before you start to use it
Tell your doctor if you have or have had any of the following medical conditions:
- heart problems (heart pain, pain in the chest, recent heart attack, heart failure, narrowing of the arteries around your heart, disease of your heart valves or any other abnormalities of your heart)
- heart condition known as hypertrophic obstructive cardiomyopathy (also known as HOCM, a condition where the heart muscle is abnormal)
- heart rhythm disorders such as irregular heart rate, a fast pulse rate or palpitations or abnormal heart trace (ECG)
- high blood pressure or swelling and weakening of blood vessels
- an overactive thyroid gland
- low blood levels of potassium
- disease of your liver or kidneys
- diabetes or high blood sugars
- a tumour of the adrenal gland
- tuberculosis (TB)
- viral or fungal chest infection
- eye problems called narrow-angle glaucoma
- prostate problems or difficulty passing urine
- an infection of the mouth or throat.
Tell your doctor if you are pregnant or plan to become pregnant or are breastfeeding. You should only use TRIMBOW during pregnancy if you are advised to do so by your doctor. It is preferable to avoid the use of TRIMBOW during labour as it may impact contractions of the uterus.
You should not use TRIMBOW during breast-feeding. You and your doctor will decide whether to discontinue breast-feeding or to discontinue TRIMBOW taking into account the benefit of breast-feeding for your child and the benefit of therapy for you.
If you have not told your doctor about any of the above, speak with your doctor before you start using TRIMBOW.
Taking other medicines
Tell your doctor or pharmacist if you are taking any other medicines, including any that you get without a prescription from your pharmacy, supermarket or health food shop.
Some medicines and TRIMBOW may interfere with each other. These include:
- medicines for HIV such as ritonavir, cobicistat
- medicines for treating high blood pressure, heart conditions and abnormal heart rhythms (such as digoxin, quinidine, disopyramide, procainamide)
- medicines to treat glaucoma
- medicines to treat migraines
- other medicines to treat COPD or asthma (such as theophylline, aminophylline or corticosteroids)
- medicines used to treat allergic reactions (antihistamines)
- medicines to treat depression or mental disorders such as monoamine oxidase inhibitors (phenelzine, isocarboxazid, furazolidone and procarbazine), tricyclic antidepressants (amitriptyline and imipramine), or phenothiazines
- L-dopa, a medicine used to treat Parkinson’s Disease
- L-thyroxine, a medicine used to treat an underactive thyroid gland
- oxytocin, used to bring on (induce) labour
- medicines containing alcohol
- fluid or water tablets
- anaesthetics used during surgery
- disulfiram, a medicine used to treat drinking problems
- metronidazole, an antibiotic to treat infection in your body.
Your doctor and pharmacist have more information on medicines to be careful with or avoid while taking this medicine.
How to use TRIMBOW
Follow all directions given to you by your doctor or pharmacist carefully. They may differ from the information contained in this leaflet.
If you do not understand the instructions in this leaflet, ask your doctor or pharmacist for help.
How much to use
Always use this medicine exactly as your doctor or pharmacist has told you. Check with your doctor or pharmacist if you are not sure.
The recommended dose is two puffs in the morning and two puffs in the evening at the same time every day.
Do not use more than your doctor tells you to use.
TRIMBOW is not used for initial therapy. TRIMBOW is used as a maintenance treatment for your COPD or asthma.
How to use the inhaler
TRIMBOW is for inhalation use.
You should inhale the medicine through your mouth and this takes the medicine directly into your lungs.
This medicine is contained in a pressurised container enclosed in a plastic inhaler with a mouthpiece. Each container provides 60 puffs or 120 puffs or 180 puffs.
If you have been prescribed a container providing 60 puffs or 120 puffs:
There is a counter on the back of the inhaler, which corresponds to the number of puffs left in the container. Each time you press the pressurised container, a puff of medicine is released and the counter will count down by one. Take care not to drop the inhaler as this may cause the counter to count down.
If you have been prescribed a container providing 180 puffs:
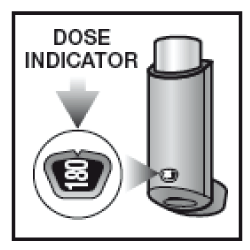
There is an indicator on the back of the inhaler, which tells you how many doses are left. Each time you press the pressurised container, a puff of medicine is released, and the dose indicator rotates by a small amount. The number of puffs remaining is displayed in intervals of 20. Take care not to drop the inhaler as this may cause the dose indicator to count down.
Testing your inhaler
Before using the inhaler for the first time, you should test your inhaler to make sure that it is working properly, as follows.
- Depending on the inhaler size prescribed for you, check that the dose counter reads 61 or 121 or 180
- Remove the protective cap from the mouthpiece
- Hold your inhaler upright with the mouthpiece at the bottom
- Direct the mouthpiece away from yourself and firmly press the pressurised container to release one puff
- Check the dose counter. If you are testing your inhaler for the first time, the counter should read:
60 - when using the container providing 60 puffs.
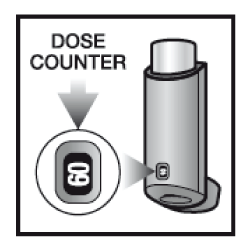
120 - when using the container providing 120 puffs

180 - when using the container providing 180 puffs
How to use your inhaler
Stand or sit up when inhaling.
IMPORTANT: Do not perform steps 2 to 5 too quickly.
- Remove the protective cap from the mouthpiece and check that the mouthpiece is clean and free from dust and dirt.
- Breathe out as slowly and deeply as possible, in order to empty your lungs.
- Hold the inhaler upright with the mouthpiece at the bottom and place the mouthpiece between your teeth without biting it. Then place your lips around the mouthpiece, with the tongue flat under it.
- Breathe in slowly and deeply through your mouth to fill your lungs with air (this should take about 4-5 seconds). Just after starting to breathe in, press down firmly on the top of the pressurised container to release one puff.
- Hold your breath for as long as possible and, finally, remove the inhaler from your mouth and breathe out slowly. Do not breathe out into the inhaler.
- Check that the dose counter has moved down by one (for 60 and 120 puff inhalers) or that the dose indicator has rotated by a small amount (180 puff inhaler).

For the second puff, keep the inhaler in the upright position for about half a minute, then repeat steps 2 to 5.
If you see 'mist' coming from the top of the inhaler or the sides of your mouth, this means that TRIMBOW will not be getting into your lungs as it should. Take another puff, following the instructions starting again from step 2.
After use
Replace the protective cap.
To prevent a fungal infection in the mouth and throat, rinse your mouth or gargle with water without swallowing it or brush your teeth after each use of your inhaler.
When to get a new inhaler
You should get a replacement when the counter or indicator shows the number 20. Stop using the inhaler when the counter or indicator shows 0, as any medicine left in the inhaler may not be enough to give you a full puff.
If you have a weak grip, it may be easier to hold the inhaler with both hands. Hold the upper part of the inhaler with both index fingers and its lower part with both thumbs.
If you find it difficult to use the inhaler while starting to breathe in, you may use a suitable spacer device. Ask your doctor or pharmacist about this device.
It is important that you read the package leaflet which is supplied with your spacer device and that you carefully follow the instructions on how to use the spacer device and how to clean it.
Cleaning the TRIMBOW inhaler
You should clean your inhaler once a week.
- Do not remove the pressurised container from the inhaler and do not use water or other liquids to clean your inhaler.
- Remove the protective cap from the mouthpiece by pulling it away from your inhaler.
- Wipe inside and outside of the mouthpiece and the inhaler with a clean, dry cloth or tissue.
- Replace the mouthpiece cap.
When to use it
You should use TRIMBOW every day, even when your COPD or asthma is not troubling you.
If you feel that the medicine is not working or you are getting breathless or wheezy, talk to your doctor. Do not use this medicine to relieve or treat a sudden attack of breathlessness or wheezing.
How long to use it
Continue using TRIMBOW for as long as your doctor tells you.
Do not stop using TRIMBOW or lower the dose, even if you are feeling better or have no symptoms.
It is important to use TRIMBOW every day.
If you have any further questions on the use of this medicine, ask your doctor or pharmacist.
If you forget to use it
Use it as soon as you remember. If it is almost time for your next dose, do not take the dose you have missed, but just take the next dose at the correct time.
Do not use a double dose to make up for the dose that you missed.
If you use too much (overdose)
Immediately telephone your doctor or Poisons Information Centre (telephone 13 11 26) or go to Accident and Emergency at your nearest hospital if you think that you or anyone else may have taken or been given too much TRIMBOW. Do this even if there are no signs of discomfort or poisoning. You may need urgent medical attention.
If you use more TRIMBOW than you should, side effects may occur (see “Side Effects”).
While you are using TRIMBOW
Things you must do
If you are about to be started on any new medicine, remind your doctor and pharmacist that you are taking TRIMBOW.
Contact your doctor if you experience blurred vision or other visual disturbances.
Tell any other doctors, dentists, and pharmacists who treat you that you are taking this medicine.
If you are going to have surgery, tell the surgeon or anaesthetist that you are taking this medicine. It may affect other medicines used during surgery.
If you become pregnant while taking this medicine, tell your doctor immediately.
Keep all of your doctor's appointments so that your progress can be checked.
Stop taking TRIMBOW, use your quick-acting “reliever” inhaler and seek medical help immediately if your breathing gets worse, this may include short of breath or start wheezing (breathing with an audible whistling sound) straight after using TRIMBOW.
Things you must not do
Do not take TRIMBOW to treat any other complaints unless your doctor tells you to.
Do not give your medicine to anyone else, even if they have the same condition as you.
Do not stop taking your medicine or lower the dosage without checking with your doctor.
Things to be careful of
TRIMBOW contains ethanol
TRIMBOW contains 8.856 mg of ethanol absolute (alcohol) in each actuation, which is equivalent to 17.712 mg per dose of two actuations. The amount in two actuations of this medicine is equivalent to less than 1 mL of wine or beer. The small amount of alcohol in this medicine will not have any noticeable effects.
Side effects
Tell your doctor or pharmacist as soon as possible if you do not feel well while you are using TRIMBOW.
All medicines can have side effects. Sometimes they are serious, most of the time they are not. You may need medical treatment if you get some of the side effects.
Do not be alarmed by the list of possible side effects. You may not experience any of them.
Ask your doctor or pharmacist to answer any questions you may have.
Tell your doctor or pharmacist if you notice any of the following and they worry you:
- sore, raised patches in the mouth, throat or oesophagus (food pipe) caused by a fungal infection (oral thrush). Rinsing your mouth out with water immediately after using the inhaler may prevent this side effect.
- dry mouth, sore mouth, burning sensation of the lips, tooth decay
- sore, dry or irritated throat, swallowing difficulties
- flu-like symptoms
- runny or stuffy nose and sneezing, nose bleeds
- feeling of tension or fullness in your sinuses
- hoarseness of the voice
- cough
- abnormal or reduced sense of taste
- headache
- inflammation of the ear
- skin rash, hives, itchy skin
- increased sweating, flushing of the skin
- bleeding or bruising more easily than normal, bleeding into the tissues
- painful, difficult or frequent urination (may be a sign of a urinary tract infection or problems)
- fungal infection of the vagina with itching, burning and white discharge (thrush)
- diarrhoea, feeling sick, upset stomach, stomach discomfort after meals
- decreased appetite
- muscle cramps or pain, pain in arms, legs or chest muscles
- swelling of the hands and feet
- restlessness, trembling, numbness, weakness
- dizziness, fatigue, sleep disorders
- irregular heartbeat, sensation of fast, slow or missed heart beats, palpitations.
The above list includes less serious side effects of your medicine.
Tell your doctor as soon as possible if you notice any of the following:
- symptoms of a lung infection such as fever or chills, increased mucus production, change in mucus colour, or increased cough or increased breathing difficulties
- severe chills, sore throat or mouth ulcers.
The above list includes serious side effects that may require medical attention.
If any of the following happen, tell your doctor immediately or go to Accident and Emergency at your nearest hospital:
- worsening shortness of breath and wheezing immediately after using TRIMBOW
- sudden signs of allergy such as rash, itching or hives on the skin, swelling of the face, lips, tongue or other parts of the body
- eye pain or discomfort, temporary blurring of vision, visual halos or coloured images in association with red eyes (signs of an acute attack of narrow-angle glaucoma)
- crushing chest pain often spreading to the arms or neck and sometimes to the shoulders and back (angina).
The above list includes very serious side effects. You may need urgent medical attention or hospitalisation.
Tell your doctor or pharmacist if you notice anything else that is making you feel unwell. Other side effects not listed above may occur in some patients. Some of these side effects (for example, changes in blood pressure, changes in blood levels of sugars, electrolytes, hormones, blood cells, cells to help your blood clot, fatty acids, ketones or proteins in the blood) can only be found when your doctor does tests from time to time to check your progress.
Using high-dose inhaled corticosteroids over a long time can cause other very rare side effects. TRIMBOW does not contain a high dose of corticosteroids, however, your doctor will monitor you for other rare side effects such as:
- problems with how your adrenal glands work
- thinning of your bones
- clouding of the lens of your eyes (cataracts)
- depression, mental disorders, feeling worried, nervous, over-excited or irritable
- growth retardation and changes in behaviour (children and adolescents).
After using TRIMBOW
Storage
Keep it where children cannot reach it.
Do not expose to temperatures higher than 50°C.
Do not pierce the pressurised container.
TRIMBOW 100/6/10
60, 120 or 180 puff inhalers:
Store below 30°C for a maximum of 2 months. Your pharmacist will enter the dispensing date on a label affixed to the inhaler.
From a multipack:
Store the unused inhaler(s) in a refrigerator (2°C-8°C) until you are ready to use. Do not freeze.
Before use, take one inhaler out of the refrigerator for some minutes to allow for the solution to warm up. After first use, store the inhaler below 30°C for a maximum of 2 months. Enter the date of first use on the removable label and affix to the inhaler. Do not put the inhaler back into the refrigerator.
TRIMBOW 200/6/10
60 puff inhaler:
Store the inhaler below 30°C for a maximum of 1 month. Your pharmacist will enter the dispensing date on a label affixed to the inhaler.
120 puff inhaler:
Store the inhaler below 30°C for a maximum of 2 months. Your pharmacist will enter the dispensing date on a label affixed to the inhaler.
From a multipack:
Store the unused inhaler(s) in a refrigerator (2°C-8°C) until you are ready to use. Do not freeze.
Before use, take one inhaler out of the refrigerator for some minutes to allow for the solution to warm up. After first use, store the inhaler below 30°C for a maximum of 2 months. Enter the date of first use on the removable label and affix to the inhaler. Do not put the inhaler back into the refrigerator.
Disposal
If your doctor tells you to stop using this medicine or the expiry date has passed, ask your pharmacist what to do with any medicine that is left over.
Product Description
What it looks like
TRIMBOW is a pressurised inhalation solution.
TRIMBOW comes in a pressurised container (coated aluminium), with a metering valve. The pressurised container is inserted in a grey plastic inhaler. This incorporates a mouthpiece provided with a plastic protective red cap (TRIMBOW 100/6/10) or green cap (TRIMBOW 200/6/10) and a dose counter.
TRIMBOW 100/6/10
Pack of 1 container with either 60, 120 or 180 puffs.
Multipack containing 240 puffs (2 containers of 120 puffs each).
Multipack containing 360 puffs (3 containers of 120 actuations each).
Not all pack sizes may be marketed.
TRIMBOW 200/6/10
Pack of 1 container with either 60 or 120 puffs.
Multipack containing 240 puffs (2 containers of 120 puffs each).
Multipack containing 360 puffs (3 containers of 120 puffs each).
Not all pack sizes may be marketed.
Ingredients
The active ingredients of TRIMBOW are beclometasone dipropionate, formoterol (eformoterol) fumarate dihydrate and glycopyrronium bromide (glycopyrrolate).
Each metered dose (the dose leaving the mouthpiece) contains 100 micrograms of beclometasone dipropionate, 6 micrograms of formoterol fumarate dihydrate and 10 micrograms of glycopyrronium (as 12.5 micrograms glycopyrronium bromide).
Each metered dose (the dose leaving the mouthpiece) contains 200 micrograms of beclometasone dipropionate, 6 micrograms of formoterol fumarate dihydrate and 10 micrograms of glycopyrronium (as 12.5 micrograms glycopyrronium bromide).
The other ingredients are:
- ethanol absolute (alcohol)
- hydrochloric acid
- norflurane as the propellant.
Supplier
TRIMBOW is supplied by:
Chiesi Australia Pty Ltd
Suite 3, 22 Gillman Street,
Hawthorn East, VIC 3123
E: [email protected]
W: www.chiesi.com.au
This leaflet was prepared in May 2022.
TRIMBOW 100/6/10:
AUST R 314166
TRIMBOW 200/6/10:
AUST R 368010
Published by MIMS July 2022






 Among the observed adverse reactions, the following are typically associated with:
Among the observed adverse reactions, the following are typically associated with:



 Co-primary endpoints (pre-dose FEV1 at Week 26 and the rate of moderate and severe exacerbation rate over 52 weeks) and the key secondary endpoints (peak0-3h FEV1 at Week 26, morning PEF over 26 weeks and the rate of severe exacerbations [pooled analysis of TRIMARAN and TRIGGER] over 52 weeks) were part of the step-down, closed confirmatory testing strategy and thus controlled for multiplicity.
Co-primary endpoints (pre-dose FEV1 at Week 26 and the rate of moderate and severe exacerbation rate over 52 weeks) and the key secondary endpoints (peak0-3h FEV1 at Week 26, morning PEF over 26 weeks and the rate of severe exacerbations [pooled analysis of TRIMARAN and TRIGGER] over 52 weeks) were part of the step-down, closed confirmatory testing strategy and thus controlled for multiplicity.


