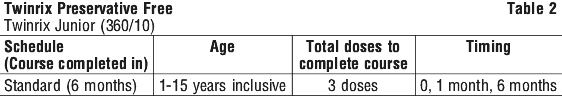WHAT IS IN THIS LEAFLET
This leaflet answers some of the common questions about TWINRIX vaccine. It does not contain all the available information. It does not take the place of talking to your doctor or pharmacist.
All medicines and vaccines have risks and benefits. Your doctor has weighed the possible risks of you or your child having TWINRIX against the expected benefits.
If you have any concerns about receiving TWINRIX talk to your doctor, nurse or pharmacist.
Keep this leaflet with this vaccine. You may need to read it again.
WHAT TWINRIX IS USED FOR
TWINRIX is a combination vaccine used to prevent hepatitis A and hepatitis B infection. The vaccine works by causing the body to produce its own protection (antibodies) against these diseases.
Hepatitis A and hepatitis B are infectious diseases, which cause the liver to become inflamed (swollen). These diseases are caused by viruses - hepatitis A and hepatitis B viruses.
Hepatitis A
Symptoms of hepatitis A usually begin 3 to 6 weeks after coming into contact with the virus. These consist of nausea (feeling sick), fever, aches and pains. After a few days the skin and/or the whites of the eyes may become yellowish (jaundice). The severity and type of symptoms can vary. Hepatitis A is often milder in young children. Most people recover completely but the illness is usually severe enough to keep adults off work for about a month.
The hepatitis A virus can be passed from person to person in food and drink, or by swimming in water contaminated by sewage. Hepatitis A is very common in many parts of the world and the risk of infection is greatest in those areas where hygiene and sanitation are poor. Vaccination is recommended for travellers to all developing countries, including people in the armed forces.
Hepatitis A occurs in Australia, but is not common. Some groups of people however are at a higher risk of exposure to the disease. Vaccination is recommended for these people:
- nursing staff and healthcare workers in contact with patients in childrens wards, infectious diseases wards, emergency rooms and intensive care units
- day-care centre staff particularly where children have not been toilet trained
- staff and residents of homes for the intellectually disabled
- sewerage workers
- food handlers
- homosexual men
- people in contact with an infected person
- people with chronic liver disease or liver transplants, or people who receive certain blood products.
Hepatitis B
The hepatitis B virus is found in body fluids such as blood, semen, vaginal secretions, or saliva of infected people. You can catch the virus if it can enter your bloodstream. Ways this can happen are through:
- injection (eg needlestick injury, or sharing needles for IV drug use)
- sexual intercourse
- sores, cuts or tiny wounds coming into contact with infected fluids (eg from a human bite, sharing razors or toothbrushes, or working with human blood or body fluids)
- an infected mother passing the virus onto her baby during or shortly after birth.
Some people infected with hepatitis B may not look or feel sick yet others will get symptoms. Symptoms may not appear for 6 weeks to 6 months after infection. Sometimes people will only have mild flu-like symptoms, but other people can become very ill. They may be extremely tired, and have dark urine, pale faeces, yellowish skin and/or eyes (jaundice), and other symptoms possibly requiring hospitalisation. There is a risk of serious liver disease, such as cirrhosis (liver scarring) and liver cancer for all chronic hepatitis B carriers.
Some groups of people are at a higher risk of exposure to hepatitis B. Vaccination is recommended for these people:
- some healthcare workers
- abusers of injectable drugs
- people with many sexual partners
- homosexual men
- haemodialysis patients or people who receive certain blood products
- people with chronic liver disease or hepatitis C
- people in contact with a hepatitis B carrier or an infected person
- staff and residents of institutions for the intellectually disabled
- inmates and prison staff at some correctional institutions
- some travellers to areas where the incidence of hepatitis B is high
There is no specific treatment for hepatitis A or hepatitis B. Vaccination is the best way to protect against infection.
TWINRIX will not protect against hepatitis caused by other agents or viruses (such as hepatitis C or hepatitis E). If a person is already infected with hepatitis A or hepatitis B virus at the time of vaccination, TWINRIX may not prevent the disease in these people.
BEFORE RECEIVING TWINRIX
DO NOT HAVE TWINRIX IF:
- you have/ your child has had an allergic reaction to TWINRIX, or any ingredient contained in this vaccine (e.g. neomycin sulphate). The ingredients are listed at the end of this leaflet. Signs of an allergic reaction may include an itchy skin rash, shortness of breath and swelling of the face or tongue.
If you have/ your child has had TWINRIX before and became unwell, tell your doctor, nurse or pharmacist before the vaccine is given. - you have/ your child has had an allergic reaction to any other hepatitis A or hepatitis B vaccine.
- you have/ your child has a severe infection with a high temperature. A minor infection such as a cold should not be a problem, but talk to your doctor about this before being vaccinated.
- the expiry date printed on the pack has passed.
- the packaging is torn or shows signs of tampering.
If you are not sure whether TWINRIX should be given, talk to your doctor or nurse. Do not give this vaccine to anyone else; your doctor has prescribed it specifically for you or your child.
BEFORE RECEIVING TWINRIX TELL YOUR DOCTOR IF:
- you have/ your child has an allergy to baker’s yeast.
- you are or think you may be pregnant or if you intend to become pregnant. Your doctor will discuss with you the possible risks and benefits of receiving TWINRIX during pregnancy.
- you are breast feeding. It is not known if TWINRIX passes into breast milk, however the vaccine is not expected to cause problems for breast-fed babies.
- you have/ your child has any medical conditions, such as:
- severe heart or lung disease
- a liver or kidney problem
- an immune deficiency condition (eg. HIV positive)
- a nervous system illness
- or a bleeding disorder; - you have/ your child has allergies to any other medicines or substances, such as dyes, foods or preservatives.
- you have/ your child has received another vaccine recently, or are taking any prescription or OTC (over-the-counter) medicines. In particular mention if you are taking medicines which suppress the immune system, such as steroids or cyclosporin.
Fainting can occur following, or even before, any needle injection, therefore tell the doctor or nurse if you/your child fainted with a previous injection.
Some vaccines may be affected by other vaccines or medicines. Your doctor or pharmacist will be able to tell you what to do if TWINRIX is to be given with another vaccine or medicine.
HOW TWINRIX IS GIVEN
The doctor or nurse will give TWINRIX as an injection. If you have any concerns about how this vaccine is to be given, talk to your doctor or pharmacist.
HOW MUCH IS GIVEN
The dose using TWINRIX (720/20) is 1 mL.
The dose using TWINRIX JUNIOR (360/10) is 0.5 mL
HOW IT IS GIVEN
TWINRIX will be injected into your upper arm muscle in adults and older children, and into the thigh muscle in infants. For some people with bleeding problems, the dose may need to be given under the skin (subcutaneously). Each dose of TWINRIX is for single use only. Any residual vaccine must be discarded.
The vaccine should never be given intravenously.
WHEN IT IS GIVEN
Adults
TWINRIX (720/20) is generally given as a total of three doses over 6 months. Each dose is given on a separate visit. The first dose will be given on an elected date. The remaining two doses will be given one month, and six months after the first dose.
- First dose: at an elected date
- Second dose: 1 month later
- Third dose: 6 months after the first dose
For adults, TWINRIX (720/20) can also be given as a total of three doses over 3 weeks (a 0, 7, 21 day schedule). However, the body’s immune response to this rapid schedule may be reduced compared to the above schedule. Therefore, this rapid schedule should only be used under special circumstances (e.g. adult travellers wanting to be vaccinated within one month of departure). A booster dose is recommended at 12 months.
Children (1 to 15 years inclusive)
TWINRIX (720/20) is generally given as a total of two doses 6 to 12 months apart. Each dose is given on a separate visit. The first dose will be given on an elected date. The second dose will be given 6 to 12 months after the first dose.
- First dose: at an elected date
- Second dose: 6 to 12 months after the first dose.
In children who are travelling to areas where there is a risk of exposure to hepatitis B, TWINRIX Junior (360/10) should be given as a total of 3 doses over 6 months ( a 0, 1, 6 month schedule).
It is important to return at the recommended times for follow up doses.
Your doctor will advise on the possible need for extra doses, and future booster dosing.
IF YOU MISS A DOSE
If you miss a scheduled dose, talk to your doctor and arrange another visit as soon as possible.
WHILE YOU ARE USING TWINRIX
THINGS YOU MUST DO:
Keep your follow up visits with the doctor or clinic. It is important that the follow-up doses of TWINRIX are given at the correct times. This will ensure the best effect of the vaccine in protecting you against hepatitis A and hepatitis B.
THINGS TO BE CAREFUL OF:
Be careful driving or operating machinery until you know how TWINRIX affects you. TWINRIX should not normally interfere with your ability to drive a car or operate machinery. But in some people vaccination can cause dizziness or lightheadedness. Make sure you know how you react to TWINRIX before you drive a car or operate machinery, or do anything that could be dangerous if you feel dizzy or lightheaded.
SIDE EFFECTS
Tell your doctor, nurse or pharmacist as soon as possible if you or your child do not feel well during or after a dose of TWINRIX.
TWINRIX helps protect most people from hepatitis A and hepatitis B, but it may have unwanted side effects in a few people. All medicines and vaccines can have side effects. Most of the time they are not serious; however, sometimes they can be. Some side effects may need medical treatment.
Ask your doctor or pharmacist to answer any questions you may have.
Most unwanted effects with TWINRIX are mild and usually clear up within a few days. These effects, generally occur around the injection site (soreness, redness, swelling). Fatigue, headache, fever and generally feeling unwell have also been reported, as have feeling sick and vomiting.
MILD EVENTS that have been reported after hepatitis A or hepatitis B vaccination.
Tell your doctor if you notice any of the following that are troublesome or ongoing:
- soreness, redness, swelling, bruising, hard lump or itching around the injection site
- headache, fatigue, fever, dizziness, disturbed sleep, fainting
- feeling sick or vomiting, gut pain, diarrhoea, loss of appetite
- cough, sore throat, respiratory infections.
- ringing in the ears, neck stiffness
- sweating, chills, flushing or generally feeling unwell
- muscle aches and pains, painful joints
- immediate injection site pain, stinging and burning feeling
MORE SERIOUS EVENTS that have been reported rarely after hepatitis A or hepatitis B vaccination.
Tell your doctor immediately if you notice any of the following:
- feelings of numbness, weakness and/ or fatigue in limbs, tingling in fingers or toes,
- generalised stiffness, visual changes, difficulty passing urine
- swollen glands, unusual bleeding, bleeding or bruising more easily than normal
- drooping eyelids or sagging facial muscles
- sudden headache, neck stiffness, dislike of bright lights, convulsions (fits)
As with all vaccines given by injection there is a very small risk of serious allergic reaction. This may occur days to weeks after vaccination. Contact your doctor immediately or go to the casualty department of your nearest hospital if any of the following happens:
- swelling of limbs, face, eyes, inside of nose, mouth or throat
- shortness of breath, breathing or swallowing difficulties
- hives, itching (especially of the hands or feet), reddening of skin (especially around the ears), or severe skin reactions
- unusual tiredness or weakness that is sudden and severe.
Other side effects not listed above, can also occur during or soon after a dose of TWINRIX.
Check with your doctor, nurse or pharmacist if you or your child have any other effects.
Do not be alarmed by this list of possible side effects. You or your child may not experience any.
STORAGE
TWINRIX is usually stored at the doctor’s clinic or surgery, or at the pharmacy. But if you need to store TWINRIX always:
- Keep TWINRIX in the refrigerator stored between +2 C and +8 C. THE PACK SHOULD NEVER BE FROZEN. FREEZING DESTROYS THE VACCINE.
- Keep the vaccine out of the reach of children.
- Keep TWINRIX in the original pack until it is time for it to be given.
Ask your pharmacist what to do with any left over TWINRIX that has expired or has not been used.
PRODUCT DESCRIPTION
WHAT IT LOOKS LIKE:
TWINRIX comes in prefilled syringes. It is a white, slightly milky liquid.
INGREDIENTS:
The active ingredients of TWINRIX are killed hepatitis A virus and the surface protein of the hepatitis B virus (from genetically engineered yeast cells). The vaccine is not infectious, and will not give you hepatitis A or hepatitis B.
Two different vaccine strengths are available:
- TWINRIX (720/20): 720 ELISA units of killed hepatitis A virus and 20 micrograms of the hepatitis B surface protein.
- TWINRIX JUNIOR (360/10): 360 ELISA units of killed hepatitis A virus and 10 micrograms of the hepatitis B surface protein.
Inactive ingredients in the vaccine are: aluminium hydroxide hydrate, aluminium phosphate, sodium chloride, amino acid supplement, formaldehyde, neomycin sulphate, polysorbate 20, -dibasic sodium phosphate heptahydrate, -monobasic sodium phosphate, trometamol and water.
TWINRIX is made without any human blood or blood products.
The manufacture of this product includes exposure to bovine derived materials. No evidence exists that any case of vCJD (considered to be the human form of bovine spongiform encephalopathy) has resulted from the administration of any vaccine product.
FURTHER INFORMATION
TWINRIX is only available if prescribed by a doctor.
TWINRIX (720/20) is available as:
Prefilled syringes in packs of 1 and 10 (AUST R 140575)
TWINRIX JUNIOR (360/10) is available as:
Prefilled syringes in packs of 1 and 10 (AUST R 140576)
MANUFACTURER
GlaxoSmithKline Biologicals S.A
rue de l'Institut 89,
1330 Rixensart,
Belgium.
SPONSOR:
GlaxoSmithKline Australia Pty Ltd
Level 4,
436 Johnston Street
Abbotsford, Victoria, 3067
Trade marks are owned or licensed to the GSK group of companies.
© 2022 GSK group of companies or its licensor.
Date of Preparation:
18 July 2022
Version 6.0
Published by MIMS December 2022