What is in this leaflet
This leaflet answers some common questions about Typhim Vi.
It does not contain all the available information.
It does not take the place of talking to your doctor or pharmacist.
All medicines, including vaccines, have risks and benefits. Your doctor has weighed the risks of you or your child having Typhim Vi against the benefits they expect it will have.
If you have any concerns about this vaccine, ask your doctor, nurse or pharmacist.
Keep this leaflet. You may need to read it again.
What Typhim Vi is used for
Typhim Vi is a vaccine used to help prevent typhoid fever, which is caused by a bacterium called Salmonella typhi.
The organism which causes typhoid fever can be passed on through contaminated food or water. If you are in an area where there is a risk of getting typhoid fever, in addition to having Typhim Vi, it is also very important to be careful with your personal, food and water hygiene. Hygiene will also help avoid illness caused by other bacteria in the water.
Typhim Vi may be given to people aged 2 years and older.
If you want more information on personal care in areas where there is a risk of getting typhoid fever, talk to your doctor, nurse or pharmacist.
How Typhim Vi works
Typhim Vi works by causing your body to produce its own protection against typhoid fever. It does this by making substances, called antibodies, which destroy the Salmonella typhi organism. If you have been vaccinated against Salmonella typhi, your body is able to attack that organism if you come in contact with it.
Your body usually takes several weeks after vaccination to fully develop this protection against typhoid fever.
You must have one dose at least 2 weeks before you may be exposed to typhoid fever.
If you are continually or repeatedly exposed to the Salmonella typhi organism, it is recommended that you have an injection of Typhim Vi every 3 years.
Most people will make enough antibodies against Salmonella typhi. However, as with all vaccines, 100% protection cannot be guaranteed.
The vaccine cannot give you or your child typhoid fever.
The chance of a severe reaction from Typhim Vi is very small, but the risks from not being vaccinated against typhoid fever may be very serious.
Before you are given Typhim Vi
When you must not be given it
Do not have Typhim Vi if you have an allergy to:
- Typhim Vi or any of the ingredients listed at the end of this leaflet.
Do not have Typhim Vi if you have, or recently have had, an:
- illness, with fever.
Typhim Vi is not recommended for use in children under 2 years of age. Typhim Vi vaccine is a polysaccharide vaccine: just as with other polysaccharide vaccines, children under 2 years are too young to make sufficient antibodies to this type of vaccine.
Do not have Typhim Vi after the expiry date printed on the pack.
Do not have Typhim Vi if the packaging is torn or shows signs of tampering.
If you are not sure whether you or your child should have Typhim Vi, talk to your doctor or pharmacist.
Before you are given Typhim Vi
Tell your doctor if you have reacted to any previous vaccination with any of the following:
- an allergic reaction. Allergic reactions may include an itchy rash (also known as 'hives'), swelling of lips, face or tongue which may cause difficulty breathing.
Tell your doctor if you have, or have had, any medical conditions, especially the following:
- lowered immunity due to illness. Examples are some blood disorders, HIV/AIDS or cancer
- lowered immunity due to treatment with medicines such as corticosteroids, cyclosporin or other medicines used to treat cancer (including radiation therapy) as you may not make the expected amount of antibody
- if you have a low platelet count or are prone to easy bruising or bleeding.
Tell your doctor if you are pregnant or intend to become pregnant. Your doctor will discuss the possible risks and benefits of having Typhim Vi during pregnancy.
Tell your doctor if you are breast-feeding. It is not known if Typhim Vi passes into breast milk. Your doctor will discuss the possible risks and benefits of having Typhim Vi during breast-feeding.
Taking other medicines
Tell your doctor or pharmacist if you are taking any other medicines, including any that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines and Typhim Vi may interfere with each other.
Your doctor or pharmacist will advise you and decide whether or not to give the vaccine.
Having other vaccines
Your doctor will advise you if Typhim Vi is to be given at the same time as another vaccine.
Your doctor and pharmacist may have more information on medicines and vaccines to be careful with, or avoid, during vaccination with Typhim Vi.
How Typhim Vi is given
Typhim Vi is given as an injection, usually into your upper arm muscle, by a doctor or nurse. For children, the muscle of the upper thigh may be preferred.
Typhim Vi should not be injected directly into a vein, or any blood vessel.
How much is given
The dose is the same for adults and children, 0.5 mL of vaccine.
Overdose
Overdose is unlikely as your doctor is giving you the injection.
If you have any concerns, ask your doctor.
When Typhim Vi is given
Typhim Vi should be given at least 14 days before any risk of coming into contact with typhoid bacteria.
If you live in an area where you are at risk of getting typhoid fever, it is recommended that you have an injection of Typhim Vi every 3 years.
Possible side effects
All medicines, including vaccines, can have side effects. Sometimes they are serious, most of the time they are not. You may need medical treatment if you get some of the side effects.
Tell your doctor or pharmacist as soon as possible if you do not feel well after having Typhim Vi.
Typhim Vi may have unwanted side effects in a few people. Usually they occur at the site of the injection, in the first 48 hours.
Ask your doctor or pharmacist to answer any questions you may have.
Tell your doctor or pharmacist if you notice any of the following and they worry you:
- local reaction around the injection site such as, pain or discomfort, tenderness, redness, or hard lump
- headache
- tiredness, weakness or fatigue
- generally feeling unwell
- decreased activity
- raised temperature
- aching muscles, muscle tenderness or weakness, not caused by exercise
- nausea
- vomiting
- diarrhoea
- stomach pains
- joint pain
- asthma
- fainting
- skin reactions such as rash, itching or itchy rash
If any of the following happen, tell your doctor immediately or go to Accident and Emergency at your nearest hospital:
- sudden signs of allergy such as rash, itching or hives on the skin, swelling of the face, lips, tongue or other parts of the body, shortness of breath, wheezing or difficulty breathing
- allergic reactions, symptoms for which include fever, swelling, skin rash, enlargement of the lymph nodes
These are very serious side effects. You or your child may need urgent medical attention or hospitalisation.
All of these side effects are rare.
Other side effects not listed above may occur in some patients. Tell your doctor or pharmacist if you notice anything that is making you feel unwell.
Do not be alarmed by this list of possible side effects. You may not experience any of them.
Storing Typhim Vi
Typhim Vi is usually stored in the doctor's surgery or clinic, or at the pharmacy. However, if you need to store Typhim Vi:
Keep it where children cannot reach it.
Keep Typhim Vi in the original pack until it is time for it to be given.
Keep it in the refrigerator, between +2°C and +8°C. Do not freeze Typhim Vi. Freezing destroys the vaccine.
Further Information
Typhim Vi is available only with a doctor's prescription.
Ask your doctor if you are unsure about anything or want more information about Typhim Vi.
What Typhim Vi looks like
Each dose of Typhim Vi consists of one syringe pre-filled with 0.5 mL of clear colourless liquid.
What Typhim Vi contains
Each dose contains:
Active ingredients:
- 25 micrograms Salmonella typhi Vi Polysaccharide (Ty2 strain).
Other ingredients:
- phenol
- sodium chloride
- dibasic sodium phosphate dihydrate
- monobasic sodium phosphate dihydrate
- water for injection
- formaldehyde (trace).
- casein (trace)
Typhim Vi does not contain lactose, sucrose, gluten, tartrazine or other azo or non-azo type of dyes.
The manufacture of this product includes exposure to bovine materials. No evidence exists that any case of vCJD (considered to be the human form of bovine spongiform encephalopathy) has resulted from the administration of any vaccine product.
Name and Address of the Sponsor
Australia:
sanofi-aventis australia pty ltd
Talavera Corporate Centre - Building D
12 - 24 Talavera Road
Macquarie Park NSW 2113
Australia
Tel: 1800 818 806
New Zealand:
sanofi-aventis new zealand limited
Level 8
56 Cawley St
Ellerslie
Auckland
New Zealand
Tel: 0800 283 684
Aust R number
Aust R 45073
Date of preparation
July 2020
typhim-ccdsv11-cmiv3-20jul20
™ Trademark of Sanofi Pasteur SA
Published by MIMS September 2020

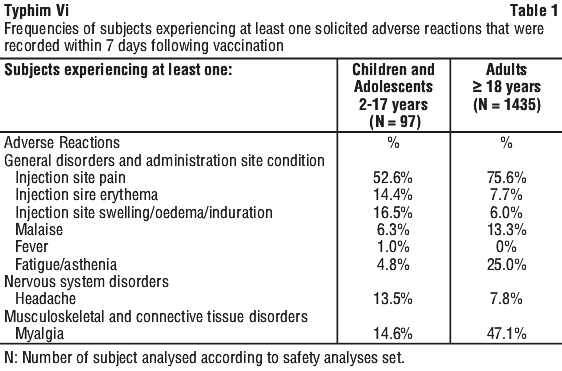 The Table 2 summarises the frequencies of subjects experiencing at least one unsolicited adverse reactions that were recorded within 28 days following vaccination in 1435 adults and 97 children and adolescents from 2 to 17 years of age.
The Table 2 summarises the frequencies of subjects experiencing at least one unsolicited adverse reactions that were recorded within 28 days following vaccination in 1435 adults and 97 children and adolescents from 2 to 17 years of age. In a U.S.
Reimmunisation study, subjects who had received Typhim Vi 27 or 34 months earlier, and subjects who had never previously received a typhoid vaccination, were randomised to placebo or Typhim Vi, in a double blind study. In this study
5/30 (17%) primary immunisation subjects and 10/45 (22%) reimmunisation subjects had an objective local reaction (erythema and/or induration at the site of injection). No severe or unusual side effects were observed. Most subjects
reported pain and/or tenderness (pain upon direct pressure). Local adverse experiences were generally limited to the first 48 hours. Results are summarised in Table 3.
In a U.S.
Reimmunisation study, subjects who had received Typhim Vi 27 or 34 months earlier, and subjects who had never previously received a typhoid vaccination, were randomised to placebo or Typhim Vi, in a double blind study. In this study
5/30 (17%) primary immunisation subjects and 10/45 (22%) reimmunisation subjects had an objective local reaction (erythema and/or induration at the site of injection). No severe or unusual side effects were observed. Most subjects
reported pain and/or tenderness (pain upon direct pressure). Local adverse experiences were generally limited to the first 48 hours. Results are summarised in Table 3.

