1 Name of Medicine
Methenamine hippurate.
2 Qualitative and Quantitative Composition
Each Uramet tablet contains 1 g methenamine hippurate.
For excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
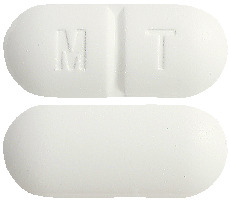
Uramet tablets are white to off-white, capsule-shaped, and embossed with "MT" with a break line on one side and plain on the other.
4.1 Therapeutic Indications
Suppression of bacteriuria associated with chronic or recurrent infection of the urinary tract. May be used for short or long-term therapy, as necessary, such as maintenance therapy after successful initial treatment of acute infections of the urinary tract with antibiotics and as long term therapy in the prevention of recurrent cystitis.
4.2 Dose and Method of Administration
Dosage.
Adult. 1 g (one tablet) twice daily.
Paediatric population. Children under 12 years.
Not recommended.
Older people. No special dosage recommendations.
Method of administration.
The tablets may be halved, or they can be crushed and taken with a drink of milk or fruit juice if the patient prefers.4.3 Contraindications
Methenamine hippurate is contraindicated in individuals with known hypersensitivity to methenamine, or to any other excipients in Uramet tablets (see Section 2 Qualitative and Quantitative Composition).
Methenamine hippurate is contraindicated in patients with hepatic dysfunction, renal parenchymal infection, renal insufficiency, or renal failure, severe dehydration, metabolic acidosis or gout.
4.4 Special Warnings and Precautions for Use
Use in hepatic impairment.
Methenamine hippurate is contraindicated in patients with hepatic impairment (see Section 4.3 Contraindications).
Use in renal impairment.
Methenamine hippurate is contraindicated in patients with renal impairment (see Section 4.3 Contraindications).
Use in the elderly.
See Section 4.2 Dose and Method of Administration.
Paediatric use.
Methenamine hippurate is not recommended for children under 12 years of age.
Effect on laboratory tests.
Methenamine hippurate taken during pregnancy can interfere with laboratory tests of urine oestriol (resulting in unmeasurably low values) when acid hydrolysis is used in the laboratory procedure. This interference is due to the presence of methenamine and/or formaldehyde in the urine. Enzymatic hydrolysis, in place of acid hydrolysis, will circumvent this problem. Methenamine hippurate also causes spuriously elevated urinary 17-hydroxycorticosteroid level due to the presence of formaldehyde in the urine when Porter-Silber method is used. Similarly, formaldehyde also interferes with fluorometric procedures for determination of urinary catecholamine and vanillylmandelic acid (VMA), causing erroneously high results.
Formaldehyde presence in the urine causes falsely decrease 5-hydroxyindole acetic acid (5HIAA) concentrations by inhibiting colour development when nitrosonaphthol methods are used.4.5 Interactions with Other Medicines and Other Forms of Interactions
Uramet should not be administered concurrently with sulphonamides because of the possibility of crystalluria, or with urinary alkalising agents. Concurrent use with acetazolamide should be avoided as the desired effect of hexamine will be lost.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility.
No data available.
(Category A)
There is inadequate evidence of safety of methenamine hippurate in human pregnancy, but it has been in wide use for many years without apparent ill consequence. Animal studies are insufficient with respect to reproductive toxicity. As a precautionary measure, it is preferable to avoid the use of methenamine hippurate during pregnancy.
Methenamine is excreted in low levels in breast milk. No adverse effects on the nursing infant have been reported.4.7 Effects on Ability to Drive and Use Machines
Methenamine hippurate does not have any effects on the ability to drive or operate any machinery.
4.8 Adverse Effects (Undesirable Effects)
Adverse events are listed below by system organ class and frequency.
Frequencies are defined as: very common (≥ 1/l0); common (≥ 1/100 and < 1/10); uncommon (≥ 1/1000 and < 1/100); rare (≥ 1/10,000 and < 1/1000); very rare (< 1/10,000); not known (cannot be estimated from the available data).
Gastrointestinal disorders.
Uncommon: gastric irritation, nausea and vomiting.
Not known: diarrhoea, abdominal pain.
Renal and urinary disorders.
Uncommon: irritation of the bladder and dysuria.
Skin and subcutaneous disorders.
Uncommon: rash, pruritus.
Reporting suspected adverse effects.
Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.4.9 Overdose
For information on the management of overdose, contact the Poisons Information Centre on 131126 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action.
Methenamine Hippurate is a urinary antibacterial agent with a wide antibacterial spectrum covering both gram-positive and gram-negative organisms.
The antibacterial action is obtained by the slow release of the bactericidal formaldehyde, from the methenamine part of the molecule, in the urine.
Clinical trials.
The available clinical data provide evidence of the efficacy and safety of methenamine hippurate in the preventative treatment of bacteriuria and recurrent urinary tract infections (UTI) in adults when used in a dose of 1 g up to three times daily and for up to 1 year.
The authors of a Cochrane review of methenamine hippurate for prevention of urinary tract infections (Lee et al, 2012) concluded that methenamine hippurate may be effective for preventing UTI in patients without renal tract abnormalities, particularly when used for short term prophylaxis, but that it does not appear to work in patients with neuropathic bladder or in patients who have renal tract abnormalities.
In a randomised, double blind, long term, crossover study (Cronberg et al, 1987) 1 g twice daily of methenamine hippurate was compared with placebo for its preventive effect on recurrent attacks of acute cystitis. Methenamine hippurate and placebo were interchanged every six months for two years. Out of 21 enrolled patients, 14 completed the first year and 13 both years of treatment, which permitted the evaluation of 27 patient years. There were 52 episodes of acute cystitis caused by reinfection: 41 occurred during placebo treatment and only 11 during the methenamine hippurate regimen (p < 0.01).
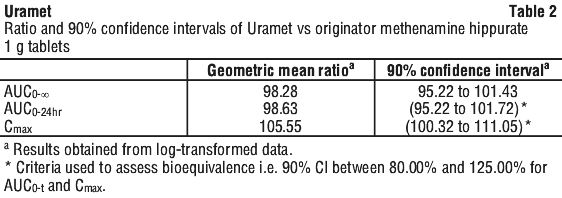
Methenamine hippurate 1 g tablets have been marketed in Australia for over 3 decades. A randomised, open label, two-period, two sequence, single dose, crossover, bioequivalence study of Uramet 1 g tablets and the originator methenamine hippurate 1 g tablets was conducted in healthy subjects under fasting conditions. The pharmacokinetic results demonstrated both products to be bioequivalent.
The primary pharmacokinetic parameters analysed were AUC0-24 and Cmax. The key secondary pharmacokinetic parameters analysed were AUC0-∞, Tmax and t1/2. The assessment of bioequivalence was based on the 90% confidence interval of the relative mean AUC0-24 and Cmax of these two products to be between 80.00% and 125.00% for log-transformed data.
The summary of the pharmacokinetic parameters for Uramet is shown in Tables 1 and 2.

5.2 Pharmacokinetic Properties
Some hydrolysis of oral methenamine occurs in the acid environment of the stomach. Methenamine hippurate is readily absorbed from the gastro-intestinal tract and distributes widely into body fluids, but very little is hydrolysed prior to excretion in the kidney and thus has minimal systemic toxic potential. Within one-half hour after a single 1 g dose of methenamine hippurate, anti-bacterial activity is demonstrable in the urine. Plasma concentrations of methenamine hippurate reach maximum 1 - 2 hours after a single dose and then decline with a half-life of about 4 hours. Methenamine recovered in the urine corresponds to about 80% of the dose given per 12 hours. Similarly, the hippurate moiety is rapidly absorbed and excreted, and it reaches the urine by both tubular secretion and glomerular filtration. This may be of importance in older patients or those with some degree of renal impairment.
5.3 Preclinical Safety Data
Genotoxicity.
No data available.
Carcinogenicity.
Methenamine did not demonstrate any carcinogenic potential in long term studies in rodents.6 Pharmaceutical Particulars
6.1 List of Excipients
Each Uramet tablet also contains the following excipients: povidone (K30), colloidal anhydrous silica and magnesium stearate.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Stored below 25°C, in a dry place.
6.5 Nature and Contents of Container
The tablets are available in amber glass bottles of 20, 25 and 100*.
*Note: not all pack sizes are marketed in Australia.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
The chemical name for methenamine hippurate is 2-benzamidoacetic acid;1,3,5,7-tetrazatricyclo[3.3.1.1]decane. The molecular weight is 319.36.
It is a white to off-white odourless crystalline powder. It is soluble in water, ethanol and chloroform. The pKa of methenamine hippurate at 25°C is 3.59.
Chemical structure.
 C15H21N5O3.
C15H21N5O3.
CAS.
5714-73-8.References
Cronberg S, Welin CO, Henriksson L, Hellsten S, Persson KM, Stenberg P. Prevention of recurrent acute cystitis by methenamine hippurate: double blind controlled crossover long term study. Br Med J (Clin Res Ed). 1987 Jun 13;294(6586):1507-8.
Lee BS, Bhuta T, Simpson JM, Craig JC. Methenamine hippurate for preventing urinary tract infections. Cochrane Database Syst Rev. 2012 Oct 17;10: CD003265.
7 Medicine Schedule (Poisons Standard)
S3 - Pharmacist only medicine.
Summary Table of Changes




 C15H21N5O3.
C15H21N5O3.